AP Syllabus focus: ‘As temperature increases, kinetic energy is spread over a wider range of particle speeds, so the system’s entropy increases.’
Temperature changes do more than speed particles up: they change how energy is distributed among many possible molecular motions. Kinetic molecular theory and kinetic energy distributions provide a particle-level explanation for why higher temperature corresponds to higher entropy.
Temperature in Kinetic Molecular Theory (KMT)
In kinetic molecular theory, temperature is tied to particle motion, especially for gases where particles are far apart and moving freely.
Temperature (Kelvin): A measure proportional to the average translational kinetic energy of particles in a sample.
The key idea for entropy is not just the average energy, but how energy can be dispersed among particles and among available energy states.
Average kinetic energy and temperature
As increases, the average translational kinetic energy increases, meaning particles (on average) move faster and collide more energetically.
= average translational kinetic energy per particle (J)
= Boltzmann constant ()
= absolute temperature (K)
= average translational kinetic energy per mole (J mol)
= gas constant ()
Even when the average kinetic energy is specified by , individual particles can still have a wide range of kinetic energies.
Kinetic Energy Distributions (Maxwell–Boltzmann)
A gas sample at a given temperature contains particles with many different speeds. The Maxwell–Boltzmann distribution describes the spread of particle speeds (and kinetic energies) in a population.
Kinetic energy distribution: The statistical spread of particle kinetic energies in a system; at a fixed temperature, some particles have low energy while others occupy a higher-energy “tail.”
How the distribution changes with temperature
When temperature increases, the kinetic energy distribution changes in two important qualitative ways:
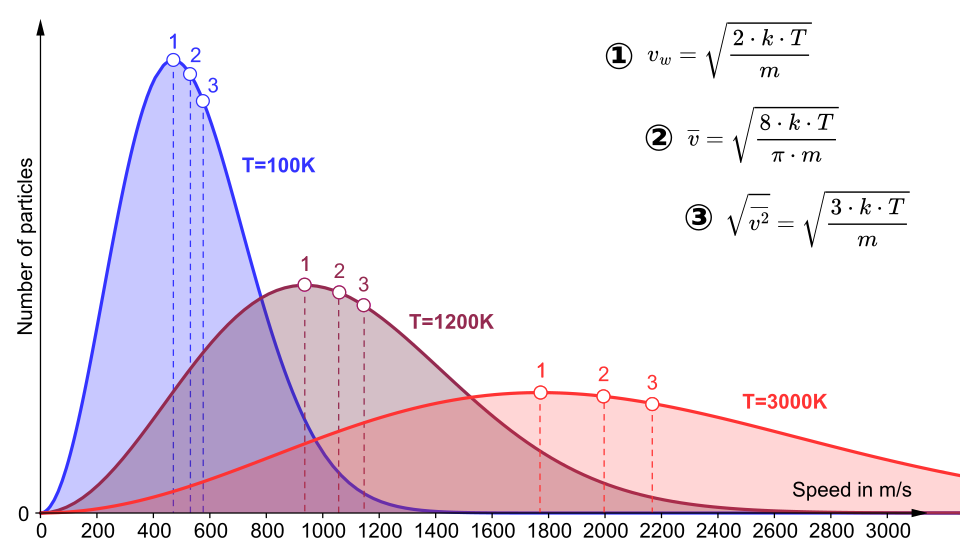
Maxwell–Boltzmann speed distributions at several temperatures on the same axes. The higher-temperature curve is broader and shifted toward higher speeds, while the peak height decreases; this visualizes how raising spreads molecules over a wider range of speeds (and kinetic energies). The marked characteristic speeds help connect “most probable,” average, and rms speeds to the distribution shape. Source
The peak lowers and shifts to higher speed/energy (the “most probable” speed increases).
The curve broadens: energies are spread over a wider range.
The high-energy tail becomes more populated, so a larger fraction of particles have very high kinetic energies.
This is the syllabus statement in particle terms: raising spreads kinetic energy across more possible particle speeds, rather than simply adding the same energy to every particle.
