AP Syllabus focus: ‘If a reaction equation is multiplied by a factor, the enthalpy change is multiplied by the same factor.’
Scaling chemical equations changes the amount of reaction being described. Because enthalpy is an extensive quantity, the reported enthalpy change must scale proportionally with the stoichiometric factor used in the balanced equation.
What “scaling a reaction” means
A thermochemical equation pairs a balanced chemical equation with a ΔH value for that equation as written. Scaling refers to multiplying every stoichiometric coefficient by the same number (including fractions).
Multiply coefficients by (e.g., double, triple): you describe times as much reaction.
Multiply coefficients by a fraction (e.g., divide by 2): you describe a proportional fraction of the reaction.
Because ΔH is tied to the reaction extent, it must be scaled exactly the same way.
Enthalpy is extensive (amount-dependent)
Enthalpy change (ΔH): The heat transferred at constant pressure for a process as written, with units typically in kJ for the stated reaction amount.
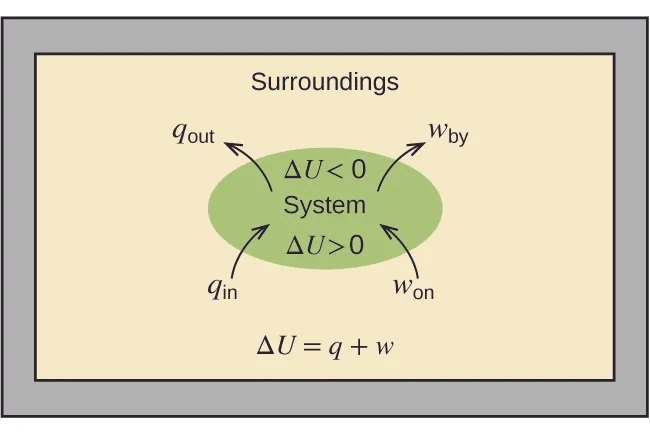
System–surroundings energy-flow diagram illustrating the first-law bookkeeping: heat and work cross the boundary and change the system’s internal energy. This kind of boundary diagram helps anchor the idea that measured heat at constant pressure corresponds to an enthalpy change for the process as written, and that energy transfer scales with how much process occurs. Source
A key implication is that ΔH is not “per molecule” automatically; it is for the stoichiometric amounts shown in the equation. If the equation’s mole amounts change, the energy change changes in direct proportion.
The scaling rule (what you must do)
When a reaction equation is multiplied by a factor, the enthalpy change is multiplied by the same factor. This is a direct statement of the syllabus focus and is treated as a rule for manipulating thermochemical equations.
= enthalpy change for the scaled equation (kJ)
= scaling factor applied to all coefficients (unitless)
= enthalpy change for the original equation as written (kJ)
Use the sign of ΔH exactly as given and scale the numerical value by :
If ΔH is negative (exothermic), scaling by makes it more negative by that factor.
If ΔH is positive (endothermic), scaling by makes it more positive by that factor.
Common scaling situations to recognise
Doubling, tripling, or halving the equation
If you double all coefficients, set and double ΔH.
If you halve all coefficients, set and halve ΔH.
Fractional coefficients are allowed
Thermochemical equations may legitimately contain fractions to match a desired “per 1 mol” basis. The same scaling rule still applies: if coefficients are divided by 4, then ΔH is divided by 4.
Scaling is not the same as reversing
Scaling changes the magnitude only (by a factor). Reversing changes the sign; do not confuse the two operations when editing equations.
How to apply the rule reliably (method)
Identify the exact factor relating the new equation’s coefficients to the original equation’s coefficients.
Confirm every species coefficient changed by the same factor (true scaling).
Multiply the original ΔH by that factor, keeping units consistent (usually kJ per “reaction as written”).
Keep the sign consistent with the original direction of the reaction unless the direction is also changed.
A fast check: if the balanced equation implies twice as many moles of every reactant/product, the energy change must also be twice as large because twice as much substance is undergoing the same chemical change.
FAQ
If the reported $\Delta H$ is already normalised (e.g., “per mol of fuel” in words), you must match that basis before scaling.
Not in standard chemical equation manipulation. A negative factor would also reverse direction; treat reversal and scaling as separate steps.
That format encodes a chosen basis (often “per mole of reaction” or per mole of a named species), reducing ambiguity when scaling.
Pick one species and divide its new coefficient by its old coefficient; if all species give the same ratio, that ratio is $n$.
Units stay consistent (kJ for the equation as written), but significant figures should follow the least precise given value after multiplication.
Practice Questions
(2 marks) A reaction has as written. The equation is multiplied by . State the new .
Correct scaling factor applied: multiply by (1)
(accept -41.6 to -41.7 kJ) with negative sign retained (1)
(5 marks) The thermochemical equation below has as written. A student writes a new equation in which all coefficients are multiplied by 2.5. (a) State the scaling factor and the new value of . (3 marks) (b) Explain briefly why must be scaled when the coefficients are scaled.
(a)
Identifies (1)
Applies scaling: (1)
with correct sign (1)
(b)
States that enthalpy change depends on amount/reacting moles (enthalpy is extensive) (1)
Links coefficients to reaction extent (more/fewer moles reacting means proportionally more/fewer energy transferred) (1)

