AP Syllabus focus: ‘The Arrhenius equation relates temperature dependence of rate to activation energy; calculations with the Arrhenius equation are not assessed on the AP Exam.’
Reaction rates are strongly temperature dependent. The Arrhenius equation provides a conceptual link between temperature, activation energy, and the rate constant by describing how the probability of successful, energy-sufficient collisions changes.
Purpose of the Arrhenius Equation
In kinetics, the rate law uses a rate constant, k, to connect reaction rate to reactant concentrations. Even when concentrations are fixed, rates change with temperature because k is temperature dependent. The Arrhenius equation is the standard model used to explain that dependence.
Arrhenius Equation (Conceptual Form)
Arrhenius equation: A model that expresses how the rate constant depends on temperature and activation energy, predicting faster reactions at higher temperatures.
A key idea is that only a fraction of collisions have enough energy to reach the transition state, and that fraction increases rapidly as temperature increases.
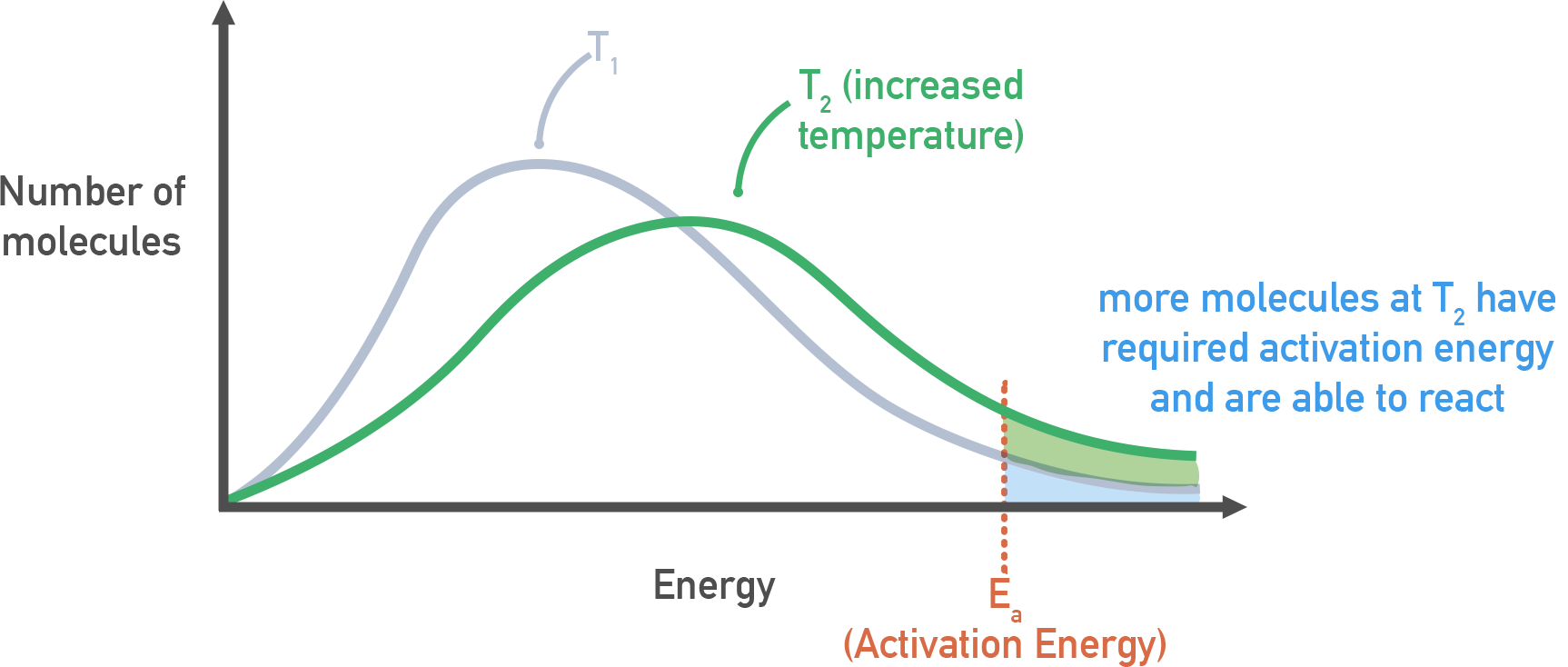
Maxwell–Boltzmann energy distributions at two temperatures ( and higher ) with an activation-energy threshold marked as . The shaded region beyond grows at higher temperature, representing a larger fraction of molecules energetic enough to react. This provides an intuitive bridge from “more high-energy collisions” to a larger rate constant . Source
= rate constant (units depend on overall reaction order)
= frequency (pre-exponential) factor (same units as )
= activation energy (J/mol)
= gas constant (J/mol·K)
= temperature (K)
This exponential form highlights that temperature affects through the term , which represents an energy barrier “penalty” on the reaction’s progress.
