AP Syllabus focus: ‘An energy profile plots energy along the reaction coordinate from reactants, through a transition state, to products; the reactant–transition state energy gap is the forward activation energy.’
An energy profile (reaction energy diagram) is a visual tool for interpreting how energy changes during a reaction. You should be able to extract activation energy and the overall energy change directly from the diagram.
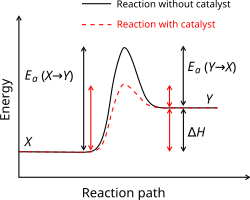
A reaction coordinate diagram that labels the activation energy as the vertical gap between reactants and the transition-state peak, and also shows the net energy change between reactants and products. The two curves illustrate how an enzyme/catalyst lowers without altering the reactant or product energy levels. This is a compact visual for separating “barrier height” from “overall energy change.” Source
What an energy profile shows
An energy profile plots the system’s energy as reactants transform into products along a reaction pathway.
Energy profile (reaction energy diagram): a graph of potential energy versus reaction progress, showing reactant energy, a maximum at the transition state, and product energy.
Key features to identify on the diagram
y-axis: Potential energy (commonly in kJ/mol)
x-axis: Reaction coordinate (a qualitative measure of progress; it is not time)
Reactants level: starting energy
Peak: transition state (highest-energy point on the pathway)
Products level: final energy
Activation energy () from the diagram
Activation energy is read as a vertical energy difference between a starting level and the peak. On an energy profile, the relevant “starting level” depends on the direction you are considering (forward vs reverse).
Activation energy (): the minimum energy barrier that must be overcome for reactants to reach the transition state and form products.
Forward activation energy
Forward reaction: reactants products
Forward : vertical gap from the reactants energy up to the peak (transition state)
Interpreting magnitude:
Larger means a bigger barrier (harder to initiate under the same conditions)
Smaller means an easier pathway (more accessible transition state)
A crucial reading skill is to focus on vertical differences (energy gaps), not the horizontal spacing.
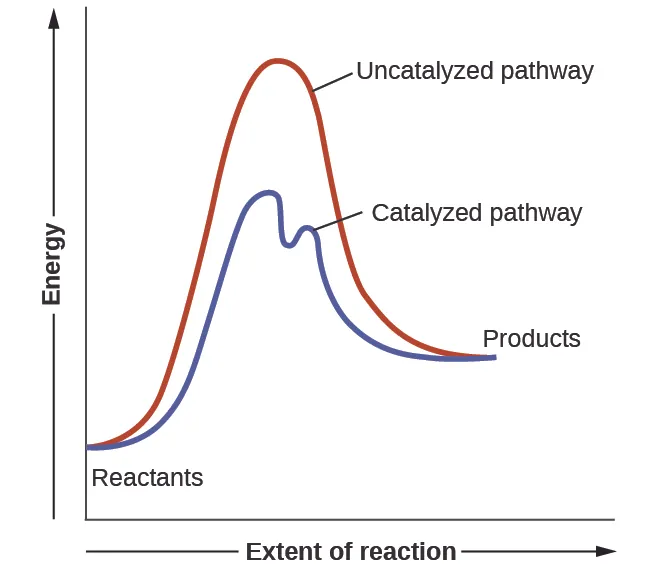
A potential-energy diagram comparing an uncatalyzed pathway to a catalyzed pathway. The catalyst changes the reaction pathway to lower the activation-energy barrier (lower peak) while keeping the reactant and product energy levels the same, so the overall energy change remains unchanged. This directly supports interpreting as a vertical energy difference from the reactants level to the transition-state peak. Source
= forward activation energy, kJ mol
= potential energy at the transition state (peak), kJ mol
= potential energy of reactants, kJ mol
= overall energy change (products minus reactants), kJ mol
= potential energy of products, kJ mol
Reverse activation energy (common extension when “reading”)
Even if the prompt emphasises forward , you should recognise that:
Reverse reaction: products reactants
Reverse : vertical gap from the products energy up to the same peak
If products lie lower than reactants, reverse will be larger than forward (and vice versa)
Overall energy change () from the diagram
The overall energy change compares the energy of products with reactants, independent of the height of the peak.
How to read
Locate the reactants energy level and the products energy level.
Compute (conceptually) the vertical difference: products minus reactants.
Interpret the sign:
(products lower): energy is released overall (often described as exothermic on potential-energy diagrams)
(products higher): energy is absorbed overall (often described as endothermic on potential-energy diagrams)
Common interpretation checkpoints
A reaction can have a large but a negative (big barrier, but energy-releasing overall).
A reaction can have a small but a positive (easy to start, but energy-absorbing overall).
The peak height tells you about the barrier; the reactant/product heights tell you about the net energy change.
FAQ
Reaction coordinate is a qualitative progress variable (bond changes and molecular rearrangements), not a clock.
Different reactions can have similar-looking profiles but occur over very different timescales.
Not necessarily. Many diagrams show potential energy changes, which often approximate enthalpy changes under typical conditions.
Whether $\Delta E$ matches $\Delta H$ depends on what energy quantity the diagram represents.
It indicates $\Delta E \approx 0$.
The reaction can still have a nonzero $E_a$, so it may require an initial energy input even though there is no net energy change.
No. The transition state corresponds to a maximum on the energy profile and is not a stable species.
It cannot be isolated because it does not reside in a potential-energy minimum.
Only vertical differences on the same y-axis scale are meaningful.
If axes are not labelled or are distorted, you can still compare relative barriers and relative product/reactant energies, but not precise numerical values.
Practice Questions
(2 marks) Define the forward activation energy, , in terms of an energy profile, and state which two points on the diagram are used to determine it.
1 mark: States is the energy gap from reactants to the transition state (energy barrier to reach the transition state).
1 mark: Correctly identifies reactants energy level and the peak/transition state as the two points.
(5 marks) An energy profile shows kJ mol, kJ mol, and kJ mol. Determine (i) , (ii) , and (iii) whether the forward reaction is energy-releasing or energy-absorbing, justifying your choice using the sign of .
1 mark: stated or used correctly.
1 mark: kJ mol.
1 mark: stated or used correctly.
1 mark: kJ mol.
1 mark: Correct interpretation: so energy-releasing overall (products lower than reactants).

