AP Syllabus focus: ‘An external energy source can make an unfavorable process occur, such as using electrical energy to drive an electrolytic cell or charge a battery.’
Driving an unfavorable chemical change is possible when you supply energy from outside the system. In AP Chemistry, the key idea is using electrical energy to force electron transfer that would not proceed on its own.
What “driving” an unfavorable process means
A reaction described as unfavorable (nonspontaneous) under the stated conditions will not proceed to a noticeable extent unless energy is continuously provided from an external source.
Nonspontaneous (thermodynamically unfavored) process — a process that requires an input of energy to occur as written under the specified conditions.
In electrochemistry, that energy input is often delivered by a power supply that pushes electrons through a circuit in a chosen direction, even if the chemical system “prefers” the opposite direction.
Electrical energy as a controllable energy input
Electrical energy is useful because you can control:
Voltage (potential difference): how strongly electrons are pushed
Current: how fast charge flows (how quickly the forced reaction proceeds)
Time: how long the forcing is applied, determining total chemical change
Electrolytic cells: forcing redox to occur
The most direct AP Chemistry context is electrolysis, where an external voltage drives a redox reaction that is unfavorable under those conditions.
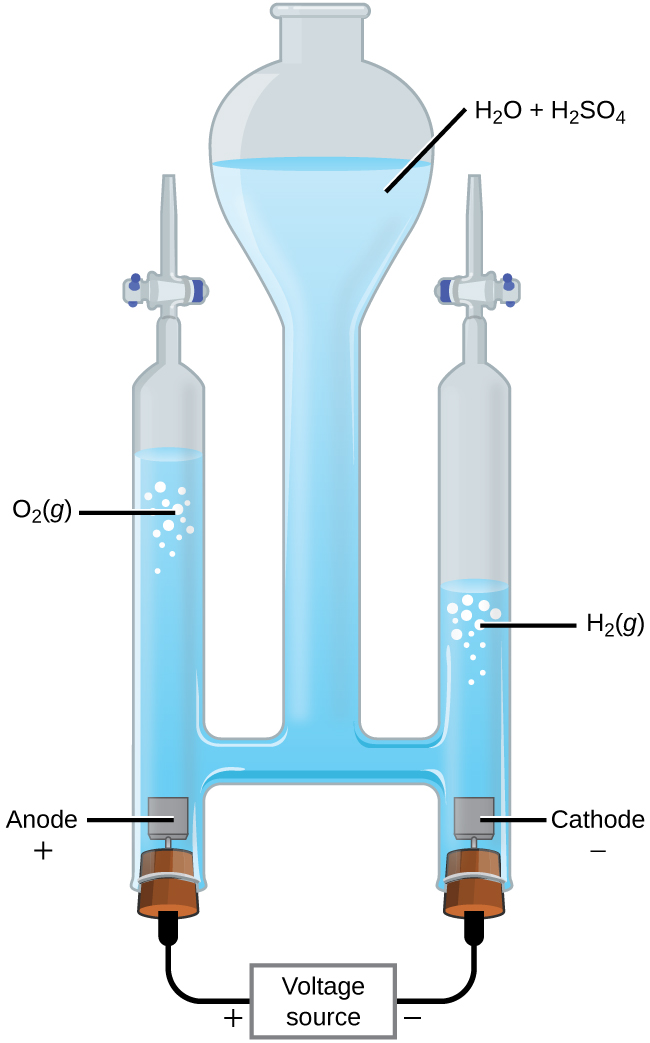
Electrolysis setup showing how an external voltage source forces electron flow through an electrolytic cell. The diagram labels the anode/cathode and indicates the products formed at each electrode, reinforcing that oxidation occurs at the anode and reduction occurs at the cathode even in a nonspontaneous (driven) process. Source
Electrolytic cell — an electrochemical setup in which an external electrical energy source forces a nonspontaneous redox reaction to occur.
In an electrolytic process:
The power supply acts like an “electron pump.”
Electrons are driven from one electrode to the other to make the desired oxidation and reduction happen.
The reaction continues only while electrical energy is supplied (turn off the power, and the forced reaction stops).
What is being “stored” or “paid for”?
