AP Syllabus focus: ‘Many processes can be broken into a series of steps, and each step has its own energy change.’
Thermochemical problems often involve energy changes that are difficult to measure directly. A powerful approach is to represent the overall process as multiple simpler steps, each with a known or measurable energy change.
What it means to “break a process into steps”
A process (physical change or chemical reaction) can be represented as a sequence of intermediate changes that, when conceptually followed in order, produce the same overall initial and final states.
Thermochemical pathway: A conceptual sequence of steps that connects the same initial state to the same final state as the overall process, with an associated energy change for each step.
The key idea in this subtopic is organisational: you are not changing what the overall process is, only choosing an alternate route made of smaller pieces so you can track individual energy changes more easily.
What counts as a “step”?
A valid step is a clearly written transformation with its own stated energy change, such as:
A reaction written as a balanced chemical equation
A physical change (for example, changing phase or dissolving), if you are given an energy change for that change
A conversion between specified chemical forms (for example, from reactants to an intermediate and then to products), as long as each conversion is explicitly stated
Each step must be defined precisely enough that an energy change can be attached to it (given, measured, or determined elsewhere).
How to represent steps and their energy changes clearly
When constructing or reading a multistep breakdown, focus on clarity and consistency.
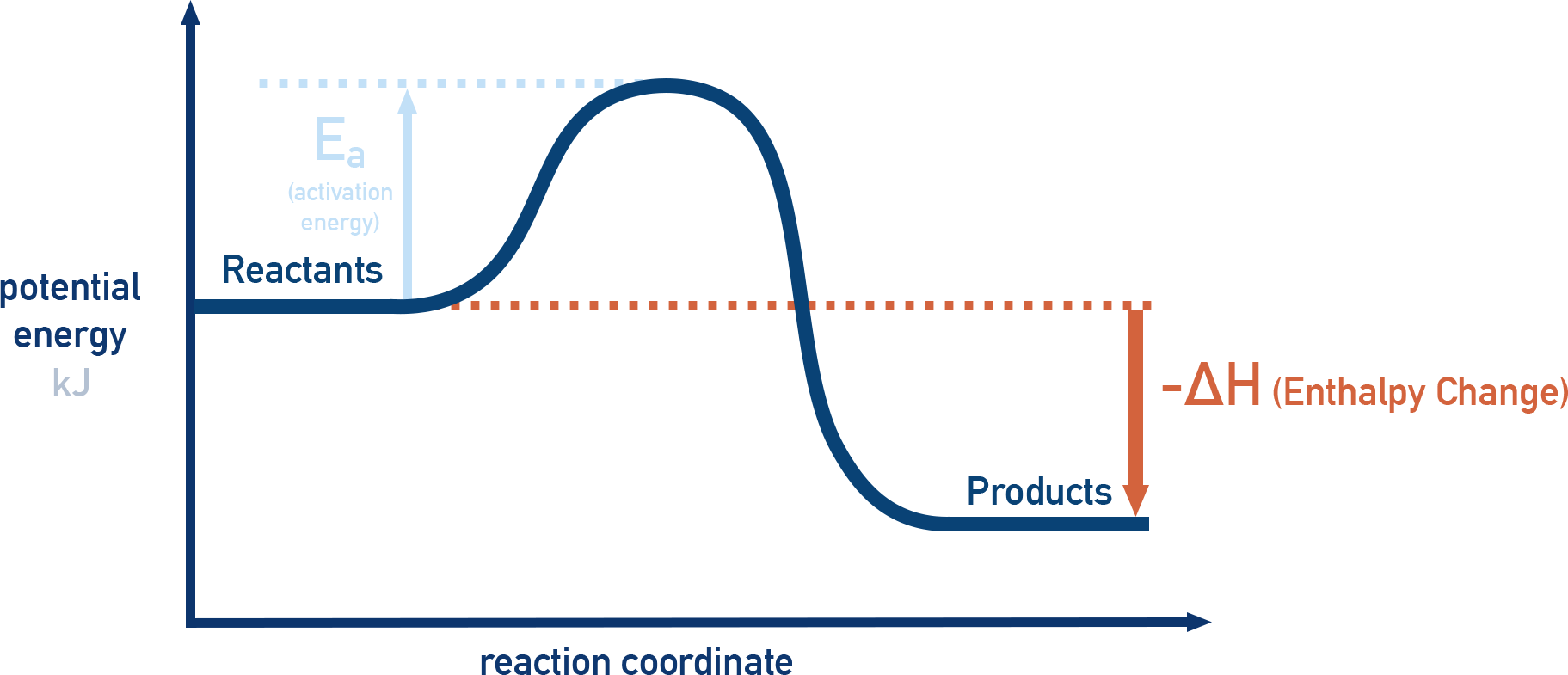
Reaction-coordinate energy profile for an exothermic process, with reactants higher than products and . The diagram labels both the activation energy (kinetic barrier) and the overall enthalpy change, helping separate “pathway shape” from the net energy difference between initial and final states. Source
Writing each step
For each step, ensure:
Chemical formulas and states are included when provided (e.g., (s), (l), (g), (aq)), because energy changes depend on physical state
The step is balanced (atoms conserved) so it represents a physically meaningful change
The energy change is labelled per the step, typically as ΔH with units (commonly kJ or kJ/mol as stated)
Keeping the overall target in view
A stepwise breakdown should be checkable by inspection:
If you conceptually “add up” the steps, anything that is created in one step and consumed in a later step is an intermediate and should cancel out in the overall description.
