AP Syllabus focus: ‘Hydronium and hydroxide concentrations are commonly expressed as pH and pOH using pH = −log[H3O+] and pOH = −log[OH−].’
pH and pOH provide compact, highly interpretable ways to report very small ion concentrations in aqueous solutions. Because they use logarithms, they also connect directly to how acidity changes across orders of magnitude.
Why pH and pOH are used
In water, the concentration of hydronium ions () and hydroxide ions () can vary across many powers of ten. Writing these values in scientific notation is accurate but can be cumbersome. The p-scale turns these concentrations into manageable numbers that still preserve their chemical meaning.
Key ideas:
pH tracks the acidic character of a solution through .
pOH tracks the basic character of a solution through .
Because the scale is logarithmic, equal changes in pH or pOH represent multiplicative changes in concentration.
pH as a measure of hydronium concentration
pH: The negative base-10 logarithm of the hydronium ion concentration, used to express on a compact scale.
Chemically, lower pH corresponds to higher , and higher pH corresponds to lower . The “p” operation is a way of saying “take a negative log,” which flips the direction so that more acidic solutions (more hydronium) have smaller pH values.
= measure of hydronium level (unitless)
= hydronium ion concentration, in (M)
= measure of hydroxide level (unitless)
= hydroxide ion concentration, in (M)
The log is base 10, so each 1-unit decrease in pH corresponds to a tenfold increase in , and each 1-unit increase corresponds to a tenfold decrease.
pOH as a measure of hydroxide concentration
pOH: The negative base-10 logarithm of the hydroxide ion concentration, used to express on a compact scale.
As increases, pOH decreases.
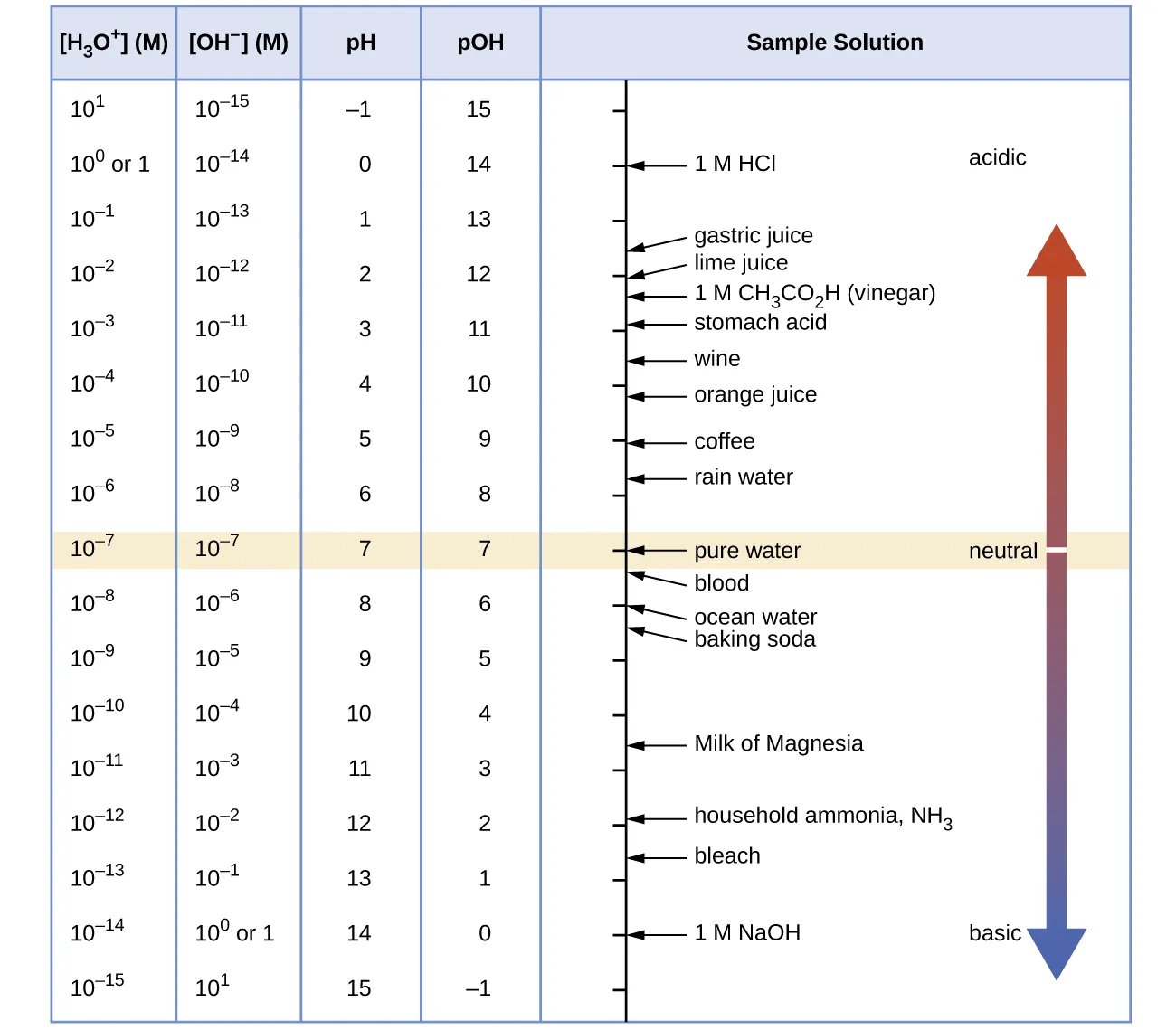
This chart shows the linked scales for , , pH, and pOH, emphasizing their inverse relationship in aqueous solution. It helps you read pH and pOH as logarithmic labels for concentration, and it anchors neutral conditions at pH 7 / pOH 7 (at 25 °C). Source
This makes pOH a convenient “mirror” scale for basicity that is particularly useful when hydroxide concentration is easier to determine directly.
