AP Syllabus focus: ‘Dissolving can be endothermic or exothermic, depending on the relative strengths of interactions before and after dissolution.’
Dissolving a substance in a solvent involves rearranging particles and the forces between them. Whether the temperature rises or falls depends on how much energy is required versus released as new interactions form.
What “dissolving” means at the particle level
A solution forms when solute particles become uniformly dispersed among solvent particles. This requires overcoming some existing attractions and creating new ones.
Key interparticle attractions involved
Solute–solute attractions: forces holding solute particles together (ionic attractions, metallic bonding, or intermolecular forces in molecular solids)
Solvent–solvent attractions: forces between solvent molecules (for water, strong hydrogen bonding)
Solute–solvent attractions: new interactions formed upon dissolution (e.g., ion–dipole attractions when ions dissolve in water)
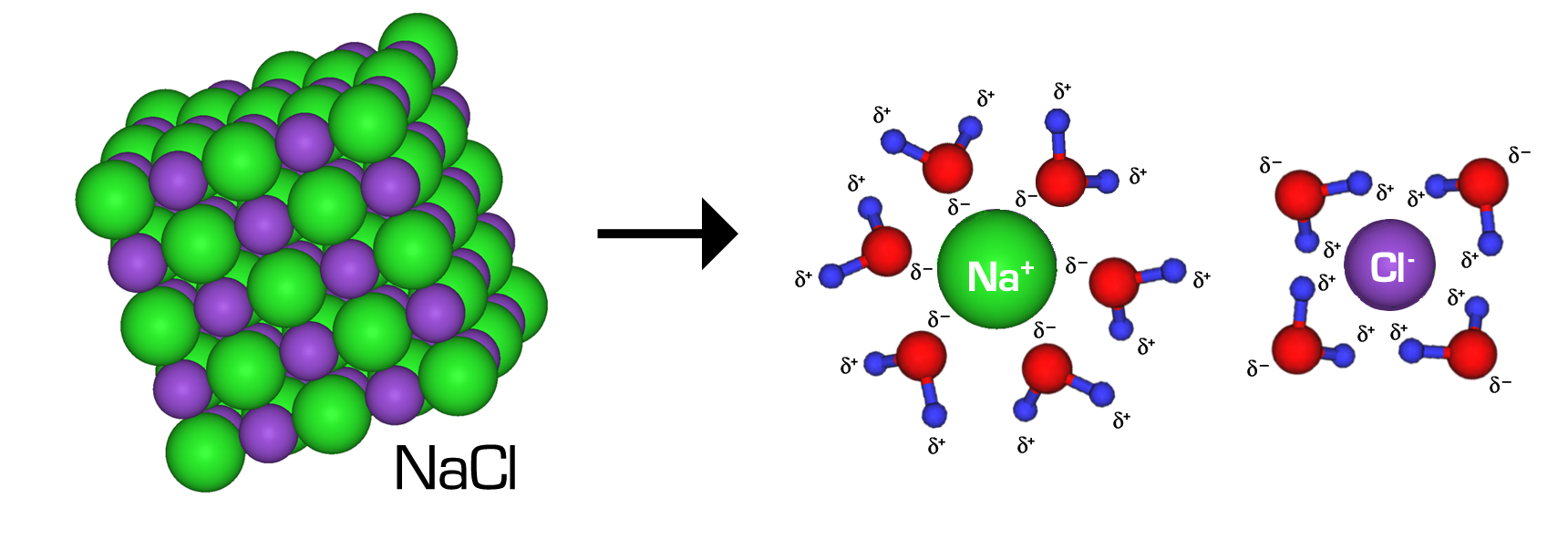
An NaCl crystal breaks apart into solvated ions as water molecules orient their partial charges around and . The diagram highlights hydration shells (ion–dipole interactions) that stabilize separated ions and help prevent recombination into the lattice. Source
When comparing energies, focus on the relative strengths of these interactions before and after dissolution.
Energy changes during solution formation
Solution formation can be viewed in energetic steps (conceptual, not necessarily separate in time):
Separating solute particles (breaking solute–solute attractions): requires energy
Separating solvent particles (making “space” in the solvent): requires energy
Bringing solute and solvent particles together (forming solute–solvent attractions): releases energy
If the energy released in forming solute–solvent attractions is larger than the energy required to separate solute and solvent particles, the overall dissolving process is exothermic. If it is smaller, the process is endothermic.
Solvation and hydration
When the solvent surrounds and stabilises solute particles, new attractions lower potential energy.
