AP Syllabus focus: ‘Temperature changes in a system indicate that the system’s energy is changing.’
Temperature is one of the most accessible signals of energy change in chemistry. By linking thermometer readings to particle motion, you can infer whether energy entered or left a system during a physical or chemical process.
Core idea: temperature is evidence of changing energy
A measurable temperature change indicates that a system’s energy has changed because temperature tracks how particle motion is distributed within the sample. When energy is transferred into or out of a system, some of that transfer often appears as a change in the system’s temperature.
Temperature is not “how much heat” something has
It is essential to separate what a thermometer reads from the broader idea of energy stored or transferred.
Temperature — a measure proportional to the average kinetic energy of the particles in a sample.
A hot object is not necessarily “high energy” in an absolute sense; it has particles with higher average kinetic energy, but the total energy also depends on the amount of matter and how energy is stored (motion vs other forms).
Thermal energy — the energy associated with the random motion of particles in a substance; it depends on both temperature and the quantity of matter.
A small spark can have a very high temperature but relatively little thermal energy because it contains very little matter.
Particle-level link between temperature and energy
At the microscopic level, temperature reflects the average kinetic energy of particles (translational motion for atoms; translational, rotational, and vibrational motion for molecules). When a system’s temperature increases, the distribution of particle speeds shifts so that, on average, particles move faster.
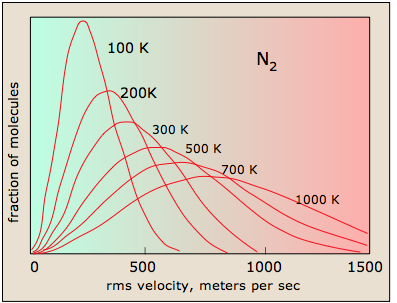
Maxwell–Boltzmann speed distributions for the same gas at multiple temperatures. As increases, the curve shifts right (higher typical speeds) and flattens (a wider spread of particle speeds), reinforcing that temperature reflects how kinetic energy is distributed across particles. Source
When temperature decreases, average particle speed decreases.
This is why a thermometer can serve as an indirect indicator that energy has been transferred and has altered particle kinetic energy.
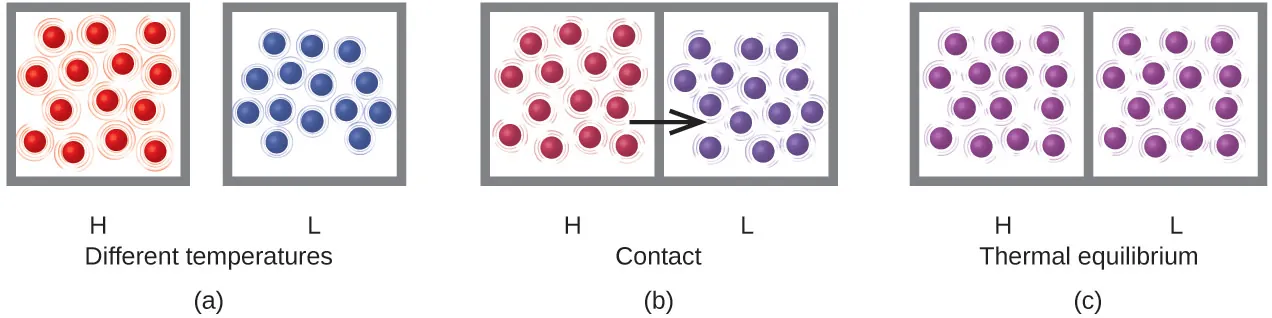
Particle-level diagram of heat transfer from a hotter object to a cooler one until thermal equilibrium is reached. The image links temperature differences to differences in average molecular kinetic energy and shows how collisions redistribute energy between objects. Source
= average kinetic energy of particles in the sample
= absolute temperature in kelvin (K)
Because the proportionality uses kelvin, a change measured in degrees Celsius corresponds to the same-size change in kelvin, but absolute temperature must be in kelvin to connect directly to particle motion.
Interpreting a temperature change as an energy change
A temperature change tells you that the system’s energy changed, but you must be careful about what exactly the thermometer is monitoring and where the energy went.
What a temperature increase implies
If the system’s temperature increases, then:
The particles’ average kinetic energy increased.
Energy was transferred to the system or converted into particle kinetic energy within the system.
The system’s energy is therefore higher than it was before the change.
Temperature rise is evidence of an energy increase, not proof of the mechanism.