Interpreting p-values (log-scale meaning)
Because pH and pOH are logarithmic, comparisons should be made in factors, not simple differences:
A difference of 2 pH units means a -fold change in .
A difference of 0.30 pH units is close to a factor of 2 change (since ).
The negative sign means larger concentrations map to smaller p-values.
Converting back to concentration (undoing the log)
To recover concentration from a p-value, use the inverse operation of the logarithm (base 10):
From pH to hydronium:
From pOH to hydroxide:
In AP Chemistry contexts, pH and pOH are treated as being based on molar concentrations, and reported pH/pOH values are typically written with attention to appropriate significant figures (reflecting the precision of the concentration data).
FAQ
Most $[H_3O^+]$ values are less than 1 M, so $\log([H_3O^+])$ is negative.
The negative sign makes pH a convenient positive number for typical aqueous solutions.
Yes. Very concentrated strong acids can produce negative pH values (because $[H_3O^+] > 1$ M).
Very concentrated strong bases can give pH values above 14.
The number of decimal places in pH/pOH is tied to the significant figures in the ion concentration.
For example, 2 significant figures in $[H_3O^+]$ typically imply 2 decimal places in pH.
pH meters respond to ion activity rather than idealised concentration.
Small differences can also arise from calibration, temperature, and electrode condition.
In water, free $H^+$ does not exist independently for long; it is associated with water molecules.
Chemistry problems often use $[H^+]$ as shorthand for $[H_3O^+]$, but the underlying meaning is hydronium concentration.
Practice Questions
Q1 (3 marks) A solution has . Calculate the pH.
Uses (1)
Correct substitution with (1)
Correct pH = 3.60 (allow 3.60 or 3.602) (1)
Q2 (5 marks) A solution has pOH = 9.25. (a) Calculate in . (2) (b) Another solution has that is 100 times larger. State the new pOH. (2) (c) State, with a reason, whether the second solution is more or less basic than the first. (1)
(a) Uses (1)
(a) Correct (allow 5.62) (1)
(b) Recognises factor 100 means pOH decreases by 2.00 units (1)
(b) New pOH = 7.25 (1)
(c) More basic because larger (or lower pOH indicates higher hydroxide) (1)

