AP Syllabus focus: ‘At the equivalence point for monoprotic titrations, moles of titrant added equal moles of analyte originally present; this relationship is used to determine analyte concentration.’
A titration uses measured volumes to relate an unknown solution’s amount of substance to a known solution. Mastering equivalence-point stoichiometry lets you convert buret readings into analyte concentration reliably and efficiently.
Core idea: the equivalence point
The equivalence point is the stoichiometric completion point of the reaction between analyte and titrant.
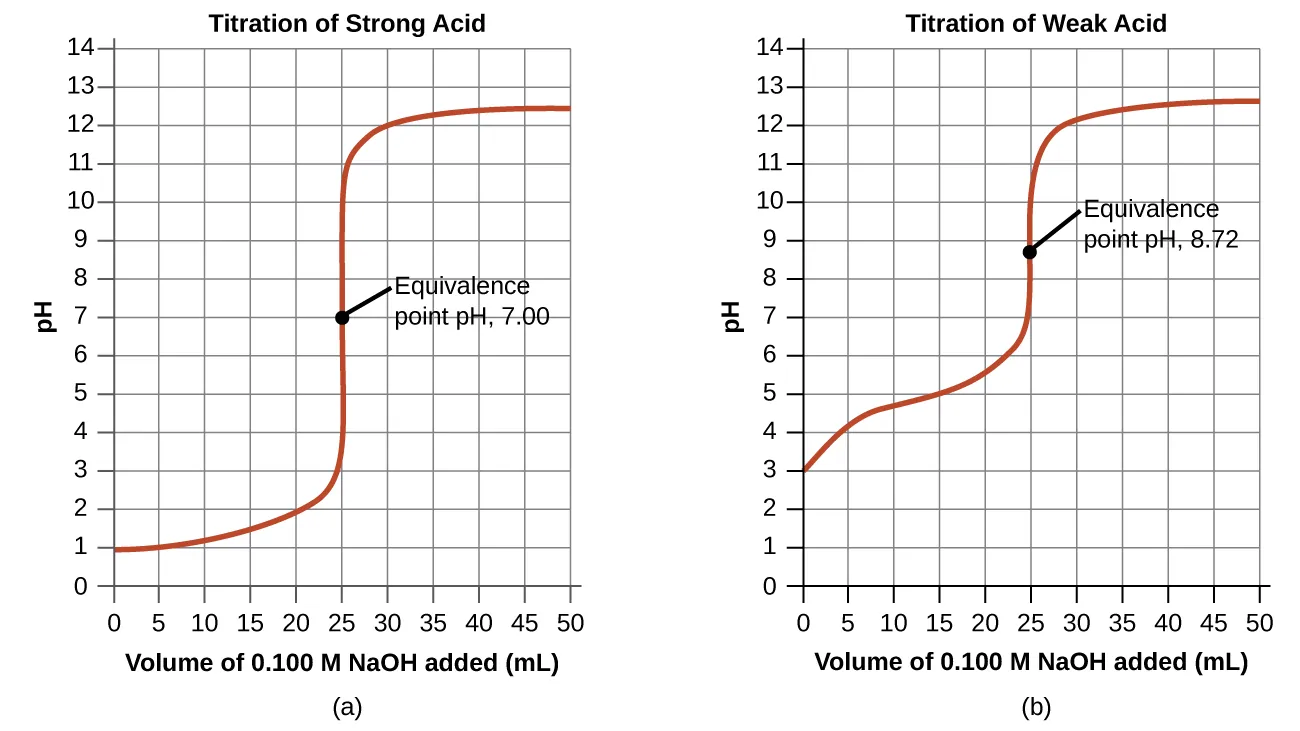
This titration curve plots pH versus volume of titrant added and highlights the characteristic steep region used to locate the equivalence point. The equivalence point corresponds to stoichiometric completion, where the reaction has consumed the limiting reactant and the solution composition changes abruptly. Reading from the curve connects the experimental measurement (volume) to the mole-equality condition used in calculations. Source
For the AP focus here, the key case is monoprotic titrations, where the reaction occurs in a 1:1 mole ratio.
