AP Syllabus focus: ‘Some catalysts form covalent bonds with reactants; acid–base catalysis is a common example where species gain or lose a proton, producing new intermediates and steps.’
Catalysts can speed reactions by changing the pathway, not the overall chemical change. In covalent and acid–base catalysis, the catalyst participates directly, creating new intermediates and steps that make the slow process faster.
Core idea: alternate pathways via new intermediates
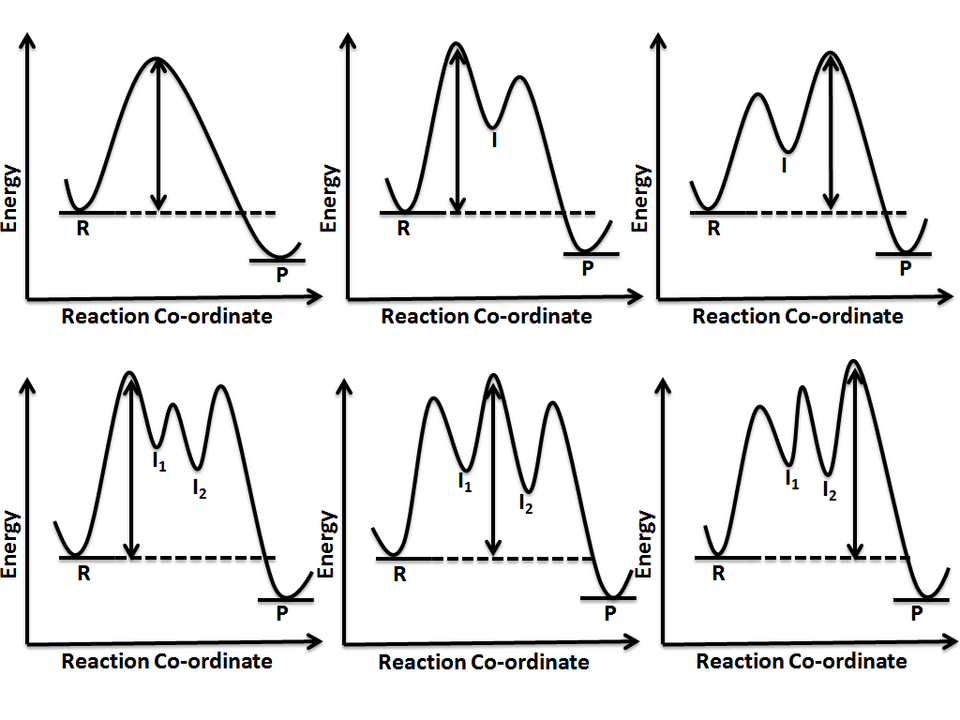
Reaction-coordinate (energy vs. reaction progress) diagrams for mechanisms with 0, 1, or 2 intermediates. The figure emphasizes that adding intermediates splits one large barrier into multiple smaller barriers, so the rate-determining step corresponds to the tallest transition-state peak. This helps visualize how a catalytic mechanism can be faster even though the overall reactant-to-product change is the same. Source
In both catalytic modes, the catalyst becomes part of the mechanism for a time, which can:
Create a more reactive intermediate
Stabilize developing charge
Avoid an especially difficult bond-breaking/bond-making event in a single step
Allow the reaction to proceed through multiple lower-barrier steps
A key check in any proposed catalytic mechanism is that the catalyst is regenerated (it appears in the first steps but is re-formed later).
Covalent catalysis
Covalent catalysis speeds a reaction by forming a temporary covalent bond between the catalyst and a reactant, producing a catalyst-bound intermediate that reacts more readily than the original reactant.
Covalent catalysis: catalysis in which the catalyst forms a temporary covalent bond to a reactant, creating a new intermediate and an alternate multi-step pathway.
Mechanistic features AP students should recognize:
Step 1: Addition/attachment. The catalyst bonds to the substrate (reactant) to form an intermediate.
Step 2: Transformation. The intermediate undergoes the key change (often easier because electron density and bonding have been altered).
Step 3: Elimination/detachment. The product leaves and the catalyst is regenerated.
Why this can increase rate:
The catalyst-bound intermediate can be more electrophilic or more nucleophilic than the original reactant.
