AP Syllabus focus: ‘Elementary reactions break some bonds and form new ones; progress can be tracked along a reaction coordinate.’
Reaction energy diagrams connect molecular-scale bond changes to what we observe macroscopically as reaction progress. This page focuses on how to interpret reaction coordinates, bond breaking/forming, and the transition state for an elementary step.
Reaction coordinate: what it represents (and what it does not)
A reaction does not occur in a single jump from reactants to products; atoms move continuously through many transient arrangements. An energy diagram compresses that complex motion into a single “progress” axis.
Reaction coordinate: An abstract measure of progress along the reaction pathway from reactants to products; it represents changing atomic positions and bonding, not time or distance.
Because the reaction coordinate is not time, a point halfway along the coordinate is not “half the time.” Instead, it is a particular arrangement (geometry) of atoms and partial bonds.
Connecting the coordinate to bond changes
For an elementary reaction, the reaction coordinate tracks:
stretching and breaking of specific bonds in reactants
reorientation and approach of reactant particles
formation of new bonds leading to products
These changes happen as electron density shifts from old bonding interactions to new ones.
Bonds along the pathway: breaking and making
In an elementary step, bond breaking and bond making are typically concerted (overlapping in time), rather than fully sequential. This matters because the highest-energy point usually corresponds to a structure where:
some original bonds are partially broken (weakened, elongated)
some new bonds are partially formed (incipient bonding)
electron density is redistributed, giving an unstable, high-energy arrangement
A common misconception is that bonds must completely break before new ones form. In many elementary steps, forming interactions begin before old ones are fully gone, which can reduce the energy required compared with a purely stepwise picture.
Structural viewpoint: what changes fastest?
Along the coordinate, the fastest-changing features are typically:
bond lengths (especially the bonds being broken/formed)
bond angles and torsions (to reach a geometry that allows bond rearrangement)
relative positions of colliding particles (distance and orientation)
The key idea is that the coordinate packages these geometric changes into one conceptual axis.
The transition state: the “top of the hill”
Energy profiles for elementary steps show a maximum at the point of greatest instability. That maximum corresponds to the transition state, a fleeting arrangement that cannot be isolated as a stable substance.
Transition state: The highest-energy arrangement of atoms along the reaction coordinate for an elementary step; it corresponds to the maximum on the energy profile.
The transition state is sometimes called the activated complex, but it is best understood as a geometry, not a “species in a flask.”
How to recognise a transition state description
A transition state description usually includes:
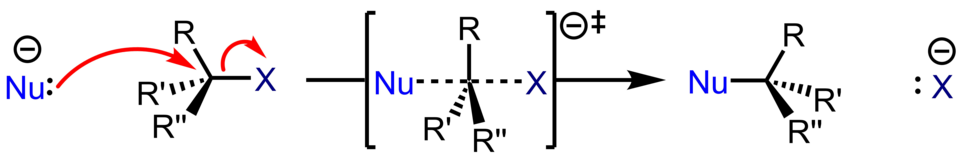
SN2 substitution mechanism highlighting the transition state (TS) where the nucleophile–carbon bond is partially forming while the carbon–leaving group bond is partially breaking (dashed bonds). The curved arrows depict electron-density flow, emphasizing that bond making and bond breaking are concerted within a single elementary step. Source
partial bonds shown with dashed lines (conceptually indicating “in the process of” breaking/forming)
unusual bond angles/lengths compared with reactants or products
high sensitivity to small geometric changes (because it sits at the peak of the energy barrier)
It is incorrect to treat the transition state as an intermediate.
Intermediate: A species that is formed in one step and consumed in a later step, corresponding to a local minimum on a multistep energy profile.
Between reactants and products for a single elementary step, there is no intermediate minimum—only one barrier peak.
