AP Syllabus focus: ‘Entropy rises for solid→liquid→gas, for gases expanding at constant temperature, and when gas-product moles exceed gas-reactant moles.’
Entropy trends let you predict the sign and relative size of without data tables. The most reliable cues are phase (solid/liquid/gas), gas volume changes, and changes in moles of gaseous particles.
What you are predicting: the sign of
Entropy is tied to how many ways matter and energy can be dispersed among particles. In AP Chemistry, you typically make qualitative predictions (increase, decrease, ~no change) using particle-level reasoning.
Entropy change (): the change in a system’s entropy during a process; positive when matter/energy becomes more dispersed, negative when it becomes more concentrated.
A positive is generally associated with greater freedom of motion and more spatial spreading of particles.
= entropy change of the system,
= heat absorbed reversibly by the system, (or )
= absolute temperature,
This relationship supports the idea that adding heat at a given temperature tends to increase dispersal, but AP questions here focus mainly on phase/volume/moles trends.
Predicting for phase changes
Phase changes are high-yield because particle freedom changes dramatically.
General phase trend (most important rule)
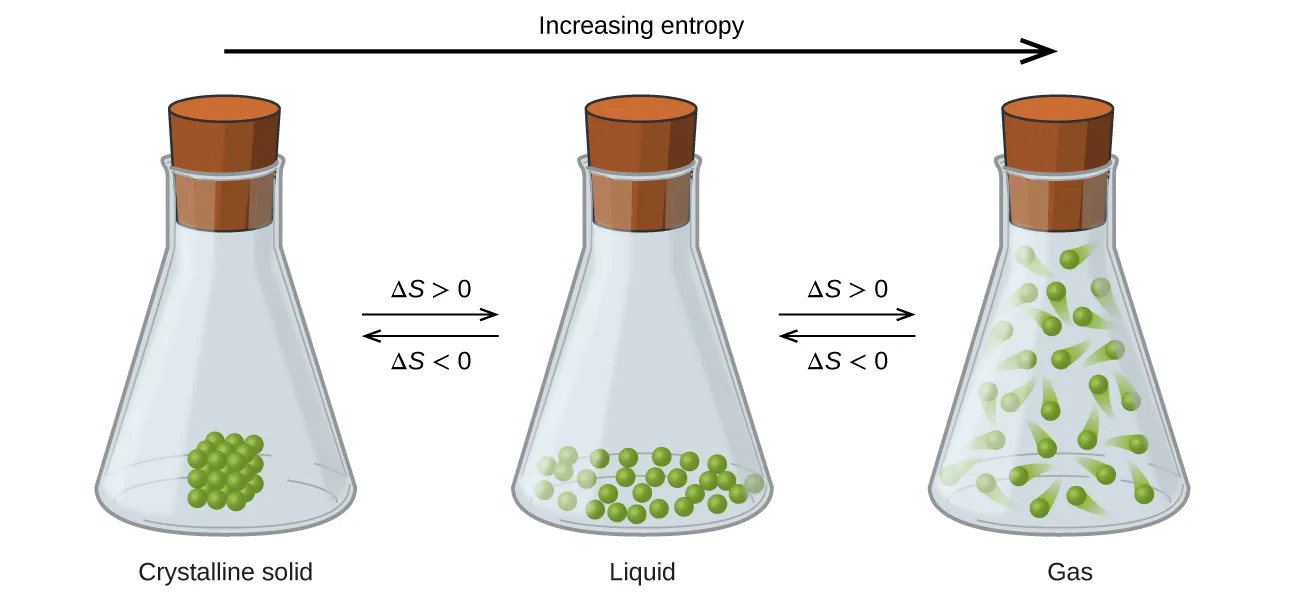
Particle diagrams compare a crystalline solid, a liquid, and a gas, emphasizing how increased freedom of motion and spatial dispersal raise entropy. The arrows and labels explicitly indicate that transitions solid liquid gas correspond to (and the reverse gives ). Source
Solid liquid gas: increases
Gas liquid solid: decreases
Why the trend works
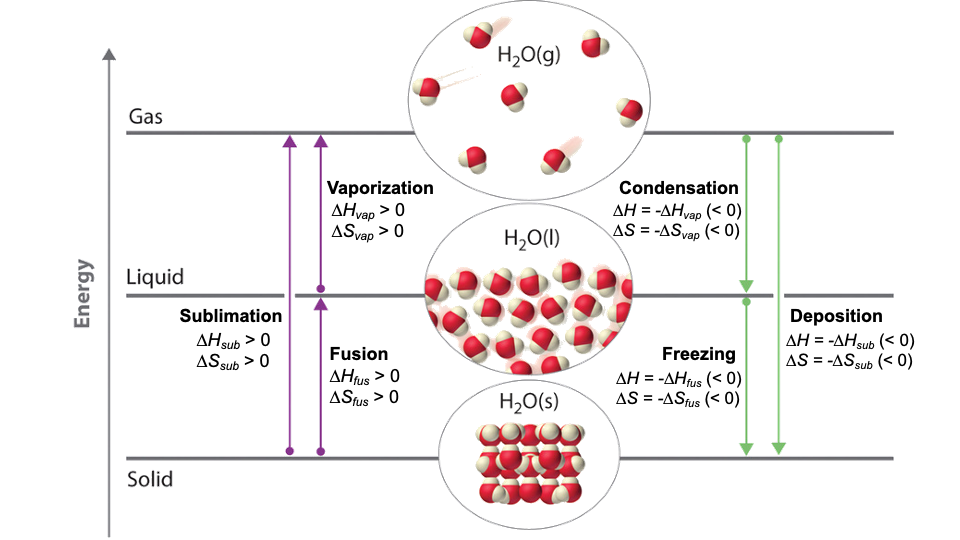
A phase-transition schematic uses arrows between solid, liquid, and gas to summarize the sign of entropy change for each process. It highlights that moving toward more separated, freer particles (solid liquid gas) gives , while the reverse direction gives . Source
Solids: particles are constrained to fixed positions (least freedom).
Liquids: particles move past each other but remain close together (more freedom).
Gases: particles move independently and occupy the container (most dispersal).
How to compare sizes (relative magnitude)
Vaporisation (liquid gas) usually gives a larger positive than melting (solid liquid), because forming a gas greatly increases spatial dispersal.
Predicting for volume changes (especially gases)
For gases, entropy is strongly linked to available volume at constant temperature.
Expansion vs compression (constant temperature)
Gas expands into a larger volume:
Gas is compressed into a smaller volume:
Particle model reasoning
Larger volume means gas particles can occupy more positions and spread out more, so there are more accessible arrangements (more dispersal).
