AP Syllabus focus: ‘A catalyst speeds a reaction by increasing effective collisions and/or providing an alternate pathway with lower activation energy than the uncatalyzed reaction.’
Catalysts are central to kinetics because they change how quickly reactions occur without changing what the reaction ultimately produces at equilibrium. Understanding catalytic speed-up requires connecting molecular-level collisions to energy barriers along a reaction pathway.
What a catalyst is (kinetics perspective)
Catalyst: A substance that increases reaction rate by changing the reaction pathway and is regenerated by the end of the reaction (not used up overall).
A catalyst changes how reactants become products (the pathway), not the overall balanced equation. In kinetics language, catalysts increase rate by increasing the number of successful (effective) molecular events per unit time.
How catalysts speed up reactions
Catalysts speed up reactions by one or both of the following syllabus-aligned ideas: increasing effective collisions and/or providing an alternate pathway with lower activation energy.
Increasing effective collisions
A collision only leads to reaction if it is “effective,” meaning it results in productive bond breaking/forming. A catalyst can make a larger fraction of encounters effective by:
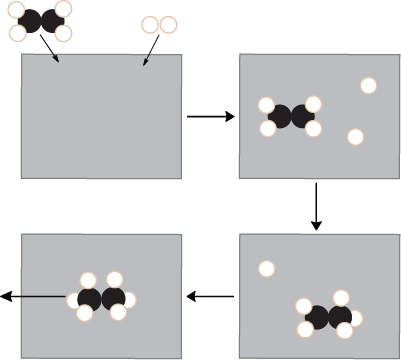
Schematic of heterogeneous (surface) catalysis showing reactants adsorbing to a metal surface, reacting while held close together, and then desorbing as products. By concentrating and orienting reactants at the surface, the catalyst increases the number of effective collisions per unit time. Source
Bringing reactants together in close proximity, increasing the frequency of productive encounters
Positioning reactants so the reactive parts of molecules meet in a bond-making/bond-breaking arrangement
Weakening key bonds in reactants during the encounter, making it easier to start the transformation
Stabilising developing charges or partial bonds during the encounter, helping the system progress toward products
All of these increase the number of productive events per second, so the measured reaction rate increases.
Providing an alternate pathway with lower activation energy
Most reactions do not occur instantly because they must pass through a high-energy configuration on the way to products.
Activation energy (): The minimum energy barrier that must be overcome for reactants to be converted into products along a given pathway.
A catalyst provides an alternate mechanism (often involving additional steps) whose highest energy barrier is smaller than that of the uncatalyzed pathway.
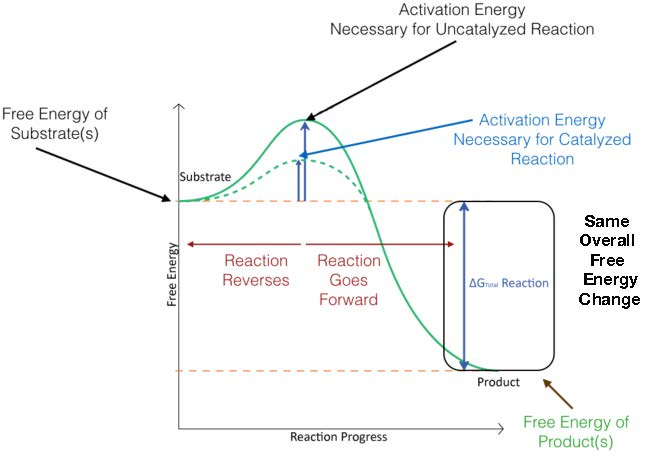
Reaction-coordinate (free energy) diagram comparing uncatalyzed and catalyzed pathways. The catalyzed pathway has a smaller activation-energy barrier while the overall free-energy change () between reactants and products remains the same, illustrating why catalysts speed reactions without changing equilibrium. Source
The key idea is that rate depends strongly on the barrier height, so reducing increases rate at the same temperature.
= activation energy barrier for the stated pathway (kJ mol)
Because the barrier is lower, a greater fraction of reacting particles can successfully reach the critical high-energy arrangement needed to proceed to products, increasing the observed reaction rate.
What catalysts do not change
Catalysts are often confused with factors that change the driving force of a reaction. For AP Chemistry kinetics, keep these straight:
A catalyst does not change the overall reaction stoichiometry
A catalyst does not change the equilibrium position; it speeds up both forward and reverse reactions by lowering the barrier for each pathway
A catalyst does not change the overall energy difference between reactants and products; it changes the path taken between them
Practical implications for rate observations
When a catalyst is present, common experimental signs include:
The reaction reaches its final equilibrium state faster
A plot of energy vs. reaction progress (conceptually) would show a lower peak for the catalyzed route than the uncatalyzed route
The rate increase is explained by either (or both):
more effective collisions, and/or
a lower alternate pathway
FAQ
They compare multiple kinetic signatures across conditions.
Orientation effects often depend strongly on molecular structure/sterics.
Lower $E_a$ effects show a stronger temperature sensitivity of the rate enhancement.
Catalysis depends on how well the catalyst stabilises the specific bond changes needed.
Small changes in reactant shape, polarity, or charge distribution can prevent key stabilising interactions in the catalytic pathway.
Poisoning occurs when another substance binds to the catalyst more strongly than the reactants.
This blocks catalytic sites or interrupts the catalytic pathway, lowering the number of productive catalytic events per unit time.
Yes. A catalyst can favour a pathway with a lower barrier leading to one product over another.
This changes product distribution by altering relative pathway rates, even when overall thermodynamics would allow multiple products.
Catalysts can deactivate without being used up stoichiometrically, for example by:
aggregation/clumping that reduces accessible catalytic area
irreversible binding of impurities
structural changes that disrupt the active form
Practice Questions
(2 marks) State two ways a catalyst can increase the rate of a reaction, as described by collision ideas and energy barriers.
Any one statement about increasing the number/fraction of effective collisions (1)
Any one statement about providing an alternative pathway with lower activation energy, (1)
(5 marks) A reaction is run once without a catalyst and once with a catalyst at the same temperature. The catalysed reaction proceeds faster, but the same equilibrium mixture is obtained in both cases. Explain this observation in terms of , pathway, and forward/reverse reaction rates.
Catalyst provides an alternative reaction pathway/mechanism (1)
Catalysed pathway has lower activation energy, , than uncatalysed (1)
Lower means a greater fraction of reacting particles can react per unit time at the same temperature (1)
Catalyst increases both forward and reverse rates (1)
Therefore equilibrium position/constant is unchanged; it is reached more quickly (1)