When you drive an unfavorable reaction electrically, you are converting electrical energy into chemical potential energy in the products. Those products are higher in free energy than the reactants for the reaction as written, which is why continuous input is required.
Charging a battery: reversing the spontaneous direction
A rechargeable battery operates in two modes:
Discharging: a thermodynamically favored redox reaction produces electrical energy.
Charging: an external power source forces the reverse (unfavorable) reaction, regenerating reactants and storing energy.
Charging is therefore a real-world example of using electrical energy to drive an unfavorable process: the charger supplies energy to push electrons opposite the direction they would flow spontaneously.
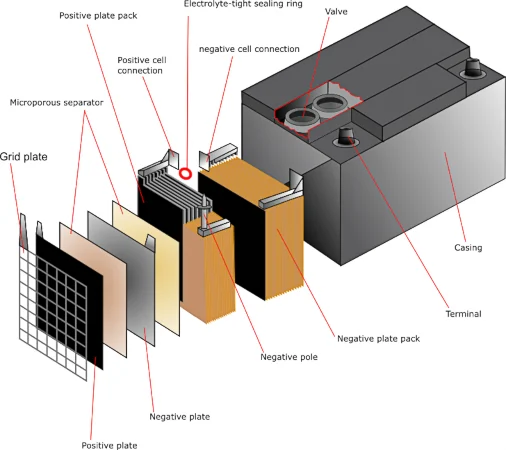
Schematic of a lead-acid battery cell identifying the positive and negative electrodes and the electrolyte. It helps visualize how the cell’s chemistry is arranged so that discharge can deliver electrical energy, while charging requires an external applied potential to drive the reverse redox changes. Source
Minimum electrical “push” required (conceptual voltage requirement)
To make a nonspontaneous redox reaction proceed, the applied electrical driving force must be large enough to overcome the reaction’s inherent tendency to go the other way.
= electrical energy supplied to the system, in J
= moles of electrons transferred per mole of reaction as written, in mol
= Faraday constant, C mol
= externally applied potential difference, in V (J C)
In practice, real systems also require extra voltage beyond the theoretical minimum due to non-ideal effects (for example, resistance and electrode surface effects), so the applied voltage must be sufficiently high to sustain the desired current.
Key takeaways for AP Chemistry
An external energy source can make an unfavorable process occur; in this subtopic, that source is electrical energy.
Electrolytic cells use a power supply to force an otherwise unfavorable redox reaction.
Charging a battery is an application of forcing the nonspontaneous reverse of the discharge reaction.
The external circuit determines the direction and extent of forced electron flow, linking electrical input to chemical change.
FAQ
The supply fixes the direction of electron flow in the external circuit.
Which reaction occurs depends on:
which species are present near each electrode
how readily each can be oxidised/reduced at the electrode surface under the imposed conditions
Because the forced direction is maintained by continuous electrical work.
Once removed, there is no external driving force to keep pushing electrons “uphill,” so sustained reaction ceases.
Overpotential is extra voltage needed beyond the ideal thermodynamic requirement.
It arises from electrode surface kinetics and can increase the necessary applied voltage even when the overall chemistry is feasible.
Some electrical energy is dissipated through internal resistance and other inefficiencies.
This converts part of the input energy into thermal energy rather than stored chemical potential energy.
Look for signs that require current:
product formation only while current flows
measurable current draw from the power source
reversal of the spontaneous discharge direction in a rechargeable cell
Practice Questions
(2 marks) Explain how electrical energy can be used to make a thermodynamically unfavourable chemical process occur.
States that an external power source supplies energy/work to the system to force the reaction (1)
Links this to electrolysis/charging a battery as an example of driving a non-spontaneous redox reaction (1)
(5 marks) A student claims: “If a redox reaction is unfavourable, it cannot be made to occur.” Using an electrolytic cell or battery charging as context, assess this claim by describing the role of an external potential difference and electron flow.
Identifies the claim as incorrect: an unfavourable reaction can be driven by external electrical energy (1)
Describes that an external potential difference forces electrons to flow in the non-spontaneous direction (1)
Connects forced electron flow to driving oxidation and reduction to occur as written (1)
States that the process continues only while energy is supplied (1)
Gives an appropriate context statement (electrolytic cell or charging a battery reverses the spontaneous direction) (1)

