AP Syllabus focus: ‘Acid–base reactions can be run under controlled conditions in a titration; a titration curve plots pH versus the volume of titrant added to summarize the results.’
Acid–base titrations let you track how solution acidity changes as measured amounts of a reacting solution are added. Titration curves condense those measurements into a clear picture of how pH responds to added volume.
What a titration is (and why it’s “controlled”)
A titration is designed so that the amount of reacting solution added is known very precisely, letting pH changes be linked directly to added volume.
Titration: An experimental method in which a solution of known concentration is added in measured amounts to a reacting solution to monitor a chemical change (here, pH changes from an acid–base reaction).
In acid–base titrations, the key control variables are:
Volume added (delivered accurately from a buret)
Mixing (so measured pH represents the whole solution)
Consistent measurement conditions (same temperature, same pH measurement method)
Essential titration vocabulary and setup
A typical setup includes a buret (for delivery), a flask or beaker (for the reacting mixture), and a way to measure pH (pH probe/meter or indicator, depending on the goal).
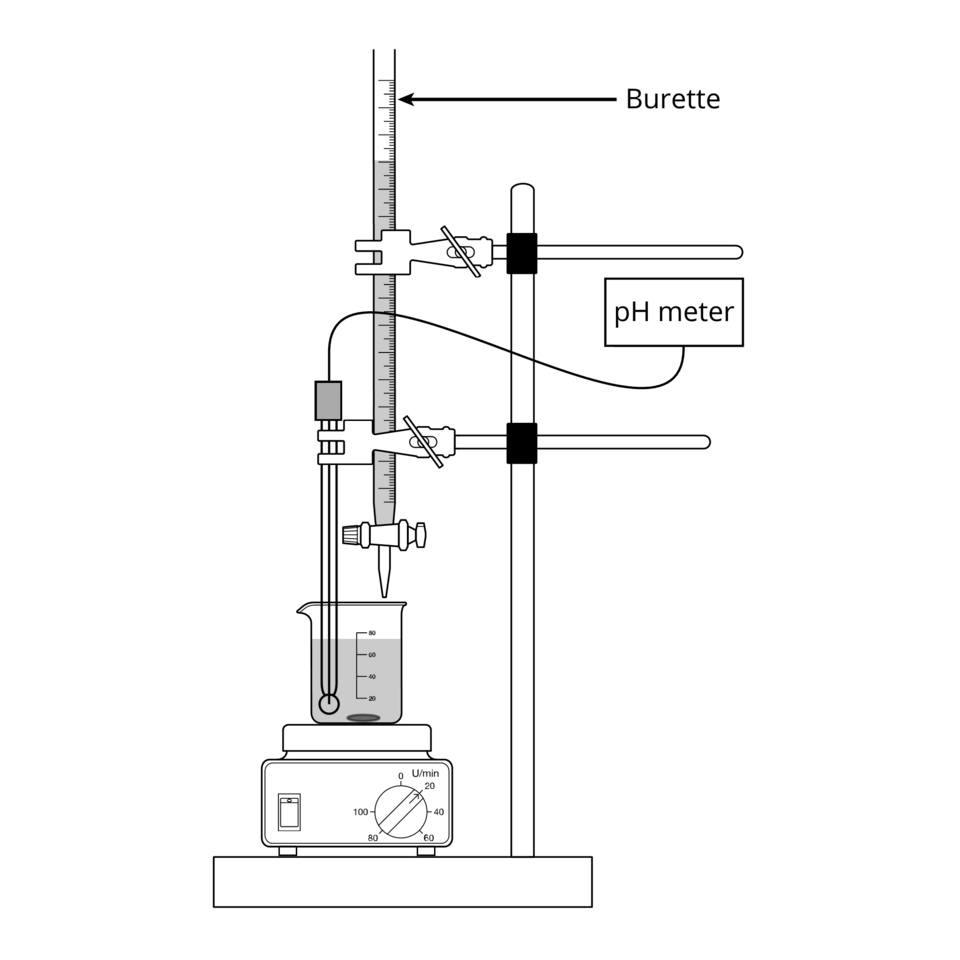
Labeled line-art diagram of a standard titration setup, showing how the buret delivers titrant into the flask while a pH probe monitors pH and a stir plate ensures uniform mixing. This helps connect technique choices (delivery accuracy, mixing, and measurement method) to the quality of a titration curve. Source
Titrant / analyte: The titrant is the solution added from the buret; the analyte is the solution initially in the flask whose pH is being monitored.
