AP Syllabus focus: ‘Each exponent is the reaction order for that reactant; adding exponents gives the overall reaction order.’
Reaction order is how sensitively a reaction’s rate responds to changing reactant concentrations. It is read from the exponents in a rate law, not from the balanced equation, and it guides predictions about rate changes.
Reaction order: what the exponents mean
A rate law expresses the measured dependence of rate on reactant concentrations using exponents.
Reaction order (with respect to a reactant): the exponent on that reactant’s concentration term in the experimentally determined rate law.
Reaction order is determined by experiment (for most reactions) and can be 0, positive, fractional, or even negative, depending on the mechanism.
General rate-law form and identifying orders
= reaction rate (typically )
= proportionality constant (rate constant)
= molar concentrations of reactants ()
= reaction orders with respect to and (unitless)
For the rate law above:
Order in is .
Order in is .
If a reactant does not appear (or has exponent 0), the rate is independent of that reactant’s concentration under those conditions.
Overall order: combining the exponents
The overall reaction order describes the net concentration dependence of the rate when all reactant concentrations change.
Overall reaction order: the sum of all exponents in the rate law for the reactants.
In the rate law , the overall order is:
Overall order
Interpreting common orders (qualitative impact)
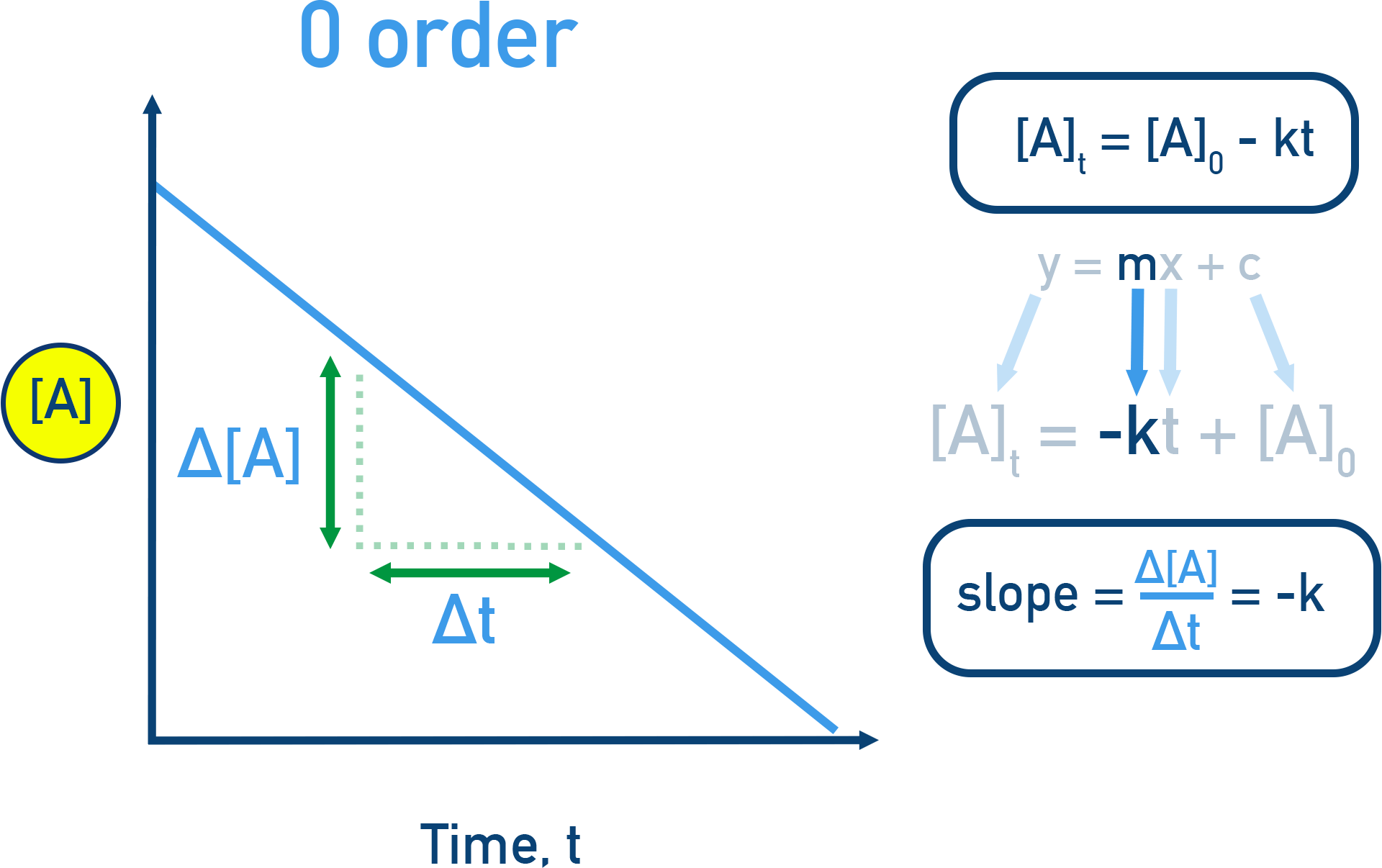
Zeroth-order kinetics shown as a linear plot of versus time with constant slope . The straight-line decrease emphasizes that the rate is constant (independent of ) until the reactant is depleted. Source
Zero order in a reactant (): changing that reactant’s concentration does not change the rate (rate factor of ).
First order in a reactant (): doubling that reactant doubles the rate (all else constant).
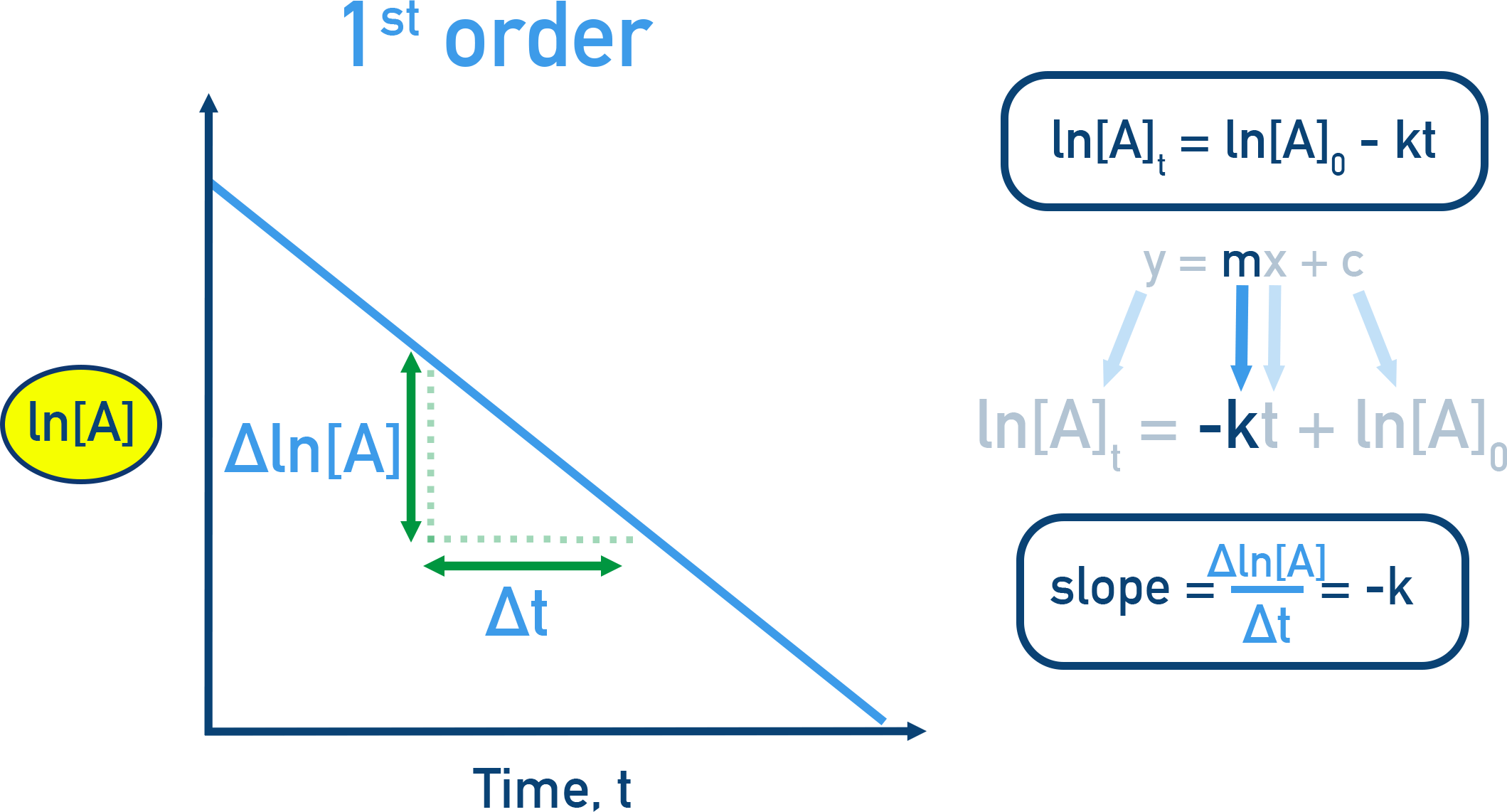
First-order kinetics illustrated by a straight-line plot of versus time, where the slope is . The linear relationship is the experimental signature that the rate is proportional to (i.e., exponent in the rate law). Source
Second order in a reactant (): doubling that reactant quadruples the rate.
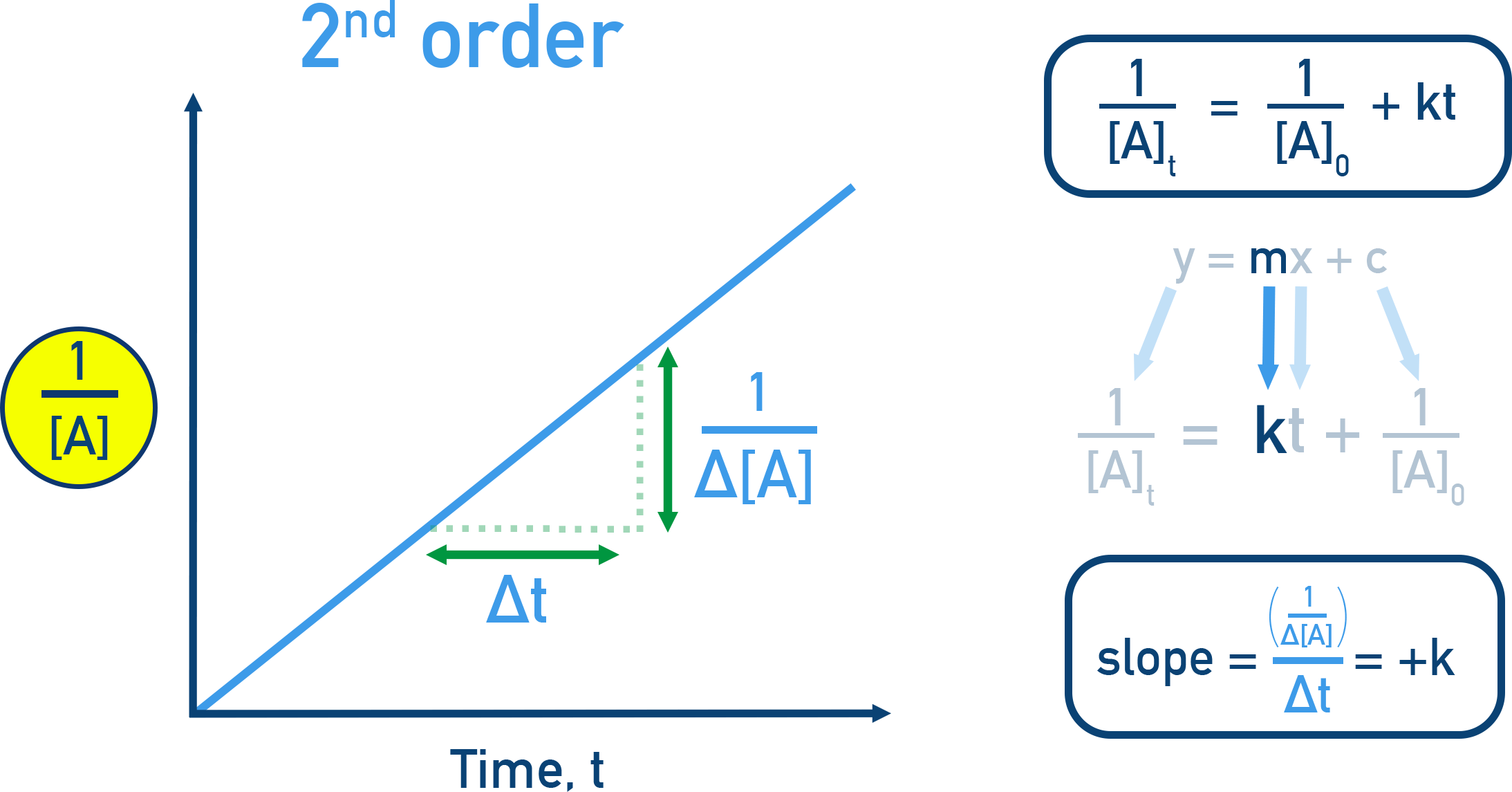
Second-order kinetics shown with the linearized integrated-rate-law plot versus time, which has slope . This visual highlights the stronger concentration dependence associated with an exponent of in a rate law (e.g., ). Source
Fractional order (e.g., ): doubling that reactant increases the rate by a factor of .
