AP Syllabus focus: ‘Some catalysts bind reactants to orient them more favorably or lower activation energy, creating a new catalyst‑bound intermediate; many enzymes work this way.’
Enzymes are biological catalysts that speed reactions mainly by binding reactants, positioning them precisely, and stabilising high-energy arrangements. This binding creates a short-lived enzyme–reactant complex and reduces the activation energy barrier.
Catalysis by Binding and Orientation: Core Idea
Many catalysts (especially enzymes) increase reaction rate by forming temporary, specific interactions with reactants. These interactions can:
Orient reactant particles so the correct atoms collide
Lower activation energy by stabilising the transition-state-like arrangement
Create a new catalyst-bound intermediate (an enzyme–substrate complex) that follows an alternative, faster pathway
Key Terms
Enzymes work through a specialised binding region.
Active site: the specific region of an enzyme where reactant molecules bind and where catalysis occurs.
Binding is selective and depends on molecular shape and intermolecular forces.
Substrate: a reactant molecule that binds to an enzyme’s active site and is converted into product.
Between these definitions, remember that enzyme catalysis is about how binding changes the likelihood of productive bond breaking/forming, not about changing the overall reaction stoichiometry.
How Binding Speeds Up Reactions
1) Orientation and Proximity Effects
For many reactions, particles collide often but rarely with the correct geometry. Enzymes improve the odds of an effective collision by:
Holding substrates close together (increased effective concentration)
Aligning functional groups so bonds can form/break along the correct pathway
Restricting rotational/translational freedom, reducing “wasted” collision angles
This means more collisions become productive without requiring higher temperature or higher bulk concentration.
2) Lowering Activation Energy by Stabilising Key Arrangements
When substrates bind, the enzyme can preferentially stabilise an arrangement that resembles the transition state. This decreases the energy barrier for reaction. Common stabilising interactions include:
Hydrogen bonding to strained or partially charged bonds
Ion–dipole attractions to developing charges
Dipole-induced dipole interactions that help polarise bonds
Temporary distortion (“strain”) that makes certain bonds easier to break
The crucial AP Chemistry takeaway is that binding can create a new pathway with a smaller activation energy while leaving the overall energy change of reaction unchanged.
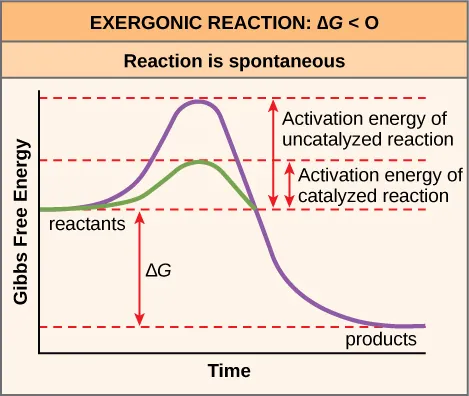
Reaction-coordinate diagram comparing catalyzed and uncatalyzed pathways. The catalyzed pathway has a lower activation energy peak (), which increases the reaction rate, while the relative energies of reactants and products remain the same. This visual reinforces that catalysts change the pathway, not the reaction’s overall energy change. Source
Catalyst-Bound Intermediates in Enzyme Mechanisms
Enzymes often form a transient, bound species during the reaction.
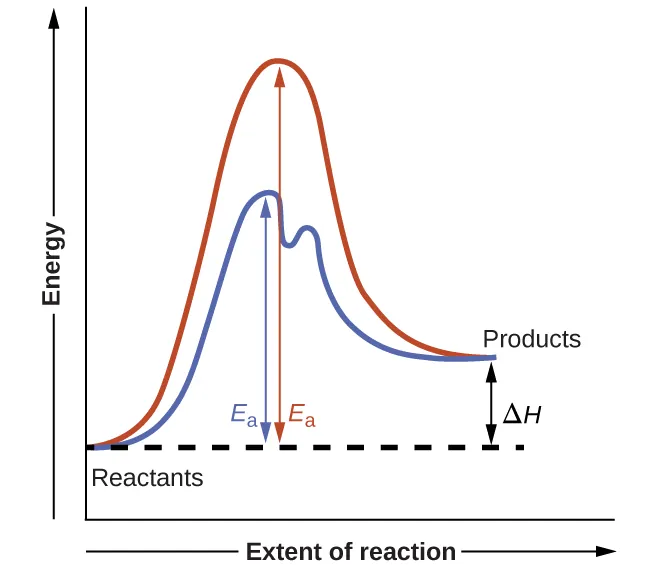
Energy diagrams comparing uncatalyzed and catalyzed reaction pathways, showing how catalysis can introduce an intermediate and multiple smaller barriers. The lower highest barrier corresponds to a faster rate-determining step. This supports the idea that enzyme mechanisms often proceed through short-lived intermediates rather than a single high-barrier step. Source
Enzyme–substrate complex: a catalyst-bound intermediate formed when substrate(s) bind to an enzyme prior to product formation.
