AP Syllabus focus: ‘Water autoionizes and establishes an equilibrium with Kw = [H3O+][OH−] = 1.0 × 10−14 at 25 °C.’
Water is not chemically inert: a tiny fraction of molecules continuously transfer protons to each other.
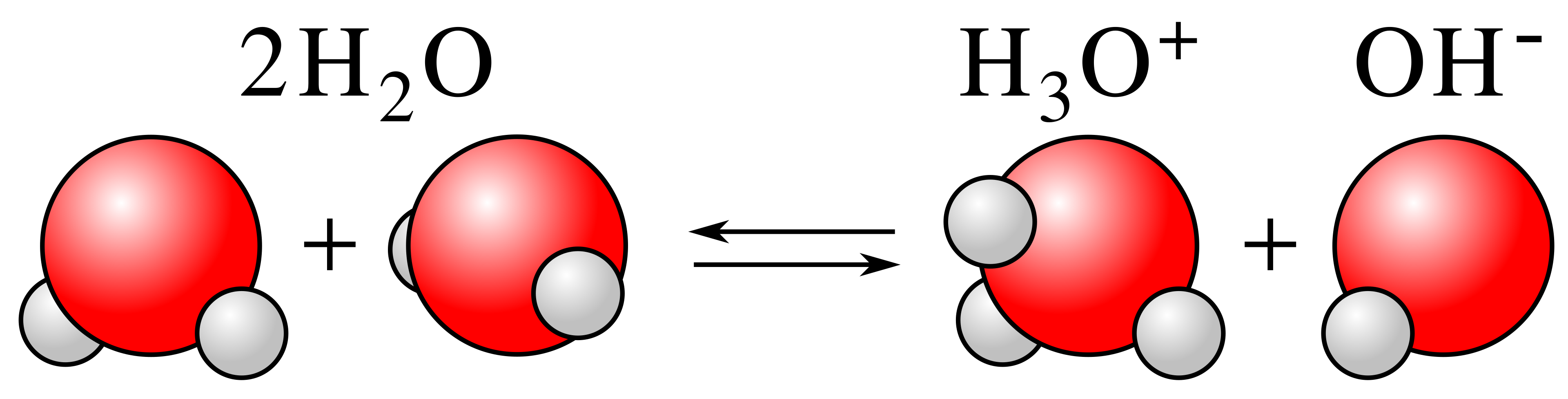
Autoprotolysis (self-ionization) of water shown as an equilibrium: two molecules exchange a proton to form and . This visual reinforces the Brønsted–Lowry idea that water can act as both an acid and a base in the same process. Source
This autoionization creates hydronium and hydroxide and defines a fundamental equilibrium constant, Kw.
The autoionization equilibrium in water
What “autoionization” means
Water molecules can act as both proton donors and proton acceptors. When one water molecule donates a proton to another, ions form; because the reactant and solvent are the same substance, this is called autoionization (also called self-ionization).
Autoionization (of water): A proton-transfer reaction in which two water molecules react to form hydronium (H3O+) and hydroxide (OH−), establishing an equilibrium.
This process is always occurring in liquid water, but it lies far to the reactant side, so only very small amounts of ions are present at any moment.
Chemical equation and equilibrium expression
= ion-product constant for water (unitless in the thermodynamic sense; treated as unitless in AP-style calculations)
= hydronium ion concentration in mol L (M)
= hydroxide ion concentration in mol L (M)
In words, Kw is the product of the equilibrium concentrations of hydronium and hydroxide ions. The liquid water itself is not included in the expression because it is a pure liquid with effectively constant concentration.
Kw: the ion-product constant of water
Definition and numerical value at 25 °C
Kw (ion-product constant of water): The equilibrium constant for water’s autoionization, equal to the product of hydronium and hydroxide ion concentrations, .
At 25 °C, the syllabus-specified value is Kw = 1.0 × 10−14.
This single constant is a cornerstone for reasoning about aqueous acid–base systems because any change in one ion concentration is coupled to the other through the equilibrium.
Interpreting Kw physically
A small Kw indicates that the equilibrium strongly favours molecular water over ions.
Even in very pure water, ions exist due to this equilibrium; they are not “impurities,” but products of water’s intrinsic chemistry.
Because Kw is an equilibrium constant, it reflects a dynamic equilibrium:
forward process: formation of H3O+ and OH−
reverse process: recombination to form H2O molecules
How Kw constrains aqueous solutions
In any aqueous solution at 25 °C (whether acidic, basic, or neither), the ion concentrations must satisfy:
the product [H3O+][OH−] equals 1.0 × 10−14
if a process increases [H3O+], equilibrium considerations imply [OH−] must decrease so the product stays constant (and vice versa)
This constraint is independent of why the ions are present (for example, whether H3O+ comes from dissolving an acid or from water itself); the equilibrium condition still applies at 25 °C.
Using Kw correctly in AP Chemistry contexts
Key ideas that prevent common errors
Kw is an equilibrium constant, not a concentration. It is the product of two concentrations at equilibrium.
Do not include [H2O] in the Kw expression. For a pure liquid, its activity is taken as constant and absorbed into the constant.
Square brackets mean concentration (M) of the species in solution; they do not represent moles.
When solving problems, treat H3O+ and H+ as representing the acidic species in water, but keep the Kw expression specifically in terms of [H3O+] and [OH−] as given by the syllabus statement.
Minimal equilibrium picture
Water establishes an equilibrium between:
predominantly H2O molecules
trace H3O+ and OH− ions
The equilibrium position at 25 °C is summarised numerically by Kw = 1.0 × 10−14, which links the two ion concentrations in every aqueous solution at that temperature.
FAQ
It represents the product of ionic species concentrations arising from the solvent’s own equilibrium.
Historically, it emphasises that both ions originate from water itself, even though other solutes can shift their concentrations.
Thermodynamically, $K_w$ is defined using activities, not raw molar concentrations.
In dilute AP-style problems, activities are approximated by concentrations, which is why $K_w = [\mathrm{H_3O^+}][\mathrm{OH^-}]$ is used without activity coefficients.
Direct measurement in pure water is difficult because conductivity is extremely low and contamination matters.
Methods typically involve careful conductivity measurements with ultra-pure water and extrapolation procedures, or electrochemical approaches that account for activity effects.
Forming separated charges in a neutral molecular liquid is energetically unfavourable.
Although water stabilises ions via solvation, the equilibrium still strongly favours neutral H2O, yielding a very small equilibrium constant.
Dissolved carbon dioxide forming acidic species, increasing $[\mathrm{H_3O^+}]$
Trace ions from glassware or salts, affecting conductivity and ionic strength
Temperature fluctuations, which shift the equilibrium constant away from its 25 °C value
Practice Questions
(5 marks) Liquid water at 25 °C undergoes autoionisation.
(a) Write the balanced equation for this equilibrium using hydronium. (1 mark)
(b) Write the equilibrium expression defining . (1 mark)
(c) State the value of at 25 °C. (1 mark)
(d) A sample has at 25 °C. Calculate . (2 marks)
(a) (1 mark)
(b) (1 mark)
(c) at 25 °C (1 mark)
(d) Rearrangement (1 mark)
(d) Correct value (1 mark)
(2 marks) At 25 °C, a student measures in an aqueous solution. Using , determine .
Uses (1 mark)
Correct value (1 mark)

