AP Syllabus focus: ‘Under nonstandard conditions, cell potential depends on concentrations of active species; the farther from equilibrium, the greater the magnitude of the potential.’
Electrochemical cell voltages are not fixed numbers: changing ion concentrations or gas pressures changes the driving force for electron flow.
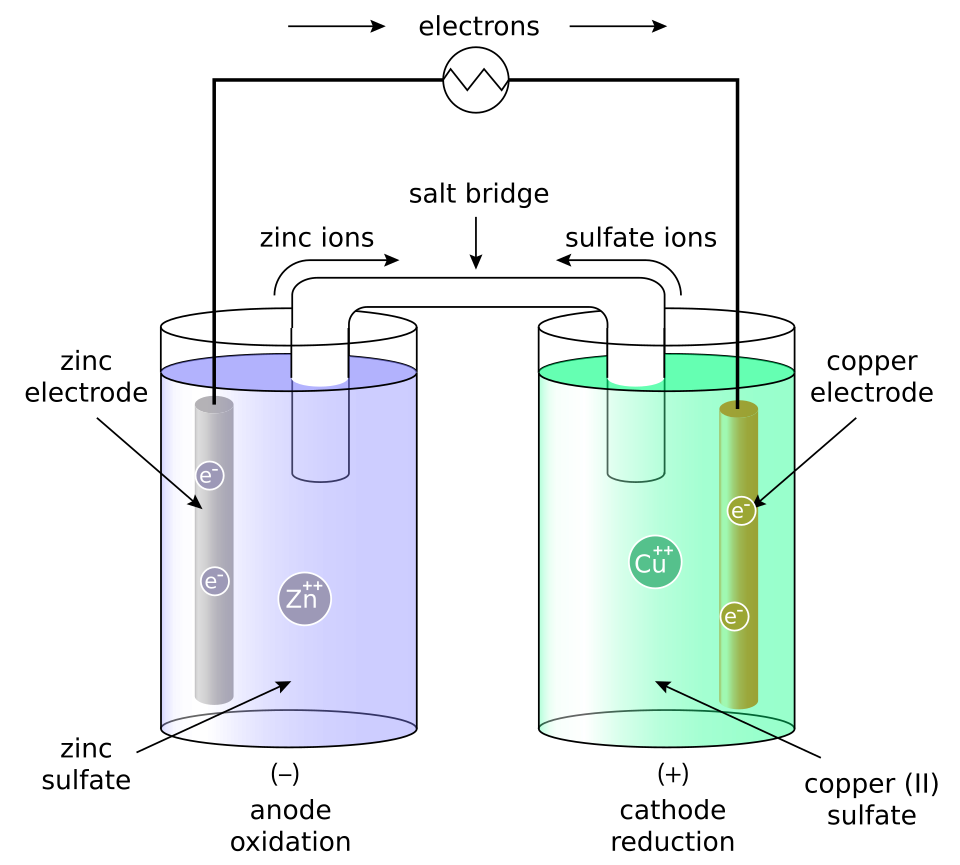
Labeled galvanic cell diagram highlighting anode vs. cathode, the direction of electron flow through the wire, and ion migration through the salt bridge to maintain charge balance. This visual helps connect concentration changes in either half-cell to changes in the reaction quotient , which in turn shifts . Source
This section explains how and why concentration changes shift measured cell potential.
Core idea: concentration affects the “push” of the redox reaction
Cell potential (): The electrical potential difference between the two electrodes of an electrochemical cell, measured in volts (V).
For a given cell reaction, depends on how strongly the system “wants” to move from its current composition toward products. That driving force changes as concentrations of active species change in either half-cell.
What counts as an “active species”
Only species whose effective concentration can change appear in the concentration dependence:
Aqueous ions and solutes (use molar concentration as an AP-level approximation)
Gases (use partial pressure as an AP-level approximation)
Exclude pure solids and pure liquids (their activity is effectively constant)
This matters because adding more solid metal electrode does not directly change , but changing the concentration of its dissolved ions does.

