AP Syllabus focus: ‘Comparing initial rates while changing starting concentrations is a method for finding the order with respect to each reactant.’
The method of initial rates uses early-time rate measurements to determine how strongly rate depends on each reactant concentration. It is a core experimental strategy for building an empirical rate law.
Core idea and key terms
What you measure
Initial rate: The reaction rate measured immediately after mixing (near ), before concentrations change significantly.
In practice, the “initial rate” is estimated from the earliest, nearly linear portion of a concentration–time (or signal–time) curve.
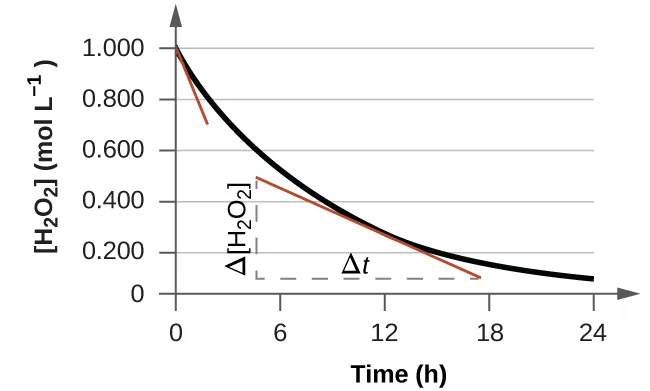
Concentration–time curve for a reacting species with tangent lines drawn to show how rate is obtained from the slope. The tangent at represents the initial rate, while a tangent at later time represents an instantaneous rate after concentrations have changed. The dashed and markers emphasize that the slope is computed as a ratio of changes in concentration and time. Source
What you are trying to determine
You compare experiments that start with different initial concentrations to infer the exponents in the rate law (the orders). A generic empirical rate law is:
= initial rate (e.g., )
= rate constant (units depend on )
= initial reactant concentrations ()
= reaction orders with respect to and (dimensionless)
The method works because changing one initial concentration while holding others constant isolates that reactant’s influence on rate.
How the method is applied (no calculations)
Designing comparisons (the “pairwise” approach)
Choose two experiments where only one reactant’s initial concentration changes (others are constant).
Use the ratio form to solve for the order of that reactant.
Repeat with different pairs to find the order for each reactant in turn.
A common ratio relationship (when only changes) is:
\dfrac{\text{Rate}_2}{\text{Rate}_1}=\left(\dfrac{[A]_2}{[A]_1\right)^m}
= initial rates from experiments 1 and 2
= initial concentrations of in experiments 1 and 2
= order in
This ratio approach avoids needing initially; can be found after orders are known by substituting one experiment’s initial rate and concentrations into the rate law.
Practical steps in an initial-rate experiment
Prepare mixtures with known initial concentrations (often by dilution).
Start timing at mixing; collect an early-time signal proportional to concentration:
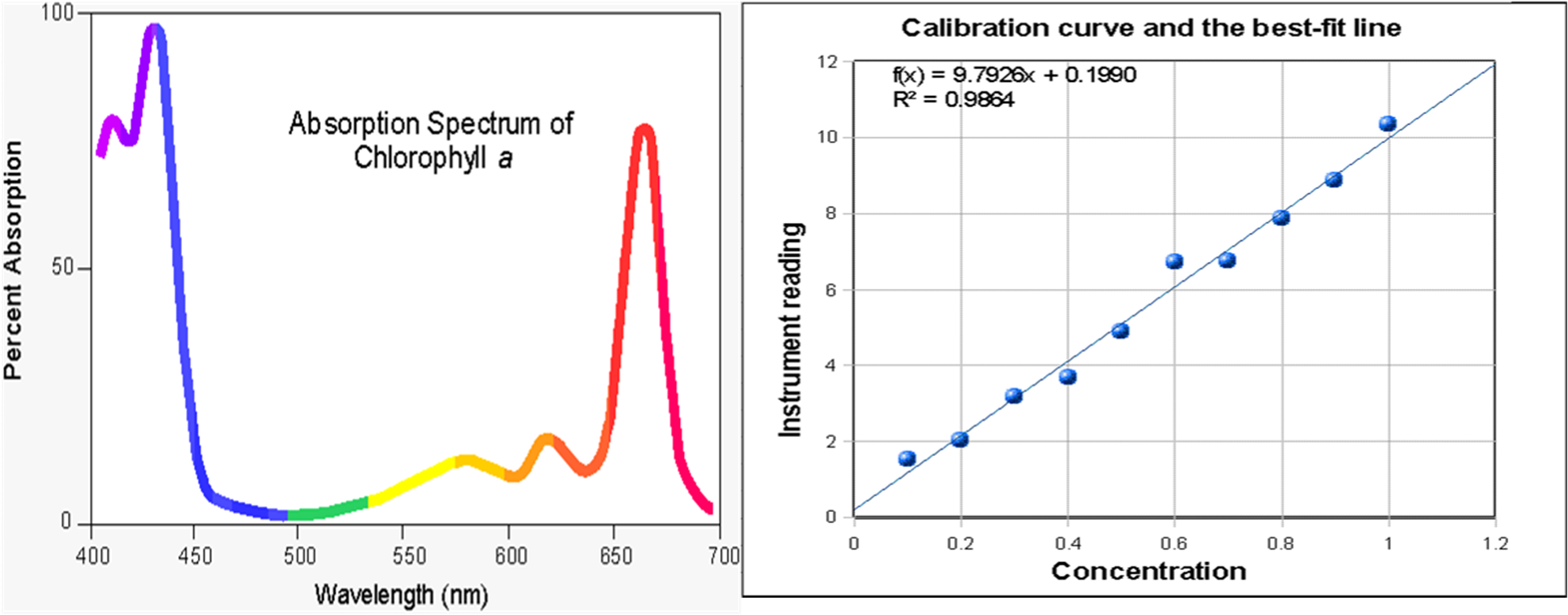
Side-by-side example of (left) an absorbance spectrum and (right) a calibration curve with a best-fit line. The calibration plot illustrates the linear relationship between measured instrument response (e.g., absorbance) and concentration over the usable range, which is what allows absorbance–time data to be converted into concentration–time data. Once concentration vs. time is obtained, the initial slope near can be used as the initial rate. Source