A useful way to visualise the process is:
Substrate(s) bind to the active site to form the enzyme–substrate complex
Bonds are rearranged within the bound complex
Product(s) leave the active site, freeing the enzyme to bind again
Because the intermediate exists only while the reaction is occurring, its formation helps explain how binding and orientation can be the central rate-boosting step.
Specificity and the “Induced Fit” Idea
Enzyme active sites are shape- and chemistry-specific, so only certain substrates bind strongly in the correct orientation. Many enzymes also undergo a small conformational change upon binding.
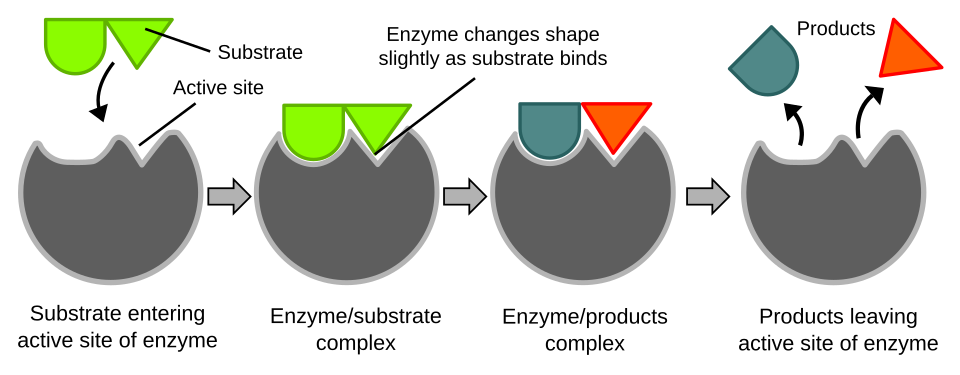
Diagram illustrating the induced-fit model of enzyme catalysis. The active site changes shape after substrate binding, improving geometric complementarity and positioning key groups for reaction. This helps connect “specific binding” to increased probability of productive bond rearrangement. Source
Induced fit: a model in which substrate binding causes the enzyme’s active site to adjust shape, improving alignment and catalytic interactions.
Induced fit helps explain why binding can both:
Improve orientation for bond rearrangement
Enhance transition-state stabilisation, lowering activation energy
What Enzymes Do Not Do
To stay conceptually precise:
Enzymes do not change the overall balanced equation of the reaction
Enzymes do not change the reaction’s overall energy change (they change the pathway, not reactant/product energies)
Strong binding to the substrate alone is not enough; effective catalysis typically involves best stabilisation of the high-energy arrangement that leads to products
FAQ
Allosteric molecules bind away from the active site and shift the enzyme’s shape.
This can subtly change active-site geometry, improving or worsening substrate orientation and the strength/placement of catalytic interactions.
If the enzyme binds the substrate too strongly, it can stabilise the starting state more than the transition-state-like arrangement.
That can increase the effective barrier to reach product, and product release may also become slow.
Some enzymes use non-protein helpers that extend the chemistry of the active site.
They can provide extra binding contacts, enforce substrate positioning, or supply functional groups that assist charge stabilisation during bond rearrangement.
pH can change the protonation state of active-site residues.
That can alter hydrogen-bond patterns and electrostatic attractions, changing how well substrates bind and whether they adopt the correct reactive orientation.
One approach is using transition-state analogue molecules that resemble the high-energy arrangement.
If an enzyme binds such analogues extremely tightly compared with substrates, it supports the idea that the enzyme is optimised to stabilise transition-state-like structures.
Practice Questions
(2 marks) Explain how an enzyme can increase reaction rate by binding reactants in its active site.
1 mark: Binding brings reactants close together and/or in higher effective concentration.
1 mark: Binding orients reactants correctly (or stabilises the transition-state-like arrangement) so more collisions lead to reaction / lower .
(5 marks) An enzyme has an active site that forms hydrogen bonds with a substrate during reaction. Describe how this can increase the rate, referring to orientation and formation of a catalyst-bound intermediate.
1 mark: Substrate binds at the active site forming an enzyme–substrate complex (catalyst-bound intermediate).
1 mark: Binding positions/orients key functional groups to favour productive bond breaking/forming.
1 mark: Hydrogen bonding stabilises developing charges/strained bonds in the high-energy arrangement.
1 mark: Stabilisation lowers the activation energy barrier () for the pathway.
1 mark: Product leaves and the enzyme is available to catalyse another cycle.