What remains after cancellation should match the overall process you are trying to represent.
This cancellation check is an essential skill because it verifies that the steps genuinely correspond to the same overall change rather than a different process.
Tracking energy changes across a multistep pathway
Each step has its own energy change, and the purpose of the pathway is to organise those changes so the overall energy change can be determined from the collection of steps.
A compact way to express this bookkeeping is:
= enthalpy change for the overall process, in kJ (or as specified)
= enthalpy change for an individual step in the pathway, in kJ (or as specified)
In practice, you should pay close attention to how each step’s energy change is stated (per reaction as written, per mole of a substance, or for a given mass), because the step’s written description and its energy label must refer to the same “amount of process.”
Why chemists use stepwise breakdowns
Breaking a process into steps is especially useful when:
The overall process is hard to run directly (too slow, too fast, unsafe, or produces side reactions)
The energy change is hard to measure directly (poor temperature signal, large heat losses, or experimental constraints)
Reliable energy changes are available for related, simpler transformations that can be combined into the desired overall change
The stepwise approach is also a communication tool: it makes clear which pieces of information are being used and how they relate to the target process.
Common pitfalls when identifying or building steps
Mixing steps that do not connect the same initial and final states (a pathway must start and end correctly)
Omitting states or using inconsistent states across steps
Attaching an energy change to an equation that is not balanced or not written to match the stated energy change
Failing to recognise an intermediate, leading to an overall result that includes species that should cancel
FAQ
Choose steps that are explicitly definable and whose energy changes are available or measurable.
Prioritise steps that share the same species and physical states as the overall process so cancellation is straightforward.
Because $X(s)$, $X(l)$, $X(g)$, and $X(aq)$ are different thermodynamic states.
A stepwise pathway that changes state implicitly changes the type of energy change being tracked, so missing states can invalidate the breakdown.
A reactant appears in the overall process (it is present initially).
An intermediate is produced in one step and consumed in another, so it cancels when the steps are combined and does not appear in the overall process.
It can, but it becomes harder to verify and to match an energy change to the step.
A safer approach is to separate distinct transformations (e.g., dissolve, then react) into separate steps so each has a clearly assigned energy change.
After conceptually combining the steps, check for:
Extra species left over (not cancelling)
Missing species from the overall process
Mismatched states (e.g., $Z(s)$ instead of $Z(aq)$)
Any of these indicate the steps describe a different net change.
Practice Questions
Q1 (2 marks) A process is represented by two steps: Step 1: with energy change Step 2: with energy change State the overall process and write an expression for the overall energy change.
Overall process stated: (1)
Expression: (1)
Q2 (5 marks) A student proposes the following steps to represent an overall process: Overall:
Step 1: with energy change
Step 2: with energy change
Step 3: with energy change
(a) Identify the intermediate species (1 mark).
(b) Explain how you would check, using only the written steps, that these steps correspond to the stated overall process (2 marks).
(c) Write an expression for in terms of (2 marks).
(a) Intermediates: and (1)
(b) Method: add steps and cancel species appearing on both sides; confirm remaining species and states match overall (2)
(c) (2)

