AP Syllabus focus: ‘If a reaction is reversed, the enthalpy change keeps the same magnitude but changes sign.’
Reversing a chemical equation reverses the direction of energy flow between system and surroundings.
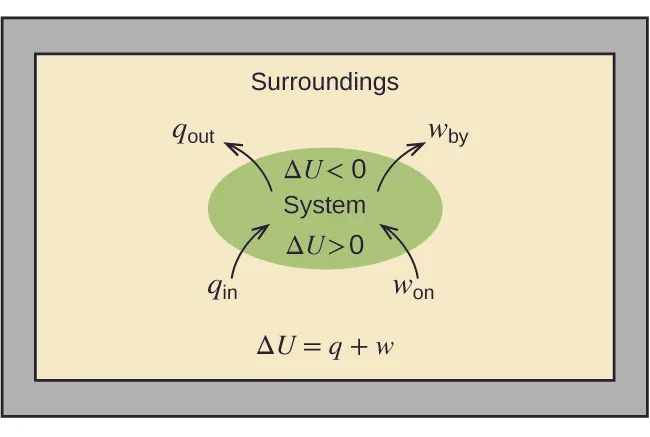
A system is drawn inside its surroundings with labeled arrows for heat and work crossing the boundary. The diagram emphasizes that energy flow direction determines the sign convention (inward heat/work increases the system’s energy; outward decreases it), which is the same logic used when interpreting the sign of for reactions written forward vs. reverse. Source
On the AP Exam, you must connect the written reaction direction to the sign of and interpret what that means physically.
Core idea: reversing a reaction reverses heat flow
A chemical reaction can be written in the forward direction or the reverse direction. The enthalpy change reported for the reaction is tied to the reaction as written.
If the forward reaction releases heat to the surroundings, the reverse reaction must absorb that same amount of heat from the surroundings.
If the forward reaction absorbs heat, the reverse reaction must release that same amount of heat.
Therefore, the numerical value stays the same, but the sign flips.
What “same magnitude, opposite sign” means
“Same magnitude” means the size of the energy change is unchanged (e.g., 125 kJ stays 125 kJ). “Opposite sign” means positive becomes negative, and negative becomes positive.
Enthalpy change is a property of the initial and final states
This rule works because enthalpy depends on the starting and ending states of the system. Reversing the reaction swaps those states, so the energy difference changes direction.
Enthalpy change, : The heat transferred at constant pressure for a process, reported with a sign to indicate whether heat is absorbed by the system () or released by the system ().
Because the sign of communicates the direction of heat transfer, reversing the chemical equation must reverse the sign to stay consistent with energy conservation.
Thermochemical equations: the reaction and are paired
A thermochemical equation is a balanced chemical equation written together with its value. The key rule is that the written equation and the stated form a matched set.
Forward equation written with describes an exothermic direction.
Reverse equation written with describes an endothermic direction.
You must reverse the equation exactly (reactants/products swap); then flip the sign of .
The sign flip rule (how to write it)
Use a compact statement to keep direction and sign consistent.
= Enthalpy change for the reaction written in reverse, in kJ (or kJ/mol as given)
= Enthalpy change for the reaction written in the forward direction, in kJ (or kJ/mol as given)
This relationship applies whether the process is described per reaction as written or per mole of a specified substance, as long as you are truly reversing the same reaction.
Common AP pitfalls when reversing reactions
Forgetting that is attached to the equation direction
If you flip reactants and products but keep the same sign, you are describing the wrong heat flow.
If you keep the equation the same but flip the sign, you are also describing a different process than the one written.
Losing track of “system” vs. “surroundings”
: heat leaves the system (surroundings warm).
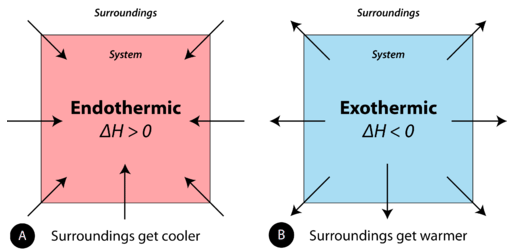
Endothermic and exothermic processes are shown as system–surroundings energy transfers, with arrows indicating the direction of heat flow. The figure directly ties the sign of to the physical observation: surroundings cool when the system absorbs heat () and warm when the system releases heat (). Source
: heat enters the system (surroundings cool).
When you reverse the reaction, which side “gains” chemical potential energy versus “loses” it is reversed, so the heat direction must reverse too.
Not keeping physical states consistent
Reversal assumes the same reactants and products in the same physical states (s, l, g, aq). If states change, it is not the exact reverse process, and the enthalpy change might not be a simple sign flip.
Confusing reversal with other equation changes
For this rule, only the direction changes. The coefficients remain exactly what they were in the original equation; only reactants and products swap sides.
FAQ
Because the same two states are involved, just swapped.
The energy difference between those states is fixed; reversing only changes the direction of the change.
Yes for the thermochemical equations describing forward vs reverse processes.
Equilibrium means both directions occur, but the defined $\Delta H$ values for forward and reverse remain equal in magnitude and opposite in sign.
No; keep the same units and magnitude, and flip the sign.
The “per mole” basis stays tied to the reaction as written (same stoichiometric event, opposite direction).
$\Delta H$ depends on the physical states of reactants and products.
If states change (e.g., $g$ vs $aq$), you are not writing the true reverse of the same process, so a simple sign flip may not apply.
Yes, conceptually: exothermic forward reactions can be written with “heat” on the product side, and the reverse with “heat” on the reactant side.
However, AP Chemistry typically expects the sign-change approach using $\Delta H$.
Practice Questions
Q1 (2 marks) The thermochemical equation is: Write the reverse reaction and state its .
Correct reverse equation written (reactants/products swapped, balanced): 1 mark
Correct : 1 mark
Q2 (5 marks) A student is given: (a) State whether the forward reaction is exothermic or endothermic. (1) (b) Write the thermochemical equation for the reverse reaction, including . (2) (c) Explain, in terms of heat flow between system and surroundings, why the sign of changes when the reaction is reversed. (2)
Exothermic: 1 mark (b)
Correct reverse chemical equation: 1 mark
Correct : 1 mark (c)
Forward: heat released from system to surroundings; reverse: heat absorbed from surroundings to system: 1 mark
Therefore energy transfer direction reverses, so must change sign while keeping magnitude: 1 mark