A difficult direct reaction can be replaced by steps that avoid an unstable high-energy arrangement.
Bond-making/bond-breaking may become more favorable due to electron donation/withdrawal by the catalyst fragment.
What to watch for:
The catalyst is not a reactant overall; it should cancel out when summing elementary steps.
The covalent intermediate is real within the mechanism, but it does not appear in the net equation.
Acid–base catalysis (proton transfer catalysis)
Acid–base catalysis accelerates a reaction through proton transfer steps. By donating or accepting , the catalyst changes charge distribution and bonding, producing new intermediates and steps.
Acid–base catalysis: catalysis in which a species donates or accepts a proton during the mechanism, generating intermediates and providing an alternate pathway.
A common pattern is protonation then reaction then deprotonation (or the reverse).
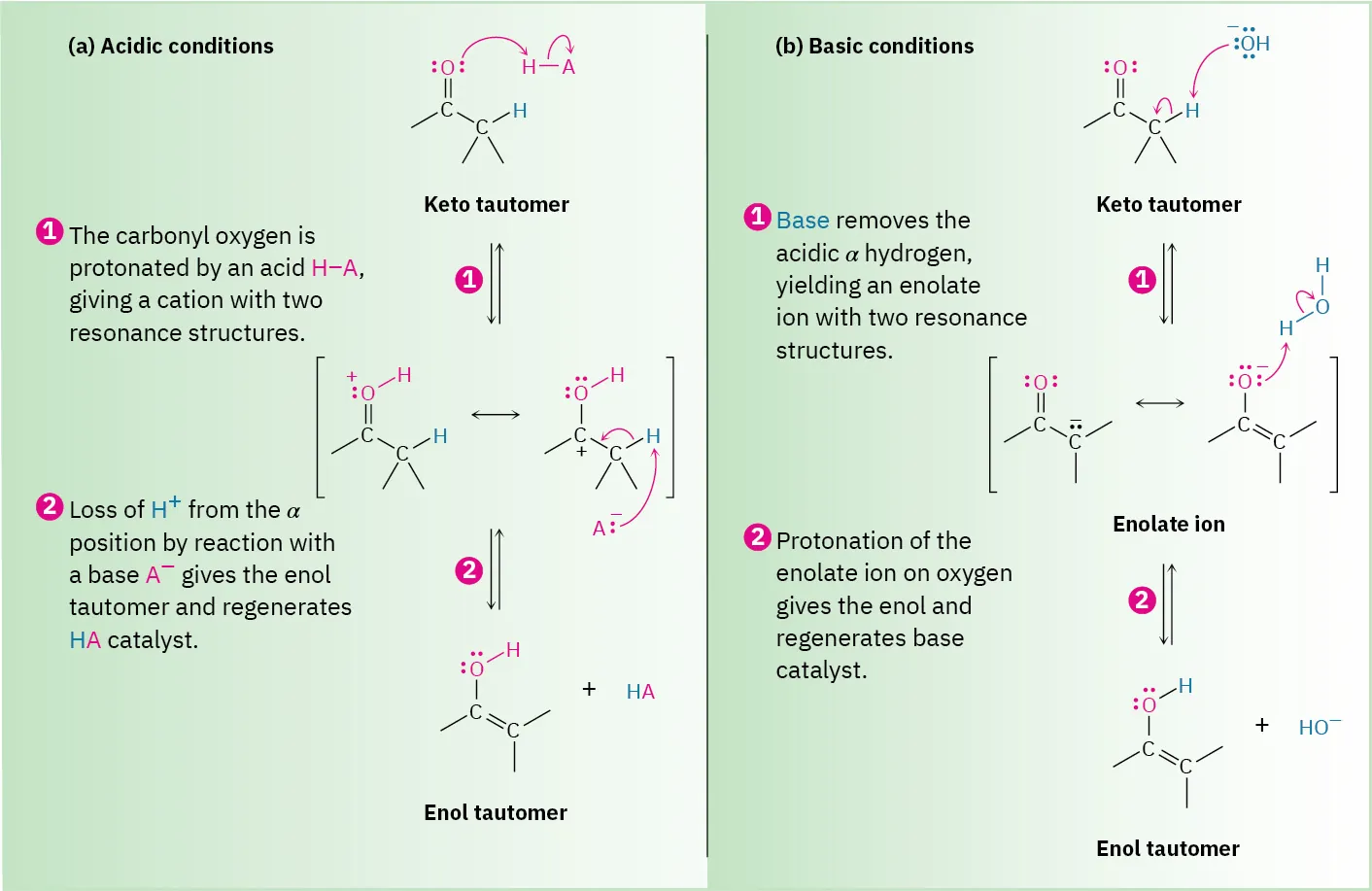
Mechanism scheme for acid-catalyzed vs. base-catalyzed keto–enol tautomerism, showing how proton transfer steps generate reactive intermediates (oxonium/enolate) and then return the proton to regenerate the catalyst. The acid pathway begins with protonation (increasing electrophilicity), while the base pathway begins with deprotonation (increasing nucleophilicity/basicity). This is a representative example of proton-transfer catalysis producing “new intermediates and steps.” Source
Proton transfer can:
Increase electrophilicity (protonation can make a functional group more susceptible to attack)
Increase nucleophilicity (deprotonation can generate a stronger nucleophile/base)
Turn a poor leaving group into a better one (protonation can help a group depart)
Acid catalysis (proton donor helps)
Catalyst acts as a proton donor in an early step, forming a protonated intermediate.
Later, a proton is transferred back so the acid catalyst is regenerated.
Net effect: same overall reaction, but with extra steps that proceed faster.
Base catalysis (proton acceptor helps)
Catalyst acts as a proton acceptor to generate a more reactive deprotonated intermediate (or to remove a proton during bond formation).
Later steps return a proton to regenerate the base catalyst.
Recognising acid–base catalysis in mechanisms
Look for:
transfer steps (often fast) that create intermediate species
The catalyst appearing as (or similar conjugate pair) in different steps
Cancellation of catalyst-related species when adding steps to get the net reaction
Shared takeaways for AP Chemistry