concentration by sampling/titration
absorbance by colorimetry (via Beer–Lambert calibration)
pressure or volume for gas production
Determine the initial slope (rate) from the earliest data points.
Compare rates across trials to determine each order, then determine .
Experimental and reasoning pitfalls (high-yield)
Keeping “all else equal”
Temperature must be constant; rate is temperature-sensitive, so uncontrolled warming/cooling distorts inferred orders.
Mixing time matters: if the reaction is fast relative to mixing, the “initial” window may already include significant concentration change.
Interpreting proportional changes correctly
If doubling leaves the initial rate unchanged, the reaction is zero-order in over that range.
If doubling doubles the rate, it is first-order in .
If doubling quadruples the rate, it is second-order in .
Data quality issues
Use multiple early points rather than a single instantaneous reading; noise can strongly affect a slope.
Ensure the measured signal truly tracks the intended species (e.g., avoid overlapping absorbances or side processes).
FAQ
Use simple multiples (e.g., $\times 2$, $\times 3$) for one reactant at a time, keeping others fixed, so the rate ratio maps cleanly to an integer or half-integer exponent.
Plan a minimal set where each reactant is isolated at least once. If that is impossible, redesign concentrations; otherwise, you must use multi-variable fitting rather than simple pairwise ratios.
Collect more frequent early measurements and estimate the tangent at $t\approx 0$ by fitting only the first small time interval where change is minimal.
The method is empirical: if the dominant pathway changes with concentration (or if a reactant is effectively in excess), the apparent exponent can shift over different starting ranges.
Use your proposed $m$ and $n$ to predict rate ratios for trials not used to derive them; consistent predicted vs observed ratios supports (but does not prove) the form.
Practice Questions
Q1 (2 marks) In an initial rates study, doubling while keeping constant doubles the initial rate. State the order with respect to and justify briefly.
Order in is 1 (1 mark)
Justification: rate changes in direct proportion to when is constant (1 mark)
Q2 (5 marks) A reaction has rate law . In three experiments, is constant. When is tripled, the initial rate increases by a factor of 9.
(a) Determine . (b) State one additional experimental comparison needed to determine . (c) State what information is required to then determine .
(a) Uses ratio idea to link and obtains (2 marks)
(b) Describes comparing two experiments where only changes while is constant (1 mark)
(c) States that an initial rate and corresponding initial concentrations (from any one experiment) are substituted into the rate law to solve for (2 marks)

