AP Syllabus focus: ‘Experimental methods track reactant or product amounts over time; these data are used to determine the reaction rate.’
Measuring reaction rates means collecting time-resolved data for a reactant or product and converting those measurements into concentration (or amount) versus time. Good experiments prioritise a clear signal, reliable timing, and controlled conditions.
What “rate” means in an experiment
A rate measurement always links a chemical change to an elapsed time and is usually reported as a change in molar concentration per second.
Reaction rate: the change in concentration of a reactant or product per unit time.
In practice, you often measure a proxy (colour intensity, pressure, mass) that can be converted into concentration or moles, then determine the slope of a suitable plot.
= molar concentration of A, in
= time, typically in
= molar concentration of P, in
Average rates use two time points; an instantaneous rate is estimated from the slope of the tangent to a concentration–time curve at a particular time (commonly at to minimise complications like reverse reaction or depletion).
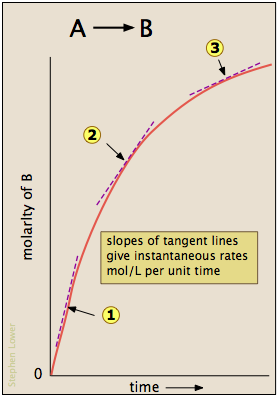
A concentration–time plot illustrating that the instantaneous reaction rate at a chosen moment is the slope of the tangent line to the curve at that time. The tangent-slope construction makes the graphical meaning of an “instantaneous rate” explicit and distinguishes it from an average slope taken over a finite time interval. Source
Designing a good rate experiment
Choose a measurable signal tied to concentration
Your method should track something that changes smoothly and predictably as the reaction proceeds:
Direct concentration measurements (common for aqueous systems)
Amount of gas produced/consumed
Mass change (e.g., gas escaping)
Optical properties (colour/absorbance)
Electrical properties (conductivity)
A key requirement is a known relationship between the signal and concentration (often via calibration or an established law).
Control variables and timing
To ensure the rate reflects chemistry rather than technique:
Keep temperature constant (water bath if needed).
Mix quickly and consistently; define a clear time zero.
Use clean glassware and consistent volumes to avoid systematic errors.
Collect enough data points early in the run if you need an early-time rate.
Common experimental methods (what you measure and what you infer)
Spectrophotometry (colour/absorbance)
If a reactant or product is coloured (or forms a coloured complex), you can monitor absorbance vs time and convert to concentration.
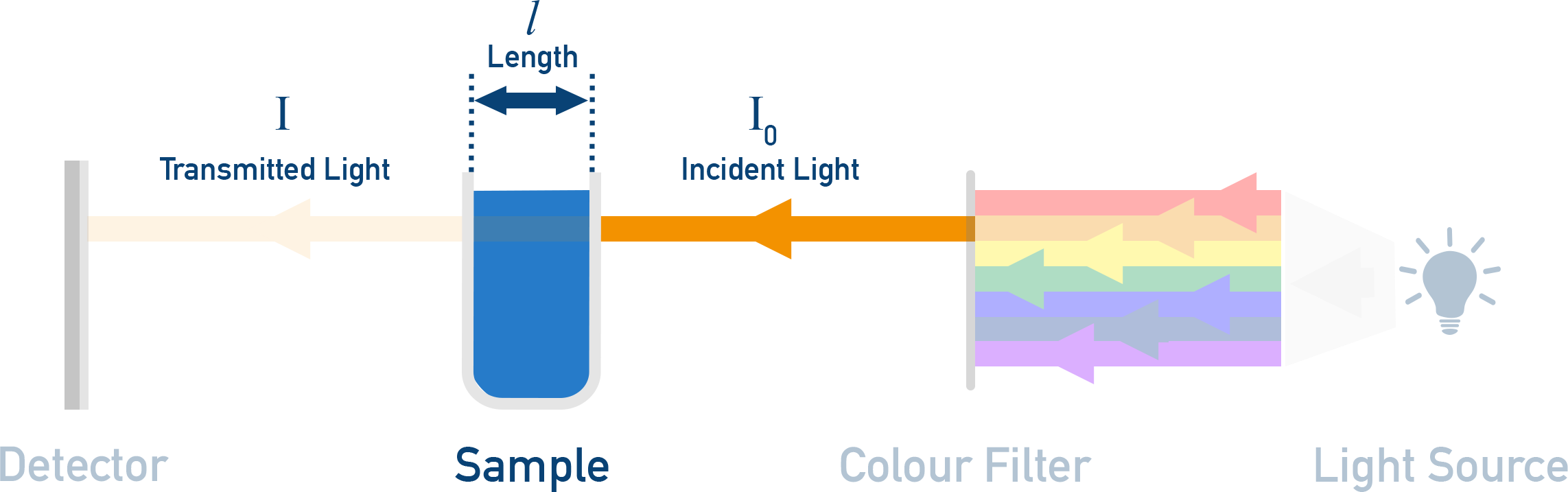
A schematic of a colorimeter/spectrophotometer setup showing incident light passing through a sample of path length and emerging as transmitted light at the detector. This diagram links the experimental geometry (especially ) to how absorbance measurements are generated during a kinetics run. Source
= absorbance (unitless)
= molar absorptivity, in
= path length of cuvette, in
= concentration, in
This approach is powerful because it is rapid and non-destructive, but it depends on using an appropriate wavelength and staying within a linear absorbance range.
