AP Syllabus focus: ‘In reactions, the system’s energy decreases (exothermic), increases (endothermic), or stays the same; energy is exchanged with the surroundings as heat or work.’
Chemical reactions redistribute energy between what you are studying and everything else. To predict temperature changes and energy signs, you must track energy flow across the boundary between the system and the surroundings.
System vs. Surroundings in a Chemical Reaction
Defining what you track
System: The part of the universe chosen for study (in thermochemistry, typically the reacting chemicals and any products).
A reaction’s energy change is described for the system, but it is observed through its interaction with what lies outside it.
Surroundings: Everything outside the system that can exchange energy with it (solution, container, air, thermometer, etc.).
The boundary between system and surroundings can be real (walls of a calorimeter) or conceptual; choosing it carefully helps you interpret measurements consistently.
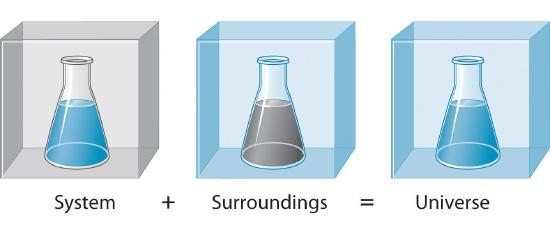
This diagram partitions the universe into the system (chemicals of interest) and the surroundings (everything else). It visually reinforces that any energy transfer you measure occurs across the system boundary, while the “universe” is the combined total. Source
What “energy flow” means for reactions
During a reaction, the system can:
Lose energy to the surroundings (often observed as warming outside the system)
Gain energy from the surroundings (often observed as cooling outside the system)
Have no net energy change overall (energy transfers may occur, but net change is zero)
Energy crosses the boundary in two main ways:
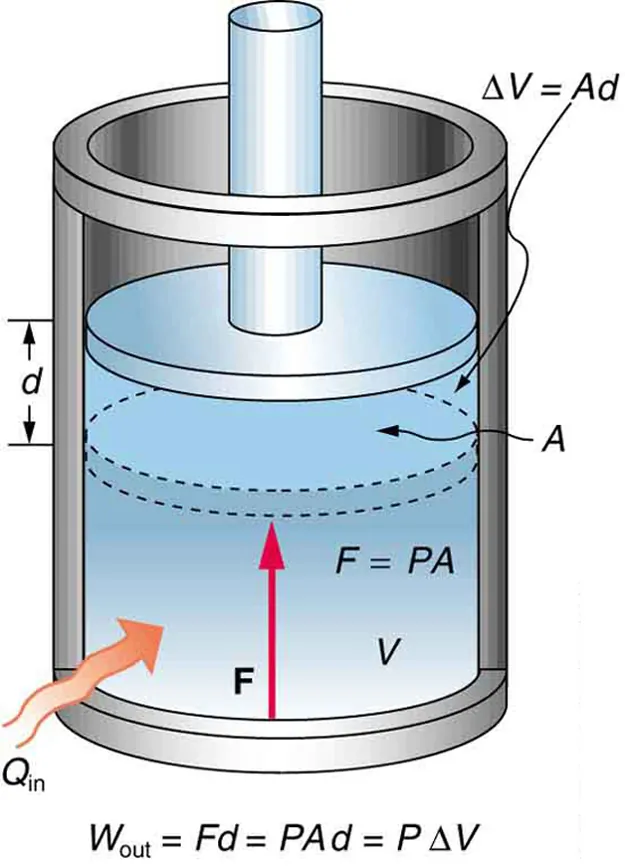
This piston–cylinder diagram shows a gas expanding at constant pressure and doing mechanical work on the surroundings. The figure ties the geometry of expansion to the work relationship , making the sign and physical meaning of pressure–volume work easier to interpret. Source
Heat (due to temperature difference)
Work (organized energy transfer, such as expansion against pressure)
Heat and Work: The Two Pathways
Heat transfer (q)
Heat (q): Energy transferred between system and surroundings due to a temperature difference.
In thermochemistry sign convention (from the system’s perspective):
: system absorbs heat (surroundings supply heat)
: system releases heat (surroundings absorb heat)
A reaction described as exothermic corresponds to a net release of energy from the system as heat under the stated conditions; endothermic corresponds to net absorption.
Work (w)
Work (w): Energy transferred when a force acts through a distance; in chemistry, commonly pressure–volume work when gases expand or compress.
Common cases in reaction chemistry:
Gas produced and expands: system often does work on surroundings (energy leaves system as work)
Gas consumed and volume decreases: surroundings can do work on system (energy enters system as work)
Energy Conservation for System + Surroundings
Energy is not created or destroyed; it is transferred. For any process, the combined energy change of system and surroundings is zero, so tracking one automatically determines the other.
= change in system internal energy, J
= heat absorbed by the system, J
= work done on the system, J
= heat absorbed by the surroundings, J
= work done on the surroundings, J
This framing matches the syllabus focus: in reactions, the system’s energy can decrease, increase, or stay the same, and the exchange with the surroundings occurs as heat or work.
Linking Energy Change to Exothermic vs. Endothermic
Interpreting “system’s energy decreases/increases”
If the system’s energy decreases, the energy must have left the system:
commonly as heat released to surroundings (exothermic)
and/or as work done by the system on surroundings
If the system’s energy increases, energy must have entered the system:
commonly as heat absorbed from surroundings (endothermic)
and/or as work done on the system by surroundings
If the system’s energy stays the same (), then:
heat and work may still occur, but they balance so
Why the “system perspective” matters
AP problems expect consistent sign conventions:
“Heat released by the reaction” refers to
“Heat absorbed by the reaction” refers to
Temperature change measured in the surroundings reflects , which has the opposite sign of
This is why a warming solution often indicates the surroundings gained heat, meaning the system released heat, consistent with an exothermic reaction description.
FAQ
Choose a boundary that matches what you can measure reliably.
If you measure the solution’s temperature change, the solution is often treated as the surroundings to the reacting species.
Include or exclude the container depending on whether its heat absorption is negligible.
Because energy can leave as heat while an equal amount enters as work (or vice versa).
For instance, $q_{sys}<0$ and $w_{sys}>0$ with equal magnitudes gives $\Delta E_{sys}=0$.
“Heat released” is about energy transfer ($q$), while temperature change depends on the heat capacity of what absorbs that heat.
A small temperature rise can still correspond to substantial heat if the surroundings have a large heat capacity.
Not necessarily. Expansion work means $w_{sys}<0$, which decreases $\Delta E_{sys}$ relative to $q_{sys}$.
Whether the reaction is termed exothermic depends on the direction of heat flow ($q_{sys}$), not directly on work.
Because chemistry uses the convention “work done on the system is positive”.
Compression: $w_{sys}>0$
Expansion: $w_{sys}<0$
Stating explicitly “on the system” vs “by the system” prevents sign errors.
Practice Questions
(2 marks) For a reaction carried out in a beaker, the solution temperature increases. From the system perspective, state the sign of and whether the process is exothermic or endothermic.
(1)
Exothermic (1)
(5 marks) A reaction occurs in a piston-cylinder and produces gas, causing the piston to rise. The surroundings gain of heat during the reaction, and the system does of work on the surroundings. Determine , , and , with correct signs.
Uses so (1)
Recognises “system does work on surroundings” so and (2)
Uses (1)
(1)