Key equipment/choices that affect data quality:
Buret reading: read the bottom of the meniscus at eye level; record to the correct decimal place.
Erlenmeyer flask: allows swirling without splashing, improving mixing.
pH probe: enables continuous/stepwise quantitative pH readings suitable for building a curve.
Stirring: magnetic stirring or consistent swirling reduces local pH gradients near the titrant entry point.
Collecting titration-curve data
To generate a titration curve, you repeatedly add small volumes of titrant and record the pH after the solution becomes uniform.
Good practice for curve-shape resolution:
Record the initial pH before any titrant is added.
Add titrant in larger increments when pH changes slowly.
Switch to smaller increments in regions where pH changes rapidly (to capture the steep part of the curve).
Wait for the pH reading to stabilise after each addition (especially with probes).
Common data-recording expectations:
Consistent significant figures in recorded volumes and pH.
Clear pairing of each pH value with the total volume added (not just the increment).
What a titration curve shows
A titration curve is the graphical summary specified by the syllabus: pH plotted against the volume of titrant added.
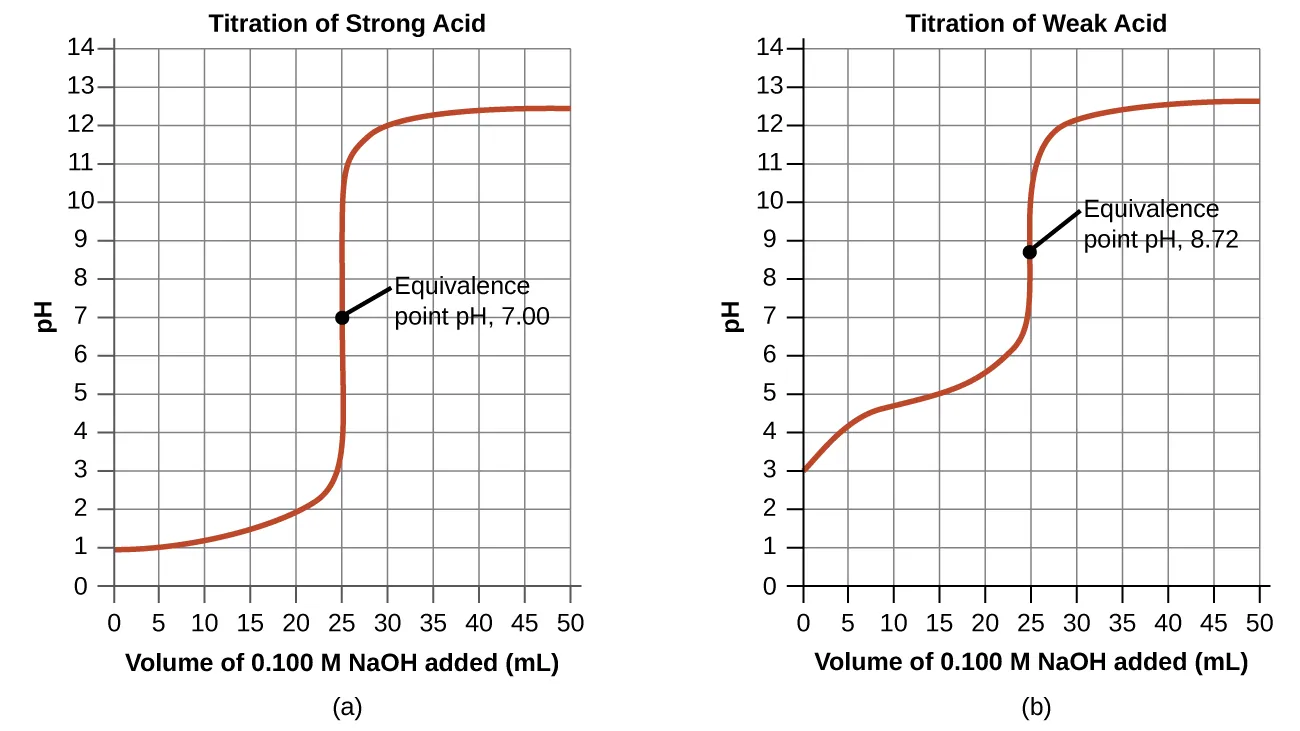
Two titration curves (strong acid–strong base and weak acid–strong base) are plotted as pH versus volume of NaOH added, highlighting the characteristic slow–fast–slow pH change pattern. The figure emphasizes how the equivalence-point pH depends on the acid/base strengths (near 7 for strong/strong, >7 for weak acid/strong base). Source
The curve’s shape reflects how the acid–base reaction progresses as titrant is introduced.
Titration curve: A plot of pH (y-axis) versus volume of titrant added (x-axis) that summarises how pH changes during a titration.
Core features students should be able to describe qualitatively:
Starting region: pH determined primarily by the initial analyte composition.
Gradual-change region(s): pH changes modestly with added titrant.
Rapid-change region: a steep section where small volume additions cause large pH shifts, indicating the reaction is approaching stoichiometric completion.
Ending region: pH is dominated by whichever reagent becomes present in excess after the main neutralisation reaction is effectively complete.
