AP Syllabus focus: ‘When reactions are added to form an overall reaction, the equilibrium constant for the overall process is the product of the individual equilibrium constants.’
Adding equilibria is a powerful bookkeeping tool: if several reversible steps sum to a net chemical equation, their equilibrium constants combine predictably. This lets you relate an overall equilibrium position to equilibria of component reactions.
Core idea: summing reactions combines their K values
When two or more chemical equations are added to obtain a single overall reaction, the corresponding equilibrium constants multiply. Conceptually, each step establishes a reactant–product ratio; performing steps in sequence compounds those ratios.
When you are allowed to multiply K values
The component reactions must be addable to give a meaningful net equation (species may cancel if they appear on both sides).
Each must correspond to the reaction exactly as written in the set you are adding.
The overall reaction is interpreted as the combined process of the individual equilibria.
Mathematical statement
= equilibrium constant for the overall (summed) reaction, unitless
= equilibrium constant for reaction step (as written), unitless
This rule is consistent with the law of mass action: multiplying equilibrium-constant expressions corresponds to adding the stoichiometric equations, because the exponents in track stoichiometric coefficients.
Why multiplication makes chemical sense
Cancellation of intermediates
Often, a species is produced in one step and consumed in a later step.
When you add the equations, that species cancels from the net reaction, and it also cancels algebraically when the expressions are multiplied. The overall therefore depends only on species present in the final net equation.
Connection to reaction “strength”
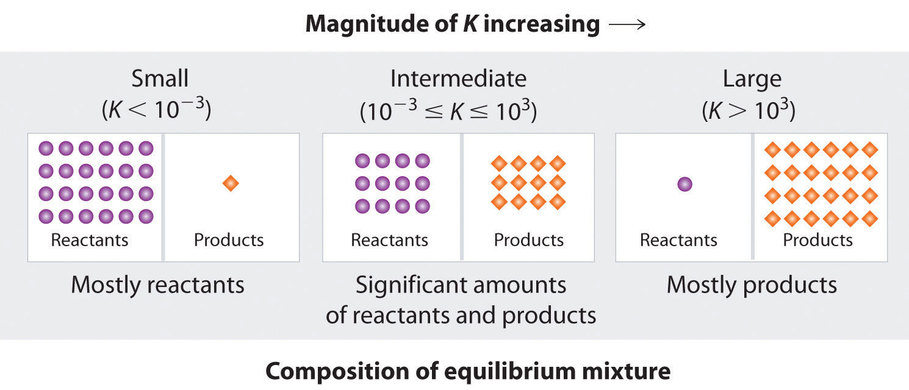
This diagram summarizes how the equilibrium composition shifts as the magnitude of changes. Large corresponds to product-dominant mixtures at equilibrium, small corresponds to reactant-dominant mixtures, and intermediate gives appreciable amounts of both. Source
If each step has (product-favoured), the product tends to be even larger, indicating an even more product-favoured overall reaction.
If one step has a very small , it can dominate the product and make the overall reaction reactant-favoured, even if other steps are product-favoured.
Log form (useful for reasoning)
Because logarithms turn products into sums, chemists often reason about combined equilibria with logs (without needing any calculation details):
This additive property helps compare contributions of individual steps to the overall equilibrium position.
Practical workflow (no arithmetic required)
How to apply the rule reliably
Write each reaction step with its given .
Add the reactions to obtain the overall reaction (cancel species that appear on both sides).
Multiply the listed values to obtain for the summed equation.
Common pitfalls to avoid
Multiplying values for reactions that do not sum to the stated overall equation.
Forgetting that the provided must match the reaction as written (direction and coefficients matter, even though those adjustment rules are handled separately).
Treating like it adds directly; it is values that multiply, not add.
What this rule enables in equilibrium reasoning
Building an overall equilibrium constant for a net transformation from chemically meaningful steps (e.g., coupling equilibria that share intermediates).
Checking consistency: if an overall reaction can be obtained by two different sets of steps, both routes should yield the same when the equations truly represent the same net process.
FAQ
Because each $K$ is built from the law of mass action.
When reactions are added, stoichiometric coefficients add; when the corresponding $K$ expressions are multiplied, exponents add, matching the summed stoichiometry.
Yes.
Even without cancellation, adding equations still defines a valid overall reaction, and the overall equilibrium constant is still $K_{\text{overall}}=\prod K_i$ for the steps as written.
Confirm both routes sum to exactly the same net equation (same species, phases, and stoichiometric coefficients).
If they do, both products of $K$ values should match; disagreement signals that at least one step equation (or its stated $K$) does not correspond to the written form.
The small-$K$ step can dominate the product.
On a log scale, $\log K_{\text{overall}}=\sum \log K_i$, so a strongly negative $\log K_i$ can outweigh several positive contributions, making the overall reaction reactant-favoured.
Yes.
Thermodynamically, $\Delta G^\circ = -RT\ln K$. For summed reactions, standard Gibbs energies add, so $\ln K$ values add, which implies the $K$ values multiply: $K_{\text{overall}}=\prod K_i$.
Practice Questions
(2 marks) Two equilibria can be added to form a net reaction: Reaction 1 has and Reaction 2 has . State the expression for the equilibrium constant of the overall reaction, , in terms of and .
States that values multiply when reactions are added (1)
Gives (1)
(5 marks) Consider the equilibria: (1) with (2) with (3) with They are added to obtain an overall reaction. (a) Write the overall reaction after adding (1), (2), and (3). (2 marks) (b) Write an expression for in terms of , , and . (1 mark) (c) Explain briefly why species does not appear in the overall equilibrium constant expression. (2 marks)
Correctly cancels (produced in (1), consumed in (2)) (1)
Overall reaction: (1) (b)
(1) (c)
States that is an intermediate that cancels from the net equation (1)
Links cancellation to multiplication of expressions: appears in numerator and denominator and cancels algebraically (1)

