AP Syllabus focus: ‘Knowing the energetics of each elementary step in a mechanism allows you to construct an energy profile for a multistep reaction.’
Multistep reactions proceed through several elementary steps, each with its own energy change and barrier. If you know the energetics of each step, you can sketch the full energy profile consistently and interpret what it implies about the pathway.
What a multistep energy profile represents
Multistep reaction energy profile: a plot of potential energy versus reaction coordinate that shows the energy changes and energy barriers for each elementary step along an overall reaction pathway.
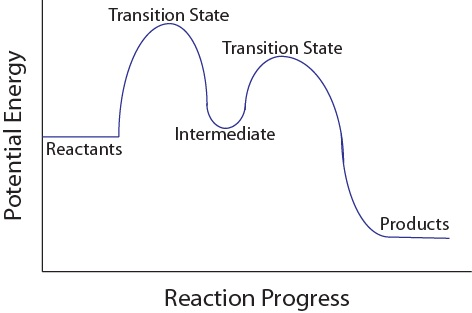
A two-step reaction coordinate diagram showing reactants and products at different potential energies, with two labeled transition states (peaks) and an intermediate (local minimum) between them. This makes the “one hump per elementary step” idea explicit and helps you see how intermediates correspond to minima on the energy profile. Source
In this subtopic, the key skill is building the qualitative shape of that plot from step energetics: the energy change for each step and the barrier height for each step.
Energetic quantities you may be given
To construct the profile, you typically need (in words, a diagram, or data):
The relative energies of reactants and products (overall energy change)
For each step:
Whether it is exothermic (energy goes down) or endothermic (energy goes up)
The activation energy barrier height for that step (how high the “hump” is above the step’s starting point)
Building the profile from step energetics (process)
Step 1: Set a baseline and choose an energy scale
Draw axes: potential energy (vertical) vs reaction coordinate (horizontal).
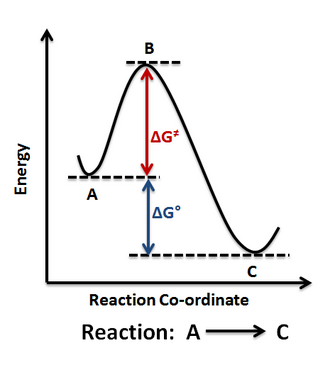
A basic reaction coordinate diagram for a one-step reaction, showing reactants rising to a transition state and then descending to products. It provides a reference picture for identifying activation energy as the vertical gap from a minimum to the peak, and overall energy change as the difference between the reactant and product energy levels. Source
Place the reactants at a chosen reference energy (often arbitrary, e.g., 0 kJ/mol), since only relative energies matter.
Step 2: Add step-by-step energy changes (the “stairs”)
For each elementary step, use the sign and magnitude of the step energy change to locate the next minimum relative to the previous one:
If step 1 is exothermic by a certain amount, the next minimum must be that amount lower than the reactants.
If step 2 is endothermic, the next minimum must be that amount higher than the previous minimum.
Continue cumulatively so that each minimum is consistent with the running total of energy changes across steps.
This cumulative bookkeeping ensures the final products land at the correct overall energy relative to reactants.
= overall energy change from reactants to products (kJ/mol or similar)
= energy change for elementary step (kJ/mol or similar)
= number of elementary steps (unitless)
In practice, you can use this relationship as a consistency check: the sum of the step energy changes must match the overall energy change shown by the endpoints of your profile.
Step 3: Add barriers for each step (the “humps”)
Each step has a barrier that rises above the step’s starting minimum:
For step 1, draw a peak that is Ea₁ above the reactant energy level, then descend to the next minimum set by step 1’s energy change.
For step 2, start from the step 2 minimum, rise by Ea₂, then drop to the next minimum, and so on.
Key construction rule: barriers are measured from the local minimum immediately before that step, not necessarily from the original reactants.
Step 4: Check for energetic realism and internal consistency
A correctly constructed multistep profile should satisfy:
The number of humps equals the number of elementary steps.
The final product energy matches the cumulative result of the step energy changes.
Large endothermic steps place subsequent minima higher; large exothermic steps place them lower.
If two steps have very different barriers, the taller hump must visibly be the larger barrier relative to its own starting minimum.
Common construction pitfalls (and how to avoid them)
Mixing up “overall” vs “step” quantities
Do not place products using only the largest step’s energy change; use the sum of all step energy changes.
Do not measure every barrier from the original reactants; measure each from the preceding minimum.
Confusing magnitude with direction
Exothermic step: minimum goes down.
Endothermic step: minimum goes up.
A step can be endothermic but still have a small or large barrier; these are independent pieces of information that both must appear in the sketch.
What your constructed profile can immediately communicate
Without invoking any extra mechanism details, a multistep profile built from step energetics can show:
Whether the overall reaction is net exothermic or net endothermic
Which step has the largest energy barrier (visually the tallest hump above its preceding minimum)
Whether energy is released/absorbed early or late in the pathway based on the sequence of minima
FAQ
Use differences between consecutive species energies as the step energy changes.
Then add a peak between each consecutive pair using each step’s stated barrier (if provided).
Yes.
Activation energy is measured from the local minimum before that step; a higher starting minimum can make a smaller absolute peak still correspond to a larger barrier relative to that minimum.
Treat it as inconsistent data.
On exams, this usually signals an error in interpretation (e.g., wrong sign) or a misread reference point for energies.
The drop from that step’s peak to the next minimum, and/or the depth of that next minimum relative to the previous minimum.
It does not, by itself, fix the barrier height.
Keep the sketch qualitative but consistent:
larger changes drawn as larger vertical shifts
larger barriers drawn as taller humps (relative to the correct local minimum)
Practice Questions
(2 marks) A mechanism has two elementary steps. Step 1 is exothermic by 20 kJ mol and Step 2 is endothermic by 5 kJ mol. State the overall energy change and whether products are above or below reactants on the energy profile.
Overall energy change kJ mol (1)
Products below reactants / overall exothermic (1)
(5 marks) A three-step mechanism has step energy changes of , , and kJ mol. The activation energies relative to each step’s starting point are 40, 15, and 35 kJ mol, respectively. Describe how to construct a consistent multistep energy profile and state the relative energy of products compared with reactants.
Reactants set at a reference energy; axes labelled (1)
Successive minima placed using cumulative step changes: after step 1 down 10; after step 2 up 25 from that; after step 3 down 30 from that (1)
Three peaks drawn; each peak height measured above its own preceding minimum (1)
Peak heights correspond to 40, 15, 35 kJ mol in steps 1–3 (1)
Products relative to reactants: kJ mol overall; products below reactants (1)

