AP Syllabus focus: ‘A multistep energy profile can show multiple transition states and intermediates, based on the energy changes of each elementary step.’
Multistep reactions rarely proceed in a single leap from reactants to products.
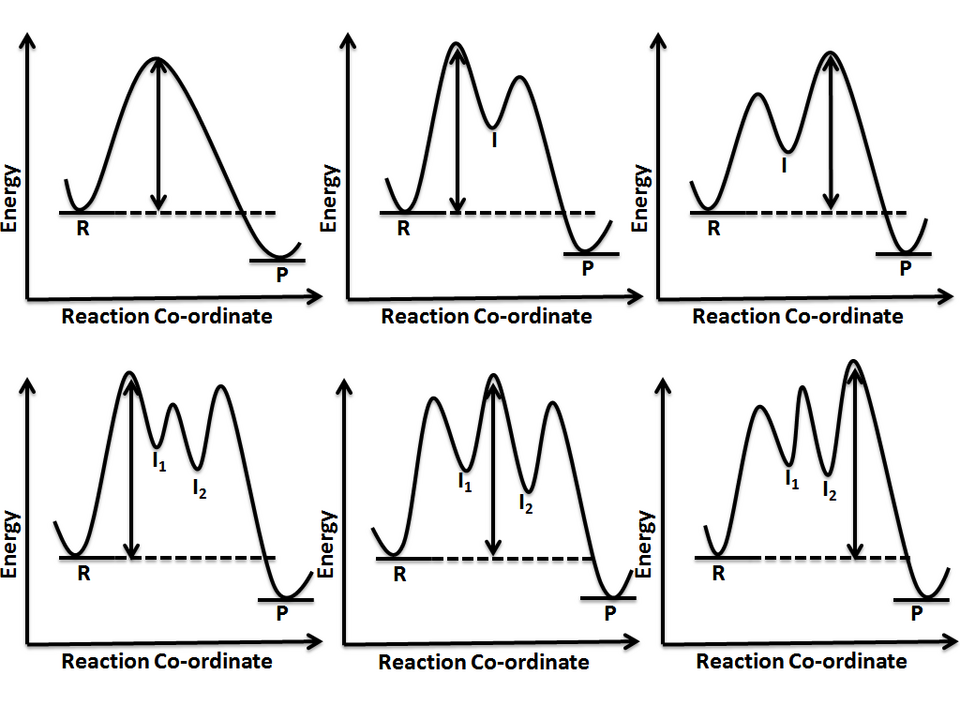
Reaction coordinate diagrams comparing pathways with 0, 1, and 2 intermediates. Each additional intermediate adds an extra valley (a local minimum) and therefore requires an additional transition state (a peak) between reactants and products. This makes the “number of steps = number of peaks” and “number of intermediates = number of valleys between peaks” rules visually explicit. Source
Energy profiles for such mechanisms reveal distinct peaks and valleys that correspond to transition states and intermediates created and consumed along the reaction pathway.
Multistep reaction energy profiles: what you are looking at
An energy profile plots potential energy (often enthalpy-like) versus the reaction coordinate (progress along the mechanism). For multistep mechanisms, the curve is not a single hump; it alternates between:
Peaks (high-energy configurations) and
Valleys (relatively stable species that can persist briefly)
Transition states appear as peaks
Transition state: A short-lived, highest-energy arrangement of atoms at the top of an energy barrier for an elementary step; it is not isolable and is represented by a peak on an energy profile.
Each elementary step has its own transition state, so a multistep profile can show multiple peaks, one per step. Key implications:
A peak corresponds to a bond-breaking/bond-forming event in that step.
The height of a peak relative to the preceding valley represents the energy barrier that must be overcome to proceed to the next species.
Transition states are sometimes labelled with a double dagger notation in mechanism diagrams (conceptually), but on an energy profile they are identified by their position at local maxima.
Between definition blocks, focus on what the curve is telling you: the reaction coordinate is not time, and the peaks are not “stages you can collect,” but energetic bottlenecks the system must pass through.
Intermediates appear as valleys
Intermediate: A species formed in one elementary step and consumed in a later step; it corresponds to a local minimum (valley) between two transition states on a multistep energy profile.
Intermediates are drawn at local minima because they are more stable than the neighbouring transition states, even if they are higher in energy than the original reactants.
On a multistep profile:
The presence of a valley between two peaks indicates an intermediate exists between those two steps.
Intermediates can be higher or lower in energy than reactants/products; the defining feature is being a minimum between maxima, not its absolute energy.
The depth of the valley reflects relative stability: a deeper minimum indicates a more stable intermediate (lower energy).
Mapping steps to features (peaks and valleys)
To connect a multistep energy profile to a proposed mechanism, use a consistent “counting” logic:
Number of steps in the mechanism = number of peaks (transition states).
Number of intermediates = number of valleys between peaks.
Reactants appear at the far left baseline; products appear at the far right baseline.
Reading energy changes step-by-step
Each elementary step has an associated energy change, which is reflected by the vertical difference between:
The energy of the species at the start of that step (reactant or intermediate) and
The energy of the species at the end of that step (intermediate or product)
This is how the syllabus statement applies: the profile’s shape (multiple transition states and intermediates) is determined by the energy changes of each elementary step, stitched together in sequence.
Common interpretation pitfalls (what to avoid)
Do not label a peak as an intermediate: intermediates are minima, not maxima.
Do not assume the “highest point overall” must be the only transition state; multistep profiles can have several peaks of different heights.
Do not read the horizontal spacing as time or rate; the reaction coordinate is a conceptual progress variable, not a clock.
Do not require intermediates to be detectable experimentally; the profile indicates they are mechanistically meaningful minima, regardless of measurability.
FAQ
“Minimum” is local, not absolute.
An intermediate is lower in energy than the two neighbouring transition states, so it sits in a valley even if the whole valley is above the reactant energy level.
Look for curvature and relative position.
A transition state is a local maximum (slope changes from + to −).
An intermediate is a local minimum (slope changes from − to +).
If the curve never turns downward then upward, it is not a true valley.
No.
Some intermediates are too reactive or too short-lived to isolate, even if the energy profile shows a local minimum. The profile indicates mechanistic stability relative to transition states, not laboratory stability.
It suggests a very unstable intermediate.
The intermediate is only slightly more stable than the transition state, so it may not accumulate appreciably and may be difficult to distinguish experimentally from a concerted-looking process.
The coordinate is an abstract progress variable.
Different bond changes and rearrangements do not proceed at a uniform pace, so equal horizontal distances do not correspond to equal time intervals; the axis only orders events along the mechanism.
Practice Questions
A multistep energy profile shows two peaks separated by one valley. Identify what the peaks and the valley represent, using correct kinetic terminology.
Peaks represent transition states (1)
Valley represents an intermediate (1)
Statement that the intermediate is formed then consumed / lies between two steps (1)
A reaction energy profile has three peaks and two valleys between reactants and products.
(a) State how many elementary steps are implied and justify your answer. (2 marks)
(b) State how many intermediates are implied and justify your answer. (2 marks)
(c) Explain how the energy changes of individual steps are reflected in the relative energies of valleys and adjacent parts of the curve. (2 marks)
(a) Three steps (1); justified because three peaks correspond to three transition states, one per step (1)
(b) Two intermediates (1); justified because two valleys between peaks correspond to intermediates (1)
(c) Step energy change described as vertical difference between start/end species of that step (1); clear link to valleys rising/falling relative to neighbouring minima/maxima depending on whether a step is uphill/downhill in energy (1)