Equivalence point: The point in a titration at which the chemically reacting species have been combined in exact stoichiometric amounts (no limiting reactant remains).
In a monoprotic acid–base titration, one mole of titrant reacts with one mole of analyte, so the equivalence condition becomes a direct equality of moles.
Monoprotic stoichiometry (the AP relationship)
For monoprotic titrations at equivalence:
moles titrant added = moles analyte initially present
This is the relationship used to determine the analyte’s concentration from titration data.
Analyte: The solution of unknown concentration being measured in a titration.
A correct setup always begins by identifying which solution is the analyte (unknown) and which is the titrant (known), then applying the 1:1 equivalence relationship only at the equivalence point.
Turning volumes into moles (and concentration)
Titration data typically provide:

This photograph shows the concave meniscus in a buret at a specific marked volume, illustrating where the correct reading is taken on the scale. Accurate buret readings are essential because the delivered titrant volume (final minus initial) is the experimental input used to compute moles via . In practice, reading the meniscus consistently at eye level helps minimize systematic volume error. Source
Titrant molarity (given)
Volume of titrant at equivalence (from buret readings)
Volume of analyte used (pipet or volumetric measurement)
You convert titrant volume to moles using molarity, then transfer those moles to the analyte using the monoprotic equivalence relationship, and finally compute analyte concentration by dividing by analyte volume.
= amount of substance (moles),
= molarity,
= solution volume used,
Always convert volumes to litres before multiplying by molarity. If volume is recorded in mL, convert using .
Analyte concentration from equivalence
At equivalence in a monoprotic titration:
Find from
Set
Compute
Be explicit about which volume belongs in the final division: it is the original analyte volume placed in the flask, not the total mixed volume after titrant is added.
Practical interpretation of “at equivalence”
“Equivalence point” refers to the chemical condition (stoichiometric completion), not merely a color change. In practice, you approximate it with an experimentally observed point (often a sharp signal change), then treat that volume as for the mole equality.
Common pitfalls to avoid
Using the initial buret reading as the titrant volume; the titrant volume delivered is final − initial.
Forgetting that the AP relationship (moles titrant = moles analyte) is specified for monoprotic titrations at equivalence, not at arbitrary points.
Dividing by the titrant volume instead of the analyte volume when finding analyte molarity.
Skipping unit checks; molarity must end in .
FAQ
Look for a single transferable $\text{H}^+$ (acids like HCl) or a single $\text{OH}^-$/single-proton-accepting site in the reaction as written.
Watch for species like $\text{H}_2\text{SO}_4$ that can donate more than one proton.
Concentration is defined for the original sample: $M_\text{analyte}=n_\text{analyte}/V_\text{analyte,initial}$.
Using total mixed volume would give the concentration after dilution, not the analyte’s original molarity.
Ensure delivered buret volume is in $\text{L}$ before using $n=MV$.
Then confirm the final division is $\text{mol}/\text{L}$, not $\text{mol}/\text{mL}$.
Limit the final molarity by the least precise measured quantity (often buret volume, typically $\pm 0.01\ \text{mL}$, and given molarity).
Carry extra digits during intermediate steps, round only at the end.
Clean glassware and proper rinsing (buret with titrant, pipet with analyte) reduce systematic dilution.
Reading the meniscus at eye level and consistent dropwise delivery near the endpoint improve volume precision.
Practice Questions
(2 marks) In a monoprotic titration, of an acid analyte is titrated to equivalence with NaOH. The volume of NaOH delivered is . Determine the analyte molarity.
1 mark: Calculate using .
1 mark: Use and .
(5 marks) of a monoprotic base analyte is titrated with monoprotic acid. Buret readings: initial , final . Calculate (i) the volume delivered, (ii) moles of titrant at equivalence, and (iii) the analyte concentration.
1 mark: (or ).
1 mark: Convert to litres correctly.
1 mark: .
1 mark: Monoprotic equivalence: .
1 mark: with correct units.