Meaning of Each Term
Activation energy,
Activation energy is the energy barrier separating reactants from the transition state.
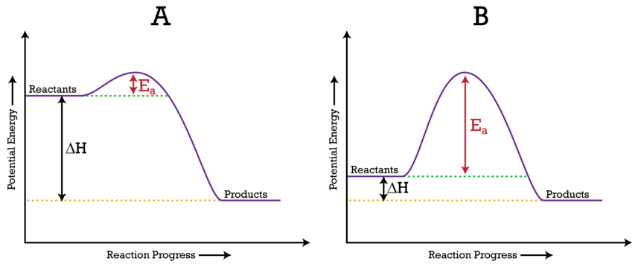
Potential energy (reaction coordinate) diagrams comparing a lower and higher activation energy barrier. The vertical gap from reactants to the peak is labeled , illustrating why a larger barrier corresponds to a slower reaction at the same temperature. The diagrams also show , emphasizing that kinetics (barrier height) is distinct from overall energy change. Source
A larger means fewer collisions have enough energy to react at a given temperature, so is smaller.
Activation energy (): The minimum energy input required for reactants to reach the transition state and undergo the elementary bond changes leading toward products.
Frequency factor,
The frequency factor groups together factors that influence how often collisions occur and how often they are oriented in a way that can lead to reaction. Conceptually:
Larger tends to increase (more “attempts” at reaction per unit time).
does not remove the barrier; it multiplies the barrier-controlled exponential term.
How Temperature Changes the Rate Constant
Temperature increases primarily because it increases the fraction of particles with enough kinetic energy to overcome . Conceptually:
As increases, becomes less negative.
Therefore, becomes larger.
So increases, often dramatically over moderate temperature ranges.
Important qualitative implications:
Reactions with larger are typically more temperature sensitive (their changes more when changes).
The Arrhenius model explains why small temperature increases can cause noticeable rate increases in many chemical systems.
Graphical Interpretation (No Calculations)
A common conceptual use is the linearised idea: plotting versus gives a straight line.
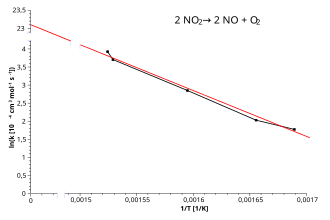
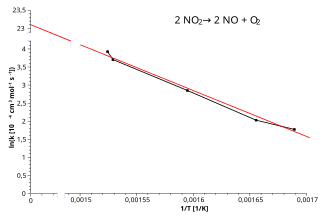
An Arrhenius plot showing versus with a straight best-fit line. The negative slope visually encodes that decreases as increases (i.e., as temperature decreases). Qualitatively, a steeper downward line corresponds to a larger activation energy barrier. Source
The line’s slope is negative, reflecting that decreases as increases (i.e., as temperature decreases).
A steeper (more negative) slope corresponds to a larger .
Two reactions can be compared qualitatively by comparing the steepness of their lines.
What the AP Exam Emphasises
Because calculations are not assessed here, focus on these takeaways:
The Arrhenius equation connects temperature dependence of rate to activation energy.
Increasing temperature increases , increasing reaction rate (if concentrations are unchanged).
Larger activation energy reduces at a given temperature and makes more sensitive to temperature changes.
FAQ
Because the exponential term uses $RT$, and $R$ is defined per kelvin.
Using °C would shift the zero point and distort the exponential dependence.
$A$ has the same units as $k$.
So its units depend on overall order (e.g., s$^{-1}$ for first order).
Yes, slightly, because collision frequency and orientation probabilities can vary with $T$.
In many introductory treatments, the exponential term dominates the temperature effect.
It can indicate the mechanism changes with temperature, or that $E_a$ is not constant over the range.
It may also reflect competing pathways.
Taking $\ln$ converts multiplicative/exponential relationships into additive/linear ones.
This makes trends easier to visualise and compare across temperatures.
Practice Questions
Q1 (2 marks): Using the Arrhenius equation, state how increasing temperature affects , and how a larger affects at the same temperature.
increases increases (1)
Larger at same smaller (1)
Q2 (5 marks): Two reactions, X and Y, have straight-line plots of versus . Reaction X has a steeper negative slope than reaction Y. Explain what this indicates about their activation energies and temperature sensitivities, using Arrhenius ideas.
On a vs plot, a steeper negative slope implies larger (2)
Therefore reaction X has larger than Y (1)
Larger means changes more with (greater temperature sensitivity) (1)
So X’s rate constant increases more rapidly with increasing than Y’s (1)

