AP Syllabus focus: ‘When all stoichiometric coefficients are multiplied by a factor c, the new equilibrium constant is the original K raised to the power c.’
Scaling a chemical equation changes the equilibrium constant in a predictable way. This page explains how multiplying every stoichiometric coefficient by the same factor affects the mathematical form and numerical value of K.
Core idea: scaling the balanced equation scales the exponent pattern
What “scaling a reaction” means
A balanced reaction can be rewritten by multiplying every stoichiometric coefficient by the same number (the scaling factor). The chemical identity of species is unchanged; only the coefficient set is rescaled.
Original: coefficients are
Scaled: coefficients become
The scaling factor must apply to all coefficients to preserve stoichiometric ratios.
Why K changes when coefficients change
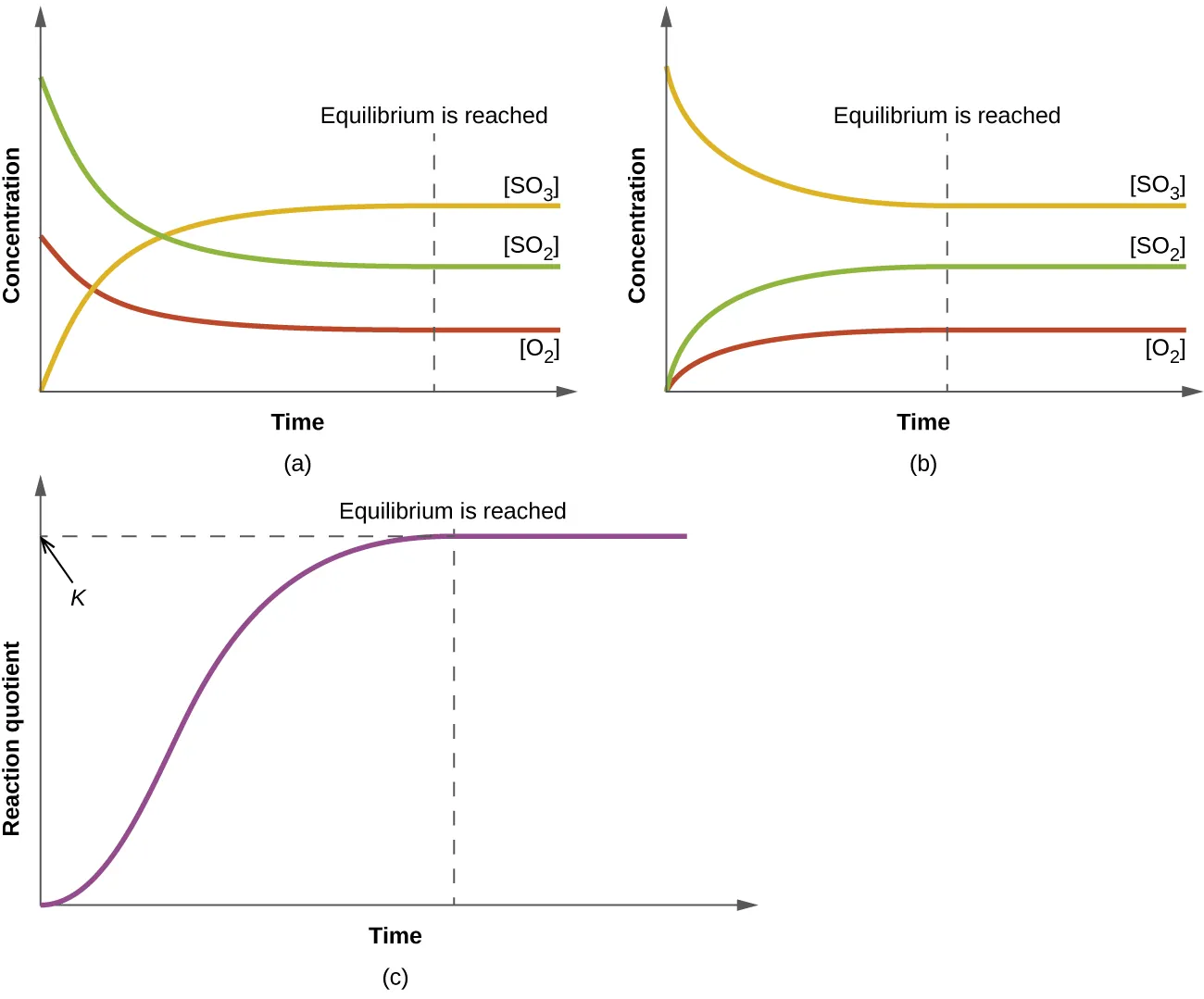
OpenStax Figure 13.6 shows concentration–time curves (and a reaction-quotient curve) leveling off when equilibrium is reached. This supports the conceptual point that equilibrium is a fixed state at a given temperature, even though the algebraic form of depends on how the reaction is written. The vertical marker makes it easy to connect “equilibrium” with “no net change in concentrations.” Source
The equilibrium-constant expression reflects stoichiometry through exponents: each species’ concentration/partial pressure is raised to the power of its coefficient in the balanced equation.
