AP Syllabus focus: ‘Entropy increases when matter becomes more dispersed; particles spread out and have more possible arrangements.’
Entropy is a way to describe how widely matter and energy are spread out in a system. In AP Chemistry, you mainly use entropy to make qualitative predictions from particle-level reasoning.
What entropy measures: dispersal and “disorder”
Entropy as dispersal (the AP Chemistry emphasis)
Entropy (): a measure of how dispersed matter and energy are within a system; greater dispersal corresponds to more possible particle arrangements and greater entropy.
The syllabus statement connects entropy to two linked ideas:
Dispersal of matter: particles are spread through a larger region of space (more “places” to be).
More possible arrangements: there are more distinct ways to distribute particles (and energy) while still describing the same overall conditions.
This is why, at the particulate level, entropy tends to increase when a system changes from “confined and structured” to “spread out and flexible.”
Microstates: the “possible arrangements” idea
Microstate: one specific, detailed arrangement of particles (and their energy distribution) consistent with the same overall, observable state of the system.
A single macroscopic situation (same amount of substance, same container, same temperature) can correspond to many microstates. Entropy increases when the count of accessible microstates increases, because the system has more ways to “be arranged” without changing the macroscopic description.
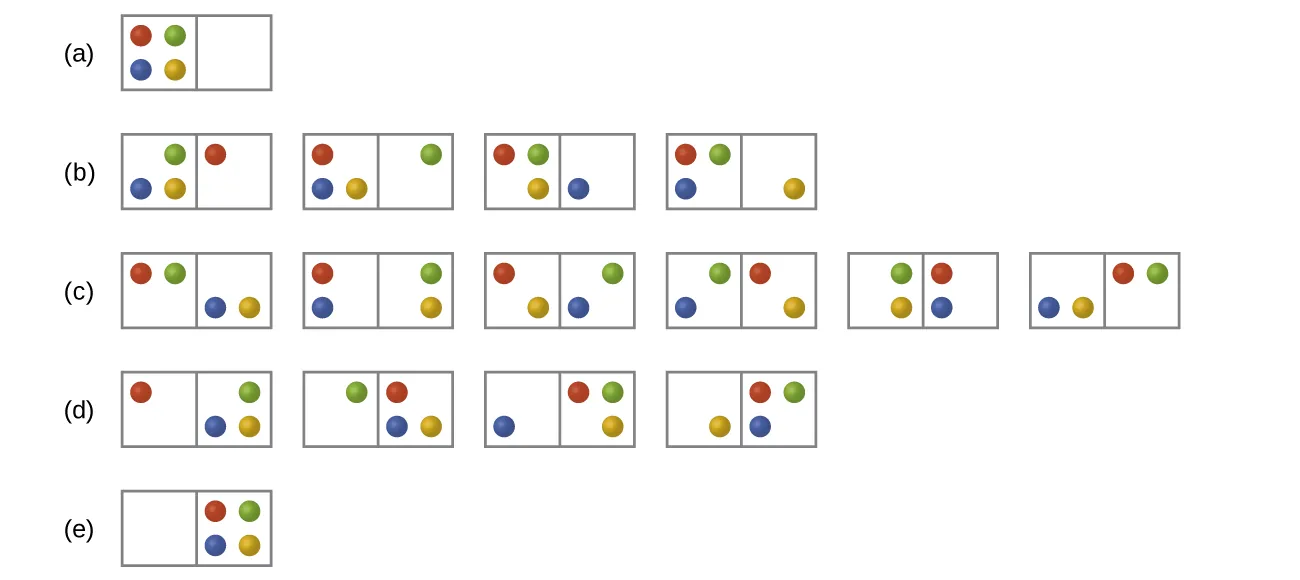
Enumerated microstates for distributing four particles between two “boxes,” grouped into macrostates (distributions) by how many particles occupy each side. The diagram makes it concrete that the most evenly dispersed distribution corresponds to the largest multiplicity , which is why it is the most probable macrostate and is associated with higher entropy. Source
To connect “arrangements” to a compact scientific statement:
= entropy (J·K)
= Boltzmann constant (J·K)
= number of accessible microstates (dimensionless)
Even if you do not calculate with this relationship in AP Chemistry, it supports the key interpretation: larger (more arrangements) means larger .

Photograph of Ludwig Boltzmann’s gravestone featuring the statistical definition of entropy, . This connects the symbolism in the equation to its historical origin and reinforces that entropy scales with the logarithm of the number of accessible microstates. Source
“Disorder” as a helpful but limited metaphor
Chemistry courses often describe entropy as disorder, but the more reliable AP-level wording is dispersal and number of arrangements.
“Disorder” works best when it clearly tracks how constrained the particles are.
“Disorder” can be misleading if used vaguely (for example, some highly “mixed” systems can still be locally structured, yet have high entropy because many microstates are available overall).
A good habit is to justify entropy changes using what particles can do (where they can move and how many ways they can be arranged), not just the word “disorder.”
Particle-level reasoning that raises entropy
When matter becomes more dispersed
Entropy tends to increase when particles occupy a larger volume or have more positional freedom because:
there are more locations available
there are more ways to arrange particles among those locations
Qualitative indicators that matter is more dispersed include:
particles are farther apart on average
particles can move independently rather than being locked into fixed positions
mixing produces many ways to distribute different particle types throughout the available space
Why gases usually have higher entropy than liquids and solids
Without focusing on phase-change rules, you can still reason from dispersal:
In a solid, particles mainly vibrate around fixed positions (few positional arrangements).