Photograph of a Daniell cell (Zn/Cu) connected to a voltmeter, illustrating how cell potential is measured in practice. Because actual ion concentrations may differ from standard conditions, the observed voltage can differ from , motivating the need for the Nernst equation. Source
Direction of the change: how concentration shifts
Concentration changes influence the tendency for oxidation and reduction by changing the “availability” of reactants/products in each half-reaction.
General qualitative rules (galvanic conditions)
For the overall cell reaction written as:
Reactants → Products
Then, at constant temperature:
Increasing reactant concentration (or pressure, for gases) tends to increase
Increasing product concentration tends to decrease
Diluting reactants tends to decrease
Removing products (lowering their concentration) tends to increase
These trends reflect the syllabus idea that depends on concentrations and changes as the system composition shifts.
“Farther from equilibrium” means a larger magnitude potential
When reactant and product concentrations are very unequal (for example, reactants high and products low), the cell has a stronger driving force, so the magnitude of the potential is larger. As the reaction proceeds, reactants are consumed and products accumulate, so the driving force weakens and the measured potential typically drops in magnitude.
The Nernst relationship (linking voltage to composition)
Reaction quotient (): A ratio built from product activities over reactant activities, each raised to stoichiometric powers, using current (not necessarily equilibrium) concentrations/pressures and omitting pure solids and liquids.
A compact way to express concentration dependence is the Nernst equation.
= cell potential at the current conditions (V)
= standard cell potential (V)
= gas constant ()
= temperature (K)
= moles of electrons transferred per balanced cell reaction (mol )
= Faraday constant ()
= reaction quotient from current concentrations/partial pressures (unitless)
In this form, concentration effects enter through : changing concentrations changes , which shifts relative to .
Interpreting the sign of the concentration term (without calculation)
If increases (more products relative to reactants), increases, so the subtraction term grows and becomes smaller.
If decreases (fewer products relative to reactants), decreases, making the subtraction term smaller (or effectively adding), so becomes larger.
Common concentration-related pitfalls
Forgetting to omit solids and liquids from
Using concentrations for species that are not meaningfully “active” (for AP, assume dilute solution behaviour unless told otherwise)
Ignoring that changing either half-cell concentration can change the overall
FAQ
Their activities are effectively constant (taken as 1) because their composition does not change with amount in a way that alters chemical potential.
Adding more solid can sustain current longer, but it does not directly change $E_{\text{cell}}$.
At higher ionic strengths, ions interact and the effective activity differs from molarity.
This can make measured potentials deviate from predictions that assume ideal dilute solutions.
By changing ionic strength, they can alter activity coefficients, shifting effective activities without changing formal concentrations.
They can also affect junction potentials in experimental setups.
Gas species enter via partial pressures (as activity proxies).
Increasing a gaseous reactant’s partial pressure tends to increase $E_{\text{cell}}$; increasing a gaseous product’s partial pressure tends to decrease it.
The factor $\frac{RT}{nF}$ scales how strongly $\ln Q$ shifts the potential.
Higher $T$ makes $E_{\text{cell}}$ more sensitive to the same concentration (or pressure) ratio changes.
Practice Questions
Question 1 (1–3 marks) A galvanic cell operates with the overall reaction: . State how changes (increases/decreases/no change) when (i) is increased and (ii) is increased, at constant temperature.
(i) increases when increases (1 mark)
(ii) decreases when increases (1 mark)
Correct reasoning linking reactant↑/product↑ to change or driving force change (1 mark)
Question 2 (4–6 marks) Two identical galvanic cells run the reaction at the same temperature.
Cell A: high and low .
Cell B: low and high .
(a) Compare the magnitudes of for A and B. (b) Explain using concentration dependence (you may refer to qualitatively). (c) State whether changing the mass of the solid electrodes (Fe or Ag) directly changes .
(a) Cell A has larger than Cell B (1 mark)
(b) Identifies as reactant and as product for the written reaction (1 mark)
(b) Explains that A has smaller (products/reactants) than B, so is larger for A (2 marks)
(b) Links “farther from equilibrium”/greater driving force to larger magnitude potential for A (1 mark)
(c) Electrode mass of pure solids does not directly affect (1 mark)