Comparing curves (qualitative, not computational)
Different acid–base pairings produce characteristically different curve shapes, but the unifying idea remains the syllabus statement: controlled additions + pH tracking → a curve that summarises results.
Qualitative comparisons you can make from curves alone:
Overall pH range covered (how acidic/basic the solution becomes)
Steepness of the rapid-change region (how sharply pH responds near stoichiometric completion)
How quickly pH changes early vs late in the titration (sensitivity to added volume)
Common technique issues that distort curves
Errors typically show up as shifted, “noisy,” or flattened curves:
Incomplete mixing → inconsistent pH readings after additions
Misread buret volumes → x-axis errors that shift the curve horizontally
pH probe issues (dirty bulb, poor calibration) → systematic pH offsets or drift
Adding volumes too large in the steep region → loss of curve detail where pH changes fastest
FAQ
Probe response time, incomplete mixing, and recording pH before stabilisation can add scatter.
Rinse with deionised water, blot (don’t wipe), and store as instructed (often in electrode storage solution).
Residual water dilutes the titrant in the buret, shifting the curve horizontally by changing delivered concentration.
Large increments can miss the steep region; small increments improve resolution but increase time and cumulative reading uncertainty.
It increases point density and consistency, capturing rapid pH changes with less timing and transcription error.
Practice Questions
(2 marks) State what is plotted on each axis of a titration curve and what the curve summarises.
pH on the y-axis (1)
Volume of titrant added on the x-axis; summarises how pH changes during the titration (1)
(5 marks) Describe how you would obtain reliable data for an acid–base titration curve using a buret and a pH meter. Include how you would choose addition volumes and ensure readings are valid.
Use buret to add measured volumes of titrant to analyte in a flask/beaker (1)
Measure and record initial pH before additions (1)
Mix thoroughly after each addition (stir/swirl) and wait for pH to stabilise before recording (1)
Use smaller volume increments where pH changes rapidly to capture the steep region (1)
Record pH against total volume added and plot pH vs volume of titrant added (1)

