AP Syllabus focus: ‘Comparing barrier heights on a multistep profile helps identify the step with the largest activation energy, which often corresponds to the slowest step.’
Multistep energy profiles provide a visual way to connect thermodynamic “ups and downs” to kinetics. By comparing the relative heights of the barriers between intermediates and transition states, you can often identify which step most strongly limits the overall reaction rate.
Interpreting “barriers” on a multistep energy profile
A multistep reaction energy profile shows several peaks (transition states) and valleys (reactants/intermediates/products).
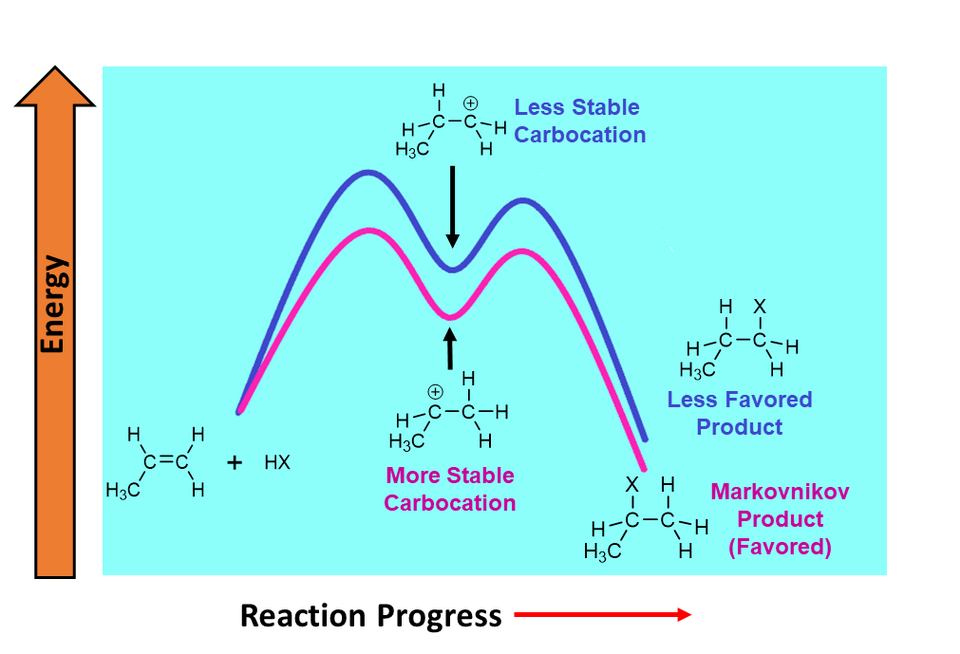
A two-step reaction coordinate diagram showing reactants, an intermediate, and two transition states (two peaks). The key kinetic comparison is the vertical gap from each valley to the next peak (each step’s ), not the absolute height of a peak relative to the original reactants. The step with the larger valley-to-peak barrier is typically the slow (rate-determining) step. Source
Each elementary step has its own activation barrier measured from its starting valley to its peak.
Activation energy (): The energy difference between a step’s starting species (reactant or intermediate) and that step’s transition state; it is the kinetic barrier that must be overcome for that step to proceed.
When the syllabus says “compare barrier heights,” it means comparing the vertical energy gaps for each step, not simply comparing absolute peak heights measured from the original reactants.
How to compare barriers correctly
Identify each local minimum (reactants or intermediates).
For each step, locate the next transition state peak immediately “ahead” along the reaction coordinate.
Compute (conceptually) each step’s barrier as: peak height minus the preceding valley height.
The largest barrier (largest ) is the step that is usually the slowest.
Why the largest barrier usually corresponds to the slow step
A higher activation barrier means a smaller fraction of collisions/encounters have enough energy to reach the transition state at a given temperature, so that step proceeds less frequently per unit time.
Slow step (rate-determining step): The step in a multistep process that proceeds at the lowest rate under given conditions, thereby limiting how fast product can form overall.
This connection is often justified by the Arrhenius relationship between rate constant and activation energy.
