AP Syllabus focus: ‘A desired product can be formed by coupling an unfavorable reaction to a favorable reaction; the reactions share one or more intermediates.’
Reaction coupling explains how chemistry can “pay” for an otherwise unfavorable transformation by linking it to a favorable one through a shared chemical intermediate, so the desired product forms in an overall coordinated process.
Core idea of reaction coupling
Coupling is not just doing two reactions in the same container. The key requirement is that the reactions are chemically linked through at least one shared intermediate, so progress in one step directly enables progress in the other.
Reaction coupling: A strategy where an unfavorable reaction is made to proceed by mechanistically linking it to a favorable reaction through one or more shared intermediates.
In a coupled pathway, the intermediate is produced by one reaction step and consumed by another; this “handoff” is what connects the driving process to the driven process.
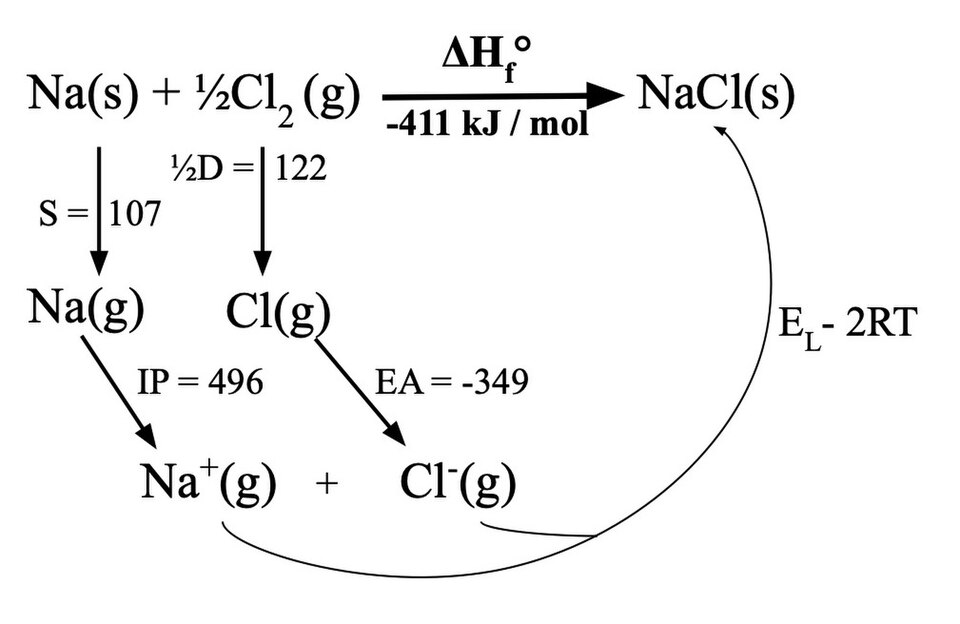
Born–Haber cycle diagram for forming an ionic solid (NaCl) from its elements, broken into labeled energetic steps such as atomization, ionization energy, electron affinity, and lattice enthalpy. The diagram emphasizes how intermediate species (gaseous atoms/ions) connect steps in a multi-step pathway, while the overall formation enthalpy is obtained by summing the step changes (Hess’s law). Source
Intermediate: A species formed in one step and consumed in a later step of a multi-step process; it does not appear in the overall balanced change.
What “favorable” and “unfavorable” mean in coupling
For AP Chemistry, classify reactions by thermodynamic tendency:
Favorable (spontaneous): can proceed under the stated conditions without continuous external work.
Unfavorable (nonspontaneous): will not proceed to a meaningful extent unless driven.
Coupling uses a favorable process as the energy source (the “driver”) and the unfavorable process as the energy sink (the “payload”). The driver must be able to generate the intermediate in a way that the payload can immediately use.
How shared intermediates make coupling work
Mechanistic linkage (not coincidence)
A coupled system is designed so that:
Step A (favorable) creates intermediate X.
Step B (unfavorable) consumes intermediate X to form the desired product.
Because X is continually removed by Step B, Step A is pulled forward (X does not accumulate), maintaining flow through both steps.
Why the intermediate matters
A shared intermediate provides a physical “currency” that transfers driving force:
Electron-transfer coupling: the shared intermediate can be electrons moving through a circuit or a redox mediator that is alternately oxidized and reduced.
Bond-energy coupling: the shared intermediate can be a high-energy reactive species that readily reacts onward, transferring its stored potential to the unfavorable step.
Recognising coupled reactions in chemical descriptions
Look for these cues:
The overall description mentions a desired reaction that is unfavorable “on its own,” but proceeds when paired with another.
Two reactions are written as a sequence where the product of one is explicitly a reactant of the next.
A species is produced and then immediately consumed (typical intermediate behaviour), sometimes described as “regenerated,” “cycled,” or “catalytic” in a pathway.
What coupling is not
Simply adding reactants of two separate reactions together without a shared intermediate.
Running two independent reactions side-by-side where neither influences the other’s progress.
A catalyst “speeding up” an unfavorable reaction (a catalyst affects rate, not thermodynamic favourability).
Common pitfalls
Treating any simultaneous reactions as “coupled” without identifying a shared intermediate.
Forgetting that coupling requires a pathway: the intermediate must be chemically accessible and reactive under the same conditions.
Confusing an intermediate with a catalyst: a catalyst is regenerated, but an intermediate is created and later consumed within the multi-step sequence.
FAQ
Evidence can include time-resolved detection of a transient species, trapping experiments, or isotope labelling that shows the species is formed then consumed.
Useful approaches:
Rapid spectroscopy
Quench-and-analyse methods
Kinetic signatures of an intermediate
Yes. In electrochemical contexts, electrons transferred through an external circuit can function as the shared intermediate connecting oxidation and reduction half-processes.
Side reactions can compete for the intermediate, diverting it away from the payload step.
Common causes:
Intermediate reacts with solvent/impurities
Multiple consumption pathways
A catalytic cycle regenerates a catalyst; coupling specifically links a favourable and unfavourable transformation via intermediate transfer.
An intermediate is typically produced then consumed; a catalyst is present throughout and is regenerated.
They choose a driving reaction that produces a compatible, controllable intermediate and ensure conditions match the payload step, while minimising competing reactions through selectivity and reaction environment control.
Practice Questions
(2 marks) Explain what is meant by reaction coupling and state the essential feature that distinguishes a coupled pair of reactions from two independent reactions occurring in the same container.
1 mark: States that an unfavourable reaction can be made to proceed by linking it to a favourable reaction.
1 mark: Identifies the essential feature: the reactions share one or more intermediates (product of one step is reactant of another), giving mechanistic linkage.
(5 marks) A student claims that any two reactions can be coupled as long as one is favourable. Critique this claim by describing the role of a shared intermediate and giving two specific reasons why some reaction pairs cannot be effectively coupled.
1 mark: States that coupling requires a shared intermediate (mechanistic connection), not merely co-occurrence.
1 mark: Explains that the favourable step must generate an intermediate that the unfavourable step consumes to form the desired product.
1 mark: Reason 1 (any valid): intermediate not reactive/selective enough under the conditions (e.g., reacts with solvent/side reactions).
1 mark: Reason 2 (any valid, distinct): incompatible conditions (pH, solvent, temperature, phases) prevent intermediate transfer/consumption.
1 mark: Additional valid critique: intermediate accumulates or is not removed, so linkage fails; or competing pathways consume the intermediate faster than the payload step.