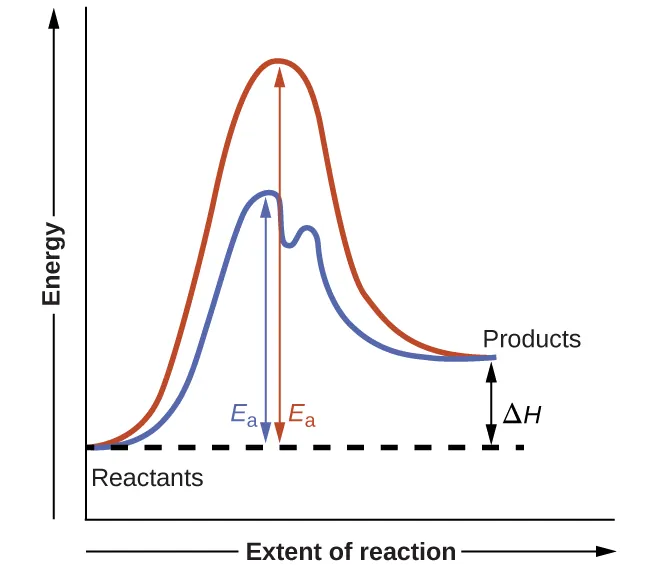
Reaction coordinate (extent of reaction) diagrams for the same overall reaction with and without a catalyst. The catalyzed pathway has a lower highest barrier (smaller ) and proceeds through two transition states separated by an intermediate (a local minimum), while depends only on the reactant and product energy levels. Source
Why the transition state matters for kinetics (conceptually)
Even without calculations, the energy maximum implies an important kinetic idea: to proceed from reactants to products, reacting particles must reach the transition-state geometry. That requires:
sufficient energy concentrated in the right modes (bond stretching, bending)
an approach that produces the correct atomic arrangement for bond rearrangement
Thus, the reaction coordinate links microscopic structure to macroscopic rate by highlighting the specific “bottleneck” geometry that must be achieved.
Reading bond changes qualitatively from the coordinate
When comparing reactant-side and product-side regions along the coordinate, you can infer:
which bonds are being broken (those present early but absent late)
which bonds are being formed (those absent early but present late)
whether the transition state is “reactant-like” or “product-like” based on which partial bonds dominate at the peak (a qualitative structural comparison)
Staying focused on this subtopic: the coordinate is the conceptual path; the transition state is the key structure at the peak; and bond breaking/forming is the molecular story that the coordinate summarises.
FAQ
It is chosen to follow the most important collective atomic motions (often a key bond length or combination of distances/angles). In computations, it may be defined by an “intrinsic reaction coordinate” connecting minima via the saddle point.
Not in the usual sense. It is a transient geometry at a saddle point on a potential energy surface; assigning a single formula can be misleading because bonding is partial and continuously changing.
Dashed bonds indicate partial bonding: electron density is shifting, so bond order is between 0 and 1 (or between 1 and 2, etc.). It is a structural convention, not a measurable bond type.
Yes. Real reactions occur on multidimensional potential energy surfaces. A one-dimensional reaction coordinate is a simplified projection capturing the dominant changes relevant to the pathway being discussed.
Indirect evidence includes kinetic isotope effects, stereochemical outcomes, and substituent effects (e.g., Hammett relationships), which can indicate whether certain bonds are more formed or broken in the transition state.
Practice Questions
(2 marks) For a single elementary reaction step, explain what the reaction coordinate represents and state what feature of the energy profile corresponds to the transition state.
Reaction coordinate is an abstract measure of progress via changing atomic positions/bonding (not time). (1)
Transition state corresponds to the maximum (peak) on the energy profile. (1)
(5 marks) A student says: “At the transition state, reactant bonds have fully broken, then product bonds start forming, and the transition state is the same as an intermediate.” Evaluate this statement using correct kinetic terminology for an elementary step.
Transition state is a highest-energy arrangement/geometry along the reaction coordinate (not an isolable species). (1)
In many elementary steps, bond breaking and bond formation are concerted/overlapping; bonds are typically partially broken/formed at the transition state. (2)
Intermediate is a distinct species formed then consumed and corresponds to an energy minimum (not the peak). (1)
Therefore transition state is not the same as an intermediate. (1)