Why “wider range” matters for entropy
Entropy increases with the number of ways energy can be distributed among particles and available energy levels. At higher :
More energy levels (translational states) become significantly accessible.
Energy can be partitioned among particles in more distinct combinations.
The system has more possible microscopic arrangements consistent with the same macroscopic state.
In other words, heating increases energy dispersal, not necessarily “disorder” in a visual sense, but the count of energetically allowed microstates.
Connecting energy dispersal to entropy (qualitative)
For AP Chemistry purposes, treat entropy here as tracking how widely energy is spread among particle motions.
At low temperature, most particles cluster near lower kinetic energies; fewer energy states are meaningfully occupied.
At high temperature, particles occupy a broader span of kinetic energies; many more microstates contribute.
This also explains why temperature is a powerful lever on thermodynamic behaviour: increasing tends to increase the entropy of a substance because energy becomes distributed across a larger set of accessible molecular motions and speeds.
Common reasoning cues (no calculation)
When comparing two samples of the same gas:
The sample at higher has:
higher average kinetic energy
broader kinetic energy distribution
higher entropy due to greater energy dispersal
When interpreting a distribution sketch:
A taller, narrower curve corresponds to lower and lower entropy (less spread in particle energies).
A shorter, wider curve corresponds to higher and higher entropy (more spread in particle energies).
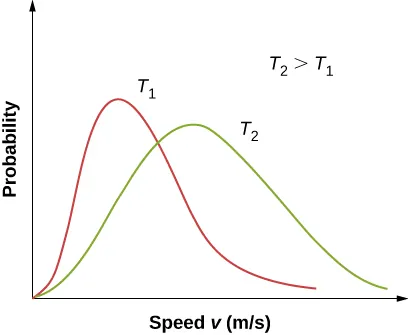
Comparison of Maxwell–Boltzmann speed distributions at two temperatures (). The higher-temperature curve is flatter, broader, and shifted to higher speeds, indicating that a greater fraction of molecules occupy higher-energy states. This is the graphical basis for saying that higher increases energy dispersal across many possible particle speeds. Source
FAQ
The total number of particles stays the same.
A broader spread over energy means the same “area under the curve” is distributed across a wider range, so the maximum height decreases.
Not always.
For temperature effects, “more energy dispersal among more accessible microstates” is more precise than visual disorder, especially for gases where randomness is already high.
No.
Heating changes the distribution: some molecules gain a lot of kinetic energy, some gain little, and collisions continually reshuffle energies, producing a wider range.
It represents a small fraction of particles with unusually large kinetic energies at that moment.
At higher $T$, that fraction increases, meaning more particles occupy very fast (high-energy) states.
Entropy tracks how many microscopic ways the system’s energy can be arranged.
A higher temperature makes many more kinetic energy values meaningfully populated, increasing the number of possible microscopic energy arrangements and therefore increasing entropy.
Practice Questions
(2 marks) For a gas sample, describe how increasing temperature changes the distribution of molecular kinetic energies, and state what happens to entropy.
Distribution becomes broader and shifts to higher kinetic energy / higher speeds (1)
Entropy increases because kinetic energy is dispersed over a wider range of particle energies (1)
(5 marks) Two sealed containers hold the same amount of the same ideal gas, one at and one at a higher temperature .
(a) Explain, using kinetic molecular theory, how the average translational kinetic energy compares between the two samples.
(b) Describe two changes you would expect in the Maxwell–Boltzmann distribution at compared with .
(c) Explain why the entropy is higher at in terms of energy dispersal.
(a) Average translational kinetic energy is proportional to temperature; higher at (1)
(b) Any two:
peak shifts to higher energy/speed (1)
curve broadens / wider spread of energies (1)
peak lowers (1)
larger high-energy tail / greater fraction at high energy (1)
(c) Entropy higher because energy is spread among more accessible energy states/microstates; more possible distributions of kinetic energy (2)