Both modes introduce additional elementary steps and intermediates.
The catalyst participates chemically but is regenerated, so its net amount is constant.
These pathways often increase rate by improving reactivity, charge stabilization, or leaving-group ability rather than changing products or equilibrium position.
FAQ
Specific acid catalysis depends only on $[H_3O^+]$ (proton transfer from hydronium is the key catalytic step).
General acid catalysis involves other proton donors (e.g., $HA$ in a buffer), so the rate can depend on the concentration/identity of those acids, not just pH.
A rate that varies systematically with pH can indicate proton transfer involvement.
Often, different pH regions show different dominant catalytic forms (acidic form vs basic form), producing a characteristic pH–rate profile.
Protonation can convert a negatively charged or strongly basic leaving group into a neutral, more stable species.
This lowers the energetic penalty for bond cleavage in the step where the group departs.
Yes. A catalyst can form a covalent intermediate and also donate/accept protons in other steps.
This is common in complex catalytic sites where multiple functionalities cooperate.
Transient formation of a catalyst-bound species can sometimes be inferred from time-dependent spectroscopy or trapping experiments.
Another clue is “burst” kinetics, where product formation briefly outpaces steady-state turnover due to intermediate accumulation.
Practice Questions
(2 marks) State what is meant by acid–base catalysis and describe one way proton transfer can increase reaction rate.
1 mark: Defines acid–base catalysis as catalysis involving proton donation/acceptance forming intermediates/extra steps.
1 mark: Explains one valid effect (e.g., protonation increases electrophilicity, deprotonation increases nucleophilicity, protonation improves leaving group).
(5 marks) A reaction is proposed to be covalently catalysed by catalyst . Outline a three-step mechanism showing (i) formation of a –substrate intermediate, (ii) conversion to a –product intermediate, and (iii) regeneration of . Explain how this differs from acting as a reactant overall.
1 mark: Step showing temporary covalent bond formation (intermediate contains ).
1 mark: Step showing chemical transformation while still bound to .
1 mark: Step showing product release and regeneration of .
1 mark: States cancels when steps are added / not in overall equation.
1 mark: Contrasts with a reactant: a reactant would be consumed and appear in the net equation.

