AP Syllabus focus: ‘Light can supply energy to drive an overall unfavorable conversion, such as converting carbon dioxide to glucose in photosynthesis.’
Light energy can power chemical changes that are not thermodynamically favored in the dark. In AP Chemistry, this is framed as using photons to supply energy that enables an overall “uphill” conversion.
What it means to “drive an unfavorable process” with light
A reaction described as unfavorable under given conditions has a positive overall Gibbs free energy change (conceptually, it is not spontaneous without energy input). Light-driven chemistry works because absorbed light raises molecules to higher-energy states, making otherwise inaccessible pathways possible.
Photochemistry and excited states
When a molecule absorbs light, it can be promoted from a lower-energy electronic state to a higher-energy one.
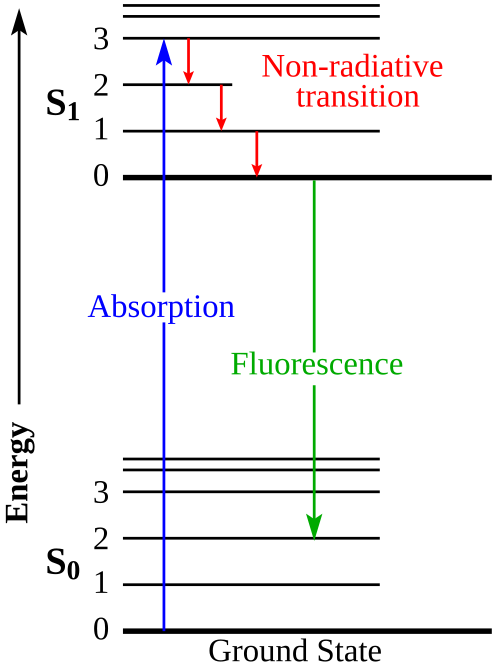
Jablonski diagram showing excitation from the ground electronic state () to an excited state (), followed by relaxation pathways such as vibrational relaxation/internal conversion and radiative emission (fluorescence). The vertical axis represents energy, making it clear why an absorbed photon can temporarily place molecules in higher-energy, more reactive states. Source
Excited state: a higher-energy electronic state formed when a species absorbs energy (often light), making it more reactive or enabling different electron-transfer pathways.
Excited states are typically short-lived, so useful light-driven chemistry must quickly convert that temporary energy into more stable chemical potential energy (stored in bonds or separated charges).
How photons supply usable energy
Light delivers energy in discrete packets called photons, and the energy depends on the light’s frequency (and inversely on wavelength). This sets an important constraint: only sufficiently energetic light can initiate a given photochemical step.
= energy of one photon (J)
= Planck’s constant ()
= frequency of light ()
In chemical terms, photon absorption can:
Promote electrons to higher-energy orbitals, enabling electron transfer that is not favorable from the ground state.
Create reactive intermediates (such as radicals or excited pigments) that follow new reaction pathways.
Generate charge separation (an electron and “hole” in different locations), which can be harnessed to do chemical work.
Photosynthesis as the key AP example: CO₂ to glucose
The syllabus highlights photosynthesis because it clearly illustrates an overall unfavorable conversion powered by light: carbon dioxide and water are converted into a more energy-rich carbohydrate.
Why CO₂ reduction is “uphill”
CO₂ is highly oxidized and relatively low in chemical potential energy. Converting CO₂ into C–H and C–C bonds (as in sugars) requires an energy input because it is effectively a reduction process that builds higher-energy products.
Light supplies that input by first energizing a pigment (e.g., chlorophyll-like molecules) and then driving a sequence of electron-transfer events that, overall, store energy in chemical form rather than releasing it as heat.
How the energy is captured rather than wasted
To actually “drive” the unfavorable conversion, the system must avoid simply re-emitting light or losing the energy by collisions. The key design features (conceptually) are:
Absorption: a chromophore/pigment strongly absorbs specific wavelengths.
Rapid electron transfer: the excited electron is transferred to an acceptor before relaxation.
Energy storage: the separated charges are used to make energy-carrying chemicals (often discussed as ATP- and NADPH-like carriers in the photosynthesis model), which then power CO₂-to-sugar chemistry.
Catalysis and stepwise pathways: the overall transformation is broken into many smaller steps so the energy can be managed and directed.
What you should be able to explain on the AP exam
Students should be able to connect the wording in the syllabus to core thermodynamic reasoning:
Light is an external energy source that can enable an overall nonspontaneous chemical change.
The role of light is not “heat,” but photon absorption that creates excited states and enables electron movement.
In the photosynthesis example, light energy is converted into chemical potential energy, allowing CO₂ to be converted into glucose despite being unfavorable without that energy input.
FAQ
Shorter wavelengths (higher frequency) carry more photon energy.
If photons are too low-energy, absorption may not reach the electronic transitions needed to initiate the key step.
Quantum yield measures how many target events occur per photon absorbed.
Low quantum yield indicates substantial losses (e.g., fluorescence, internal conversion), reducing the ability to drive an unfavourable net change.
Pigments broaden and strengthen absorption in useful spectral regions.
They can also spatially and energetically channel excitation to a reactive site, improving the chance of productive electron transfer.
Fluorescence or phosphorescence (light emission)
Internal conversion (non-radiative relaxation)
Collisional deactivation (heat transfer to surroundings)
These compete with productive chemistry.
Separated charges can recombine, returning the system to lower energy without doing chemical work.
Effective systems separate charges quickly and use them in fast follow-up reactions to “lock in” the energy.
Practice Questions
(2 marks) In terms of energy and reaction favourability, explain how light can enable an overall unfavourable chemical conversion such as to glucose.
Light provides an external energy input via photon absorption / excitation (1)
The absorbed energy is stored as chemical potential energy (e.g., in higher-energy bonds or separated charges), allowing an otherwise nonspontaneous overall change (1)
(5 marks) A photochemical system absorbs visible light and then carries out a net reduction process that is unfavourable in the dark. Explain, using chemical reasoning, how photon absorption can lead to a sustained chemical change rather than immediate energy loss.
Absorption promotes a species to an excited electronic state / higher-energy electron distribution (1)
Excited state enables electron transfer pathways not accessible from the ground state (1)
Rapid charge separation or formation of reactive intermediates competes with relaxation (1)
Energy is converted into longer-lived chemical potential energy (e.g., bonds or redox carriers) (1)
Stepwise/catalysed pathways allow the net “uphill” conversion to proceed using the stored energy (1)