When coefficients are multiplied by a factor, all exponents in the K expression are multiplied by that same factor, which raises the overall value of K to a power.
Definitions you must know
Equilibrium constant (K): A constant (at a fixed temperature) that equals the value of the reaction quotient when the system is at equilibrium, written using equilibrium amounts with exponents from the balanced equation.
A key implication is that K is tied to the way the reaction is written (its stoichiometric coefficients), not just to the identities of reactants and products.
Scaling factor (c): The number by which every stoichiometric coefficient in a balanced chemical equation is multiplied to produce an equivalent, rescaled equation.
The scaling rule for equilibrium constants
The AP-required relationship is the direct mathematical consequence of exponent scaling in the law of mass action.
= equilibrium constant for the scaled equation (unitless)
= equilibrium constant for the original equation (unitless)
= scaling factor applied to all stoichiometric coefficients (dimensionless)
This rule applies whether K is written in concentration form or partial-pressure form, as long as you are consistent with the reaction as written.
Interpreting the rule without doing calculations
Common scaling cases you should recognise
If , then the new K is the square of the original K.
If , then the new K is the cube of the original K.
If (for example, dividing all coefficients by 2), then the new K is a root of the original K (a power less than 1).
What changes and what does not
Changes: the numerical value of K for the rescaled equation.
Does not change: the actual equilibrium state of a given physical system at a fixed temperature (equilibrium composition is the same state), because you have not changed the chemistry—only how the balanced equation is written.
Still temperature-dependent: scaling does not remove the fact that K depends on temperature; scaling is a separate, purely algebraic transformation.
Typical pitfalls
Forgetting that all coefficients must be scaled by the same factor; scaling only one coefficient produces a different reaction.
Mixing equilibrium constants from differently scaled equations; K values can only be compared or combined when the underlying reaction equations are written compatibly.
Assuming scaling changes the direction the reaction “favours.” Scaling changes the reported K value, but it does not create a new chemical equilibrium system—just a new way to express the same stoichiometric relationship.
FAQ
Yes, if the entire equation is divided by a number (e.g., $c=\tfrac{1}{2}$), then $K_{\text{new}} = K^{1/2}$. This corresponds to taking a root of the original K.
In rigorous terms, K is defined using activities (relative to a standard state), making it dimensionless. Concentration-based forms are approximations that preserve the same scaling rule.
No. Changing only one coefficient changes the reaction stoichiometry, so it is no longer a simple scaling transformation and the power rule does not apply.
Yes. Even though pure solids and liquids are omitted from K expressions, any scaled coefficients for gaseous/aqueous species still scale the exponents, so $K_{\text{new}}=K^c$ remains valid.
Adopt a consistent convention such as writing K alongside the exact balanced equation used, or using subscripts (e.g., $K$, $K_{\text{new}}$) that explicitly track the scaling factor.
Practice Questions
(2 marks) For a reaction with equilibrium constant , the balanced equation is multiplied by 2 to give a new equation. State the equilibrium constant for the new equation in terms of .
States (1)
Correctly links the exponent 2 to doubling all stoichiometric coefficients / scaling factor (1)
(5 marks) A reaction has equilibrium constant . The equation is rescaled by a factor . (a) Write an expression relating to and . (2 marks) (b) Explain, in terms of exponents in the equilibrium-constant expression, why this relationship holds. (3 marks)
Writes (2) (allow 1 mark if exponent rule is stated in words but symbols are unclear) (b)
States that stoichiometric coefficients become exponents in the K expression (1)
Explains scaling by multiplies each exponent by (1)
Concludes that multiplying all exponents by raises the entire expression value to the power , giving (1)