Negative order (e.g., ): increasing that reactant decreases the rate (often due to inhibitory steps in a mechanism).
Reaction order is not stoichiometric coefficient
A frequent mistake is assuming exponents must match coefficients from the balanced chemical equation. In general:
Balanced equations describe overall stoichiometry, not how the rate depends on concentration.
Rate-law exponents come from the reaction pathway, so they are typically not predictable from the overall equation alone.
When a reactant’s concentration changes, the rate changes by a power-law factor set by its order. This lets you compare “which reactant matters most” for rate control under a given rate law, and how the rate scales when multiple concentrations change simultaneously.
FAQ
Yes. If any exponent is fractional or negative, the sum can be fractional or negative.
A negative overall order means the rate decreases as concentrations (in the way specified by the rate law) increase, indicating net inhibition within the measured concentration range.
A negative exponent suggests the reactant (or something proportional to it) reduces the concentration of an active species needed for reaction.
This can happen via formation of an unreactive complex, surface blocking, or shifting a fast equilibrium away from a reactive intermediate.
Order in $A$ is an experimentally observed exponent in the overall rate law. Molecularity refers to how many particles collide in a single elementary step.
They only necessarily match for an elementary step’s rate expression, not for an overall reaction.
They can, if the dominant pathway changes across concentration ranges or if a pre-equilibrium shifts which species controls the rate.
In such cases, a single simple rate law may only apply over a limited set of conditions.
A pseudo-order arises when one reactant is in such large excess that its concentration is effectively constant during the measurement.
Its concentration term can be absorbed into an “apparent” constant, making the observed rate law look like a lower-order dependence on the remaining reactant(s).
Practice Questions
(2 marks) A reaction has rate law . State the order with respect to , the order with respect to , and the overall order.
Order in is 2 (1 mark)
Order in is 1 and overall order is 3 (1 mark for both correct)
(5 marks) For a reaction with : (a) State the order with respect to and with respect to . (2 marks) (b) State the overall order. (1 mark) (c) If is quadrupled and is doubled, by what factor does the rate change? (2 marks)
(a) Order in is (1 mark); order in is (1 mark)
(b) Overall order is (1 mark)
(c) Rate factor from : (1 mark); rate factor from : ; net factor (1 mark)