Gas pressure or volume measurements
For reactions producing gas, rate can be obtained by tracking:
Gas volume vs time (gas syringe)
Pressure vs time (pressure sensor)
You then convert gas amount to moles (and, if needed, to concentration in the reaction mixture). Ensure leaks are minimised and temperature is stable, since gas measurements are temperature-sensitive.
Mass loss over time
If a gas escapes from an open system, the balance reads a decreasing mass. The rate of mass loss can be related to moles of gas formed (using molar mass), then to reaction progress. Key limitations include drafts, evaporation, and splashing.
Sampling and quenching, then titration/analysis
For reactions without a convenient continuous signal, you can:
Remove timed aliquots
Quench (stop) the reaction in the sample (cooling, neutralisation, dilution, or a scavenger)
Determine concentration by titration or another assay
This method is flexible but depends strongly on fast, consistent quenching and accurate timing.
Conductivity or pH probes
If ions are produced/consumed, conductivity vs time can track changing ionic concentration. Similarly, pH vs time can track changes for acid–base involved processes. These signals may require calibration and can be influenced by side equilibria, so the interpretation must match the chemistry.
Turning raw data into a rate
Data handling workflow
Record the signal at known times.
Convert signal concentration (or moles) using calibration, Beer–Lambert, or stoichiometric/physical relationships.
Plot concentration vs time (or an equivalent progress variable vs time).
Determine rate as a slope:
Average rate: slope between two points.
Instantaneous/initial rate: tangent slope at a chosen time (often near ).
Units and reporting
Typical rate units are (or sometimes if using moles). Always state:
what species is being tracked,
the time interval (for average rates),
and the sign convention (reactants decrease, products increase).
FAQ
Pick an interval where the signal changes measurably above noise, but not so long that conditions change dramatically (e.g., significant temperature drift or reactant depletion).
For very fast reactions, shorten intervals and increase sampling frequency.
Continuous monitoring records a signal without removing material (e.g., absorbance, pressure).
Sampling and quenching removes aliquots at set times, stops reaction in each sample, then analyses concentration; it is slower but works when no continuous signal exists.
Prepare standards of known concentration for the coloured species and measure absorbance at the chosen wavelength.
Plot $A$ vs $c$ to obtain a line; use it to convert absorbance readings to concentration during the kinetics run.
If the solution is not homogeneous immediately, the measured signal may reflect local concentrations rather than the bulk.
Sensor response times can smooth sharp changes, making the earliest slope appear smaller than the true initial rate.
Use an experimentally determined conversion (calibration curve) over the working range, or redesign the method to track a different species/signal.
Nonlinear regions (e.g., high absorbance) should be avoided to keep rate extraction straightforward.
Practice Questions
(2 marks) A reaction produces a coloured product. The absorbance at 500 nm increases linearly from 0.20 to 0.44 over 80 s. State what graph you would plot to determine the average rate over this interval, and what feature of the graph gives the rate.
Plot absorbance (or concentration) against time (1)
Rate is determined from the gradient/slope over the interval (1)
(5 marks) Describe how you could measure the rate of a reaction that produces in an aqueous mixture using a pressure sensor. Include what you would record, how you would process the data to obtain a rate, and two key controls/precautions.
Record pressure (or pressure change) in the sealed vessel as a function of time (1)
Convert pressure data to amount of gas (moles) using an appropriate gas relationship, then relate to reaction progress (1)
Determine rate from the gradient of a suitable plot (e.g., moles of or concentration vs time), mentioning tangent/initial slope or average slope (1)
Control temperature / ensure constant temperature (1)
Prevent leaks / consistent mixing and clear time zero / adequate sensor calibration (any one) (1)