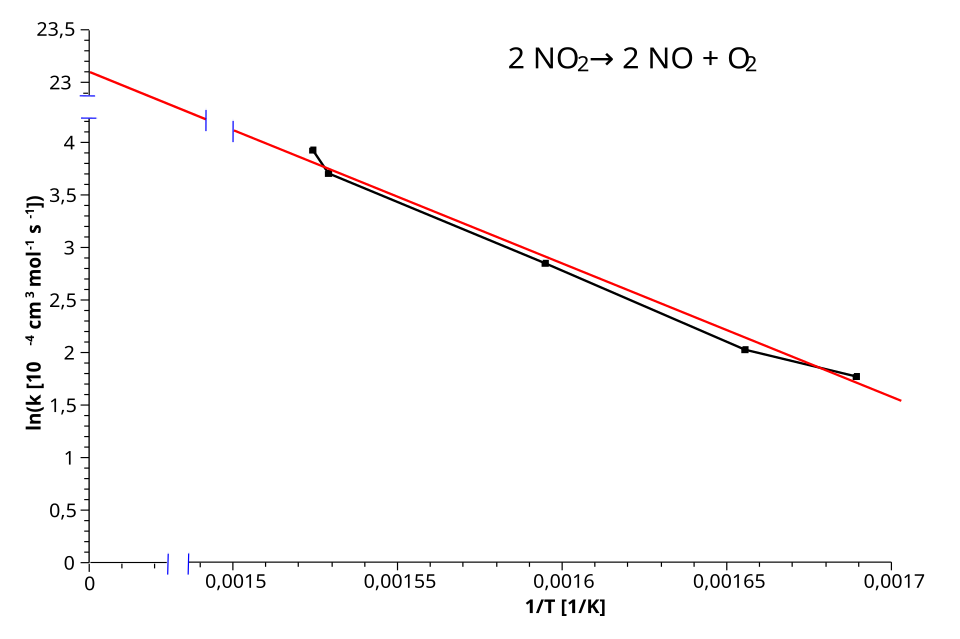
An Arrhenius plot of versus illustrating the linear form of the Arrhenius equation. The line’s slope is proportional to , so a larger activation energy corresponds to a steeper negative slope and a stronger temperature dependence of the rate constant. The intercept relates to the pre-exponential factor . Source
= rate constant for a given step (units depend on that step’s molecularity)
= activation energy for that step (J mol)
= absolute temperature (K)
Even without calculation, the key idea is: larger ⇒ exponentially smaller ⇒ slower step, so the largest barrier commonly controls the overall timescale.
Connecting the “largest barrier” to what you observe experimentally
In many multistep profiles:
The highest barrier corresponds to the smallest step rate constant.
Intermediates before that barrier may accumulate relative to others, because they form faster than they are consumed.
The overall reaction appears to “wait” for that difficult barrier-crossing event.
Forward vs reverse direction matters
Barrier comparisons must be made in the direction of interest:
The forward slow step is linked to the largest forward barrier (from each valley to the next peak).
If you considered the reverse reaction, you would compare barriers from products/intermediates toward reactants; the largest reverse barrier might be different.
Common pitfalls when using energy profiles to identify the slow step
Comparing absolute peak heights instead of valley-to-peak gaps for each step.
Assuming the tallest peak from the baseline reactants is always rate-limiting, even when there are high-energy intermediates later.
Ignoring that a step can be “large barrier” yet not control the observed rate if it is not on the dominant pathway or if conditions change (temperature can change the relative importance of barriers).
Confusing overall energy change (reactants vs products) with kinetics; being more exothermic or endothermic does not, by itself, determine which step is slow.
FAQ
They may be comparably rate-limiting under the same conditions.
In practice, small differences in the pre-exponential factor $A$, molecularity, or reactant availability can break the tie, so experimental kinetics would be needed to distinguish them.
Because barriers are measured from the preceding valley, not from the original reactants.
A high-energy intermediate raises the “starting point” for a later step, reducing that step’s valley-to-peak gap even if the peak is high in absolute terms.
Raising $T$ increases the fraction of molecules with sufficient energy to cross any barrier.
Because of the exponential dependence in $e^{-E_a/(RT)}$, higher-$E_a$ steps are typically sped up proportionally more, which can sometimes shift which step is effectively slowest.
You may observe a transient increase (then decrease) in the concentration or signal of an intermediate.
This can happen when the intermediate forms quickly but is consumed slowly across the large barrier.
Yes. Stabilising or destabilising a transition state or intermediate changes the relative energies on the profile.
For example, a polar solvent may lower the energy of a charged transition state more than a neutral intermediate, reducing that step’s effective barrier.
Practice Questions
(2 marks) A reaction energy profile has three steps. Step 1 has a small barrier, Step 2 has the largest barrier, and Step 3 has a medium barrier. Which step most likely limits the overall rate, and why?
Identifies Step 2 as the slow/rate-determining step (1).
Explains that the largest activation barrier corresponds to the smallest (or lowest fraction reaching the transition state), so it is slowest (1).
(5 marks) A multistep profile shows valleys at energies 10, 25, and 5 kJ mol, with subsequent peaks at 40, 55, and 30 kJ mol (forward direction).
(a) Rank the forward activation barriers for Steps 1–3 from largest to smallest. (3 marks)
(b) State which step is most likely the slow step and give a kinetic reason. (2 marks)
(a) Correct barriers: Step 1 , Step 2 , Step 3 kJ mol (any clear working implied) (2).
(a) Correct ranking: Step 1 = Step 2 > Step 3 (1).
(b) States Step 1 or Step 2 (tie) is most likely slow (1).
(b) Links larger to smaller via (conceptually) (1).

