AP Syllabus focus: ‘In pure neutral water at 25 °C, pH = pOH = 7.0 and pKw = 14.0; because Kw depends on temperature, neutral pH differs from 7.0 at other temperatures.’
Pure water is not chemically “inactive”: it self-ionizes slightly, and the extent of that ionization changes with temperature.
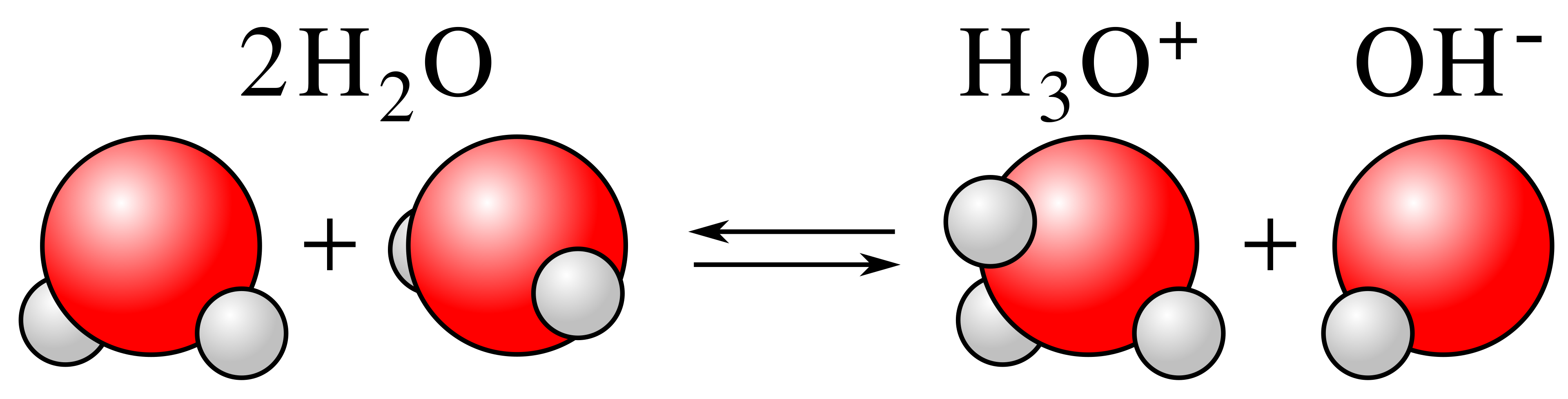
Diagram of water autoprotolysis (autoionization): two water molecules exchange a proton to form hydronium, , and hydroxide, . The figure makes it explicit that both ions are produced simultaneously, so in pure water their concentrations are equal at neutrality. This equilibrium underlies the definition of . Source
This shifts Kw, pKw, and therefore the pH of neutral water away from 7.00 except at 25 °C.
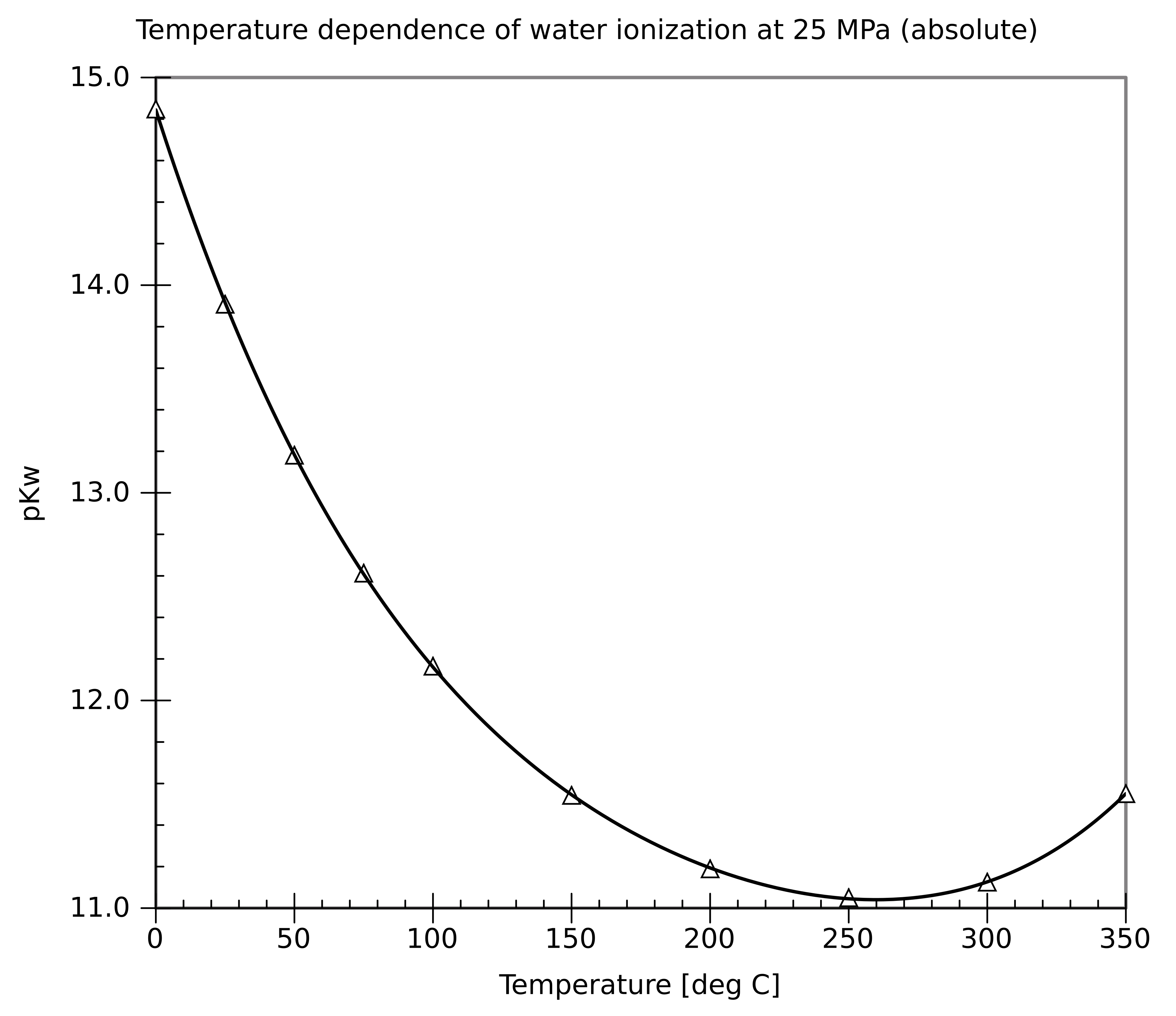
Temperature dependence of water’s ionization constant () shown as a plotted trend versus temperature. As temperature changes, the autoionization equilibrium shifts, so does not remain fixed at . This is why the neutral-point pH is not always 7.00. Source
Neutral water at 25 °C
What “neutral” means (and what it does not mean)
Neutrality is defined by equal hydronium and hydroxide concentrations, not by a fixed pH value.
Neutral solution: a solution in which (so there is no net acidic or basic character).
At 25 °C, that equality happens to correspond to pH = 7.0 and pOH = 7.0, but that numerical value is temperature-specific.
The role of pKw at 25 °C
The equilibrium constant for water’s autoionization is Kw, and its logarithmic form pKw is often used to connect acidity and basicity on the pH scale. At 25 °C, pKw = 14.0, which is consistent with neutral water having equal pH and pOH values.
= negative base-10 logarithm of (unitless)
= water ion-product constant (unitless, defined in terms of concentrations for AP-level work)
Because neutral water has , the neutrality condition links directly to pKw: when the two ion concentrations are equal, their p-notation values are equal as well (pH = pOH). At 25 °C, that shared value is 7.0 because pKw is 14.0.
Why neutral pH changes with temperature
Kw depends on temperature
The syllabus requirement is that you understand this relationship qualitatively: Kw depends on temperature, so the pH of neutral water differs from 7.0 at other temperatures. In other words, “neutral” does not mean “pH 7” unless the temperature is 25 °C.