Smaller volume restricts positions, reducing dispersal.
Predicting from moles of gas in chemical reactions
When comparing reactants and products, focus on how many moles of gaseous particles are present, since gases dominate entropy changes.
Core mole-count rule (AP favourite)
If moles of gas increase from reactants to products: tends to be positive
If moles of gas decrease: tends to be negative
If moles of gas are unchanged: may be small, so look for other cues (phase changes, dissolution/precipitation, complexity).
How to apply it correctly
Use the balanced equation and count only gaseous species for this heuristic.
The sign prediction is strongest when the change in gas moles is large (for example, 1 mol gas 3 mol gas).
Common situations
Producing a gas from liquids/solids/aqueous species often gives .
Consuming gas to form liquids/solids often gives .
Reactions with equal gas moles can still have nonzero , but the mole rule alone may not decide the sign.
FAQ
Look for phase changes (forming/consuming a gas is most decisive).
Also consider whether particles become more “free-moving” (e.g., liquid to aqueous ions tends to increase dispersal), but the gas/phase cue usually dominates.
At constant temperature for a gas, increasing pressure typically decreases volume, so entropy decreases.
Decreasing pressure allows expansion, so entropy increases.
It strongly suggests $\Delta S<0$, but it is still a heuristic.
If other major changes occur (for example, producing a gas from condensed phases elsewhere in the balanced equation), they can compete with the mole-count trend.
Gases occupy far more volume and have much greater freedom of motion, so changes involving gases usually change dispersal far more than rearrangements within condensed phases.
Use the stoichiometric coefficients from the balanced equation as the mole counts for gaseous species.
Then compare total gaseous moles on each side to predict the sign.
Practice Questions
Predict the sign of for each process: (a) (b) A gas at constant temperature is compressed to half its original volume. (c)
[3 marks]
(a) (solid to liquid increases entropy). [1]
(b) (compression decreases entropy). [1]
(c) (3 mol gas reactants to 2 mol gas products). [1]
Consider the following changes. For each, state whether is positive, negative, or approximately zero, and justify using phase/volume/gas-mole arguments. (a) (b) (c) A sealed syringe of gas is allowed to expand at constant temperature when the plunger is released.
[6 marks]
(a) with justification: gas to liquid reduces dispersal. [2: 1 sign, 1 reasoning]
(b) with justification: 4 mol gas to 2 mol gas. [2: 1 sign, 1 reasoning]
(c) with justification: volume increases at constant (expansion). [2: 1 sign, 1 reasoning]