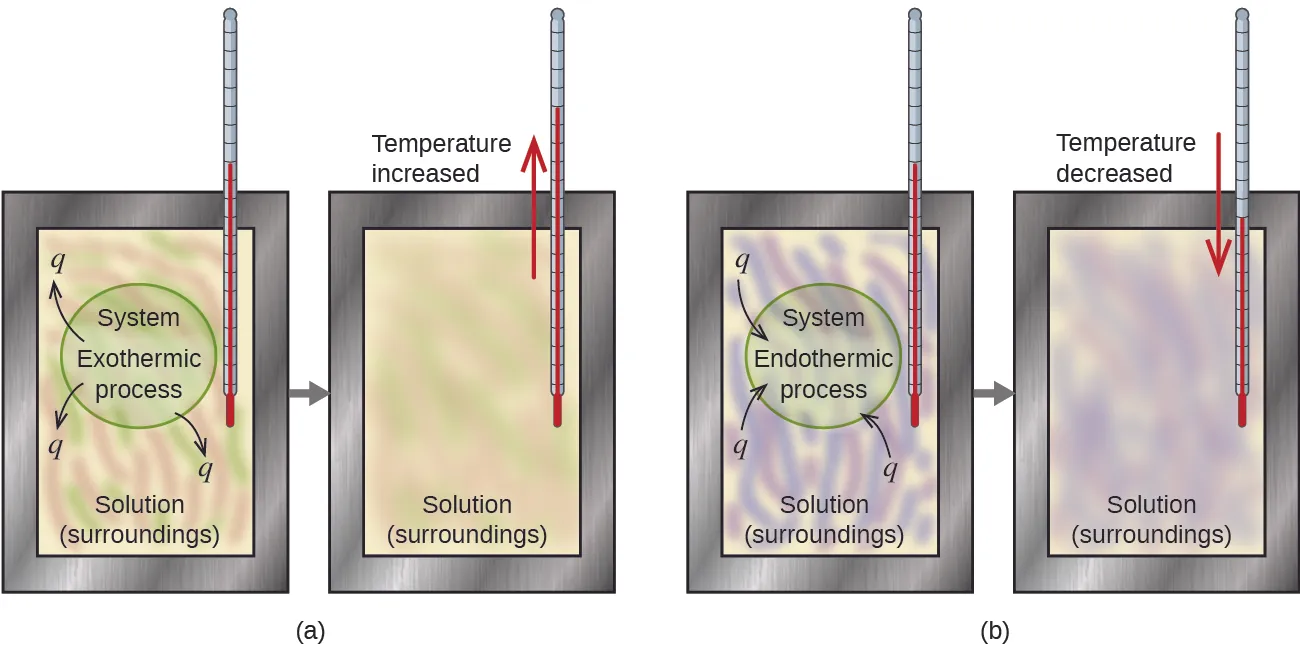
Calorimetry schematic comparing exothermic and endothermic processes, emphasizing how the direction of temperature change reflects energy transfer. The figure ties an observed to the sign convention for heat (system vs surroundings) in a controlled measurement setup. Source
The energy could come from the surroundings, from electrical input, or from internal conversion of stored energy into kinetic energy.
What a temperature decrease implies
If the system’s temperature decreases, then:
The particles’ average kinetic energy decreased.
Energy left the system or was converted from kinetic energy into another form.
The system’s energy is therefore lower than it was before the change.
In practice, a cooling system often indicates energy transfer away from the system, but the direction of transfer must be inferred from how the experiment is set up and what is defined as “the system.”
Using temperature change as experimental evidence
Temperature is a convenient observable because it can be tracked continuously as a process occurs. In AP Chemistry contexts, you commonly use temperature change to support claims such as:
“Energy was transferred during this process.”
“The system’s energy increased/decreased during the observed interval.”
“A process caused a measurable change in particle kinetic energy.”
Common measurement considerations (what can obscure the evidence)
Real measurements can complicate the link between temperature change and energy change.
Heat exchange with the environment: If the container is not well insulated, the measured temperature change may be smaller than the true change caused by the process.
Incomplete mixing: Temperature gradients can form; the thermometer may sample a warmer or cooler region than the bulk.
Response time and placement: A probe measures its immediate surroundings; delays can make the maximum or minimum temperature easy to miss.
Multiple processes happening at once: Simultaneous energy transfers (for example, warming from the room while another process cools the sample) can partially cancel, producing a small net temperature change.
No temperature change does not always mean no energy change
A constant temperature reading is not, by itself, proof that energy is not changing. A system can exchange energy while maintaining nearly constant temperature if energy is being redistributed in ways a thermometer does not directly capture, or if energy transfer in and out is balanced over the observation time. Therefore, temperature data are best treated as evidence that must be interpreted in context rather than a standalone verdict.
FAQ
Kelvin is an absolute scale with zero at the point where thermal motion is minimised in the classical model.
Using kelvin preserves proportional relationships between temperature and average kinetic energy.
Yes. Thermal energy depends on both temperature and the amount and nature of the substance.
A larger mass (or a substance that stores more energy per degree) can contain more thermal energy at the same temperature.
A probe measures the local temperature around its tip.
If the beaker is not well stirred, different regions can be different temperatures, so placement can bias the recorded change.
Good insulation or a lid to reduce environmental heat exchange
Consistent stirring to reduce temperature gradients
Fast-response probe and frequent readings to capture peaks/troughs
Small differences in initial temperatures, mixing rate, timing, evaporation, or heat exchange with the bench and air can change the net observed temperature shift.
Instrument resolution and calibration also contribute to variability.
Practice Questions
(2 marks) A student observes that the temperature of a reaction mixture increases by 6 °C after two solutions are combined. What does this observation indicate about the energy of the system? Explain briefly.
1 mark: States that the system’s energy increased (or particles’ average kinetic energy increased).
1 mark: Links temperature rise to increased average kinetic energy / energy transfer evidenced by temperature change.
(5 marks) A student uses a temperature probe to monitor a process in a beaker and records only a small temperature decrease. Give two reasons why the measured temperature change might underestimate the true energy change of the system, and explain how each reason affects the temperature reading.
1 mark: Identifies heat exchange with surroundings (poor insulation).
1 mark: Explains that heat transfer to/from the environment reduces the magnitude of the observed temperature change.
1 mark: Identifies incomplete mixing / temperature gradients OR probe placement/response time.
1 mark: Explains how non-uniform temperature or probe lag leads to an incorrect/smaller recorded change.
1 mark: Provides a second valid reason with a correct explanation (e.g., simultaneous competing energy transfers; recording interval misses the minimum).