A temperature change alters the position of the autoionization equilibrium for water. When Kw changes, the product changes, and the “equal-ion” point (neutrality) shifts to a different numerical pH.
pKw is the temperature-sensitive reference point
Because is defined from , it also varies with temperature. That matters because the numerical relationship between pH and pOH is anchored to pKw, not automatically to 14.
= (unitless)
= (unitless)
= temperature-dependent sum of and (unitless)
When temperature changes:
changes
therefore changes
therefore the neutral-point condition pH = pOH = shifts
so neutral pH is not necessarily 7.00
Interpreting “neutral” across temperatures (AP expectation)
For AP Chemistry, focus on these takeaways:
At 25 °C (the standard reference temperature), neutral water has pH = pOH = 7.0 and pKw = 14.0.
At other temperatures, you should not assume pH 7 indicates neutrality.
If you are given a non-25 °C value of Kw or pKw, neutrality is found by enforcing (equivalently, pH = pOH) and using the temperature-appropriate pKw.
FAQ
Water’s autoionisation is endothermic overall.
Increasing temperature favours products, increasing $K_w$; decreasing temperature lowers $K_w$.
No.
“Neutral” only describes $[H_3O^+] = [OH^-]$; it says nothing about toxicity, oxidising power, or other reactivity.
It comes from using concentration-based expressions in practice.
Strictly, equilibrium constants are defined using activities (unitless); concentrations are an approximation.
Electrode response depends on temperature, so meters use temperature compensation.
Without compensation, the displayed pH can drift even if the chemical composition is unchanged.
The definition $pH = -\log[H_3O^+]$ does not change.
What changes is the reference point for neutrality, because $K_w$ (and thus $[H_3O^+]$ in pure water) changes with temperature.
Practice Questions
(2 marks) State the pH of pure neutral water at and state the value of at .
pH (1)
(1)
(5 marks) At , . Determine the pH of neutral water at this temperature. Show your working.
Uses neutrality: (1)
Sets (1)
Finds (1)
Computes (1)
Final pH (\approx 6.77) (allow suitable rounding) (1)