Solvation: The process in which solvent particles surround and interact with solute particles; in water this is called hydration.
In ionic compounds dissolving in water, hydration involves strong ion–dipole attractions between ions and the partially charged ends of water molecules, often competing with the ionic attractions in the crystal.
Enthalpy of solution: organising the energy bookkeeping
Chemists often summarise the overall heat effect of dissolving using the enthalpy change of solution, which reflects the net result of “energy in” and “energy out” from interaction changes.
Enthalpy of solution (): The enthalpy change when a specified amount of solute dissolves in a specified amount of solvent to form a solution.
A useful way to express the idea “separate then mix” is:
= net enthalpy change for dissolution (kJ mol)
= energy required to overcome solute–solute attractions (kJ mol)
= energy required to overcome solvent–solvent attractions (kJ mol)
= energy released when solute–solvent attractions form (typically negative) (kJ mol)
This relationship emphasises the syllabus idea: dissolving is endothermic or exothermic depending on the relative strengths of interactions before and after dissolution.
Interpreting endothermic vs exothermic dissolving using forces
Endothermic dissolving (solution cools)
Solute–solute and/or solvent–solvent attractions are strong and costly to disrupt
Solute–solvent attractions formed are comparatively weaker
Net result: more energy absorbed than released, so
Common particle-level intuition: if water must disrupt extensive hydrogen bonding but the new interactions do not compensate fully, the process tends to be endothermic.
Exothermic dissolving (solution warms)
Solute–solvent attractions formed are very strong (large energy release)
They outweigh the energy needed to separate solute and solvent particles
Net result: more energy released than absorbed, so
This often occurs when hydration/solvation is especially favourable, producing strong stabilising interactions.
“Like dissolves like” as an energy idea
“Like dissolves like” reflects the energetic requirement that solute–solvent attractions should be comparable in strength to the attractions being replaced.

Oil and water separate into distinct layers because their intermolecular attractions are not comparable in strength (polar hydrogen-bonding network vs. nonpolar dispersion-dominated interactions). This macroscopic phase separation is the everyday evidence behind the rule-of-thumb “like dissolves like.” Source
Polar/ionic solutes tend to dissolve in polar solvents (strong new attractions possible)
Nonpolar solutes tend to dissolve in nonpolar solvents (dispersion forces can be similar in both)
FAQ
No. Stirring mainly changes the rate at which solute and solvent make contact.
It can, however, make the temperature change easier to observe by helping the solution reach a uniform temperature more quickly.
“Disappearing” means particles disperse; it does not indicate energy release.
If disrupting the crystal and rearranging water molecules costs more energy than hydration releases, thermal energy is drawn from the surroundings, so it feels cold.
Higher charge and smaller ionic radius generally increase charge density, strengthening ion–dipole attractions.
Stronger ion–dipole attractions usually make hydration more exothermic, increasing the magnitude of energy released upon solvation.
Yes. Solubility reflects overall favourability, which can include factors beyond enthalpy.
Two solutes may both form solutions, yet one may release more energy on solvation (exothermic) while another absorbs more (endothermic) due to different interaction strengths.
As concentration increases, new solute particles interact with an environment that is already partly structured by dissolved solute.
This can change effective solute–solvent and solvent–solvent interactions (e.g., fewer “free” solvent molecules), so the measured heat effect per mole can vary with concentration.
Practice Questions
Q1 (1–3 marks) A student dissolves substance X in water and observes the solution temperature decreases. Classify the dissolving process as endothermic or exothermic and state what this implies about the relative strengths of interactions before and after dissolution.
Correct classification: endothermic (1)
States that more energy is required to separate solute–solute and/or solvent–solvent attractions than is released by forming solute–solvent attractions (1)
Links to weaker solute–solvent attractions compared with those disrupted (1)
Q2 (4–6 marks) Explain, in terms of interparticle forces, why dissolving an ionic solid in water can be either exothermic or endothermic. Your answer should refer to (i) separating particles and (ii) forming new interactions.
Mentions breaking/overcoming solute–solute attractions in the ionic lattice requires energy (1)
Mentions separating water molecules (overcoming solvent–solvent attractions, e.g. hydrogen bonding) requires energy (1)
Mentions formation of ion–dipole attractions during hydration releases energy (1)
Explains exothermic when hydration energy released exceeds energy required for separation (1)
Explains endothermic when hydration energy released is less than energy required for separation (1)
Uses clear comparative language about relative interaction strengths before vs after (1)