In a liquid, particles can move past one another but remain close together (more arrangements).
In a gas, particles move freely through the container (many more arrangements; matter is highly dispersed).
The key AP idea is that spreading out (especially into three-dimensional space with lots of empty volume) typically corresponds to many additional microstates.
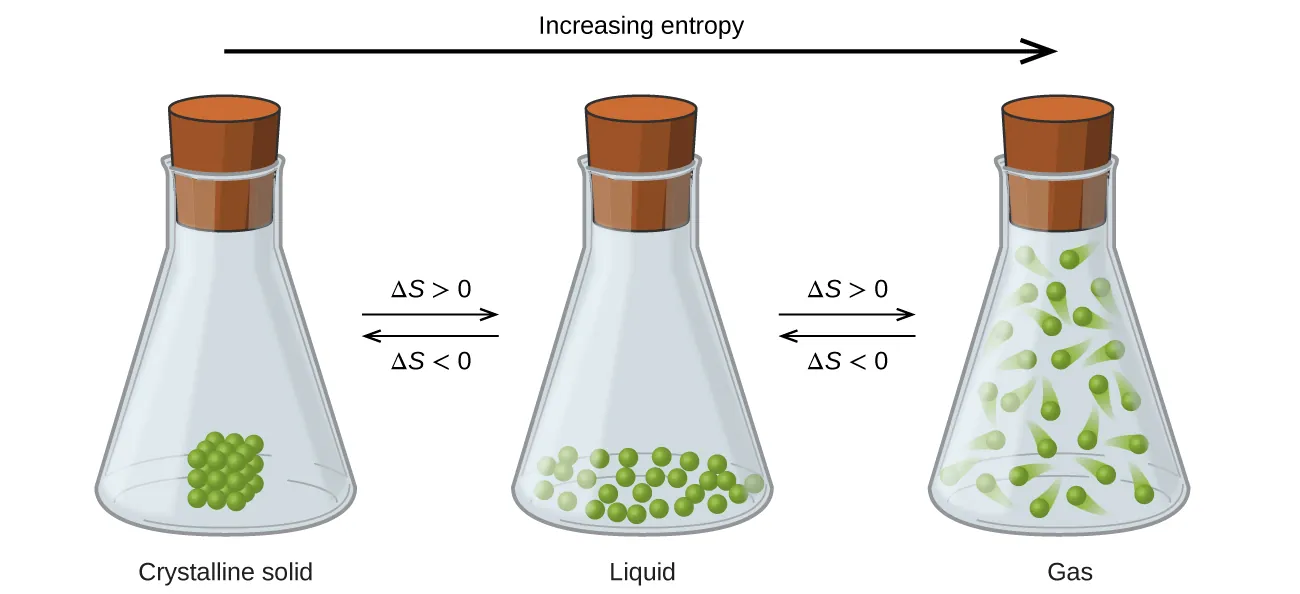
Particulate diagrams of the same substance as a crystalline solid, liquid, and gas, with arrows indicating the direction of increasing entropy. The figure emphasizes how greater freedom of motion and occupation of a larger volume increases the number of accessible arrangements, so . Source
Using diagrams and language correctly
What to look for in particulate diagrams
When comparing two pictures of the same amount of substance, higher entropy is associated with:
particles occupying a larger region of the container
particles being less constrained (fewer fixed lattice-like positions)
more ways to place particles without changing the overall appearance (more “arrangements” consistent with the same macrostate)
Common pitfalls in wording
Saying “entropy increases because it’s more random” is weak unless you tie it to more arrangements.
Do not confuse entropy with enthalpy: entropy is not “how much energy” the system has, but how dispersed energy and matter are across possible arrangements.
Avoid treating entropy as a moral direction (“nature wants disorder”); instead, describe the probabilistic availability of many microstates.
FAQ
Both matter and energy dispersal contribute to entropy.
In AP Chemistry this subtopic stresses matter dispersal (particles spreading out), but the “possible arrangements” idea also includes how energy can be distributed among particles.
A different microstate is a different detailed assignment of particle positions (and, more broadly, energy distribution) that still matches the same overall conditions.
Two microstates can look identical macroscopically but differ microscopically.
More available volume means more possible locations for each particle.
That increases the number of ways to distribute particles in space, so the number of accessible microstates increases.
Yes. Visual “order” is not a reliable metric.
If the “ordered-looking” state allows more accessible microstates overall (for instance, many equivalent arrangements consistent with the same macroscopic pattern), entropy can be higher.
Concentration/density describe how much matter is in a given volume.
Entropy describes how many microscopic arrangements are accessible; density can influence entropy, but entropy is not itself a measure of “how concentrated” something is.
Practice Questions
(2 marks) Explain, in terms of particle arrangements, why entropy increases when matter becomes more dispersed.
Mentions particles spread out/occupy larger region or are less constrained (1)
Links dispersal to more possible arrangements/microstates (1)
(5 marks) A student claims: “Entropy just means disorder, so it always increases when a system looks messier.” Critique this claim using the idea that “particles spread out and have more possible arrangements.” Your answer should refer to microstates and dispersal of matter.
States entropy relates to dispersal and number of possible arrangements rather than appearance alone (1)
Defines or correctly uses the idea of microstates as specific arrangements consistent with the same macrostate (1)
Explains that greater dispersal typically increases accessible microstates (1)
Notes “disorder” is an imperfect metaphor and must be justified by particle freedom/constraints (1)
Uses clear particle-level reasoning (e.g., more locations/ways to arrange particles) rather than vague statements (1)

