Elements, compounds and mixtures
· Element = a substance made of one type of atom only and cannot be chemically broken down into simpler substances.
· Compound = atoms of different elements chemically bonded in a fixed ratio.
· Mixture = two or more substances not chemically bonded together in no fixed ratio; components can be separated by physical methods.
· Elements and compounds are pure substances; mixtures are not.
· Homogeneous mixture = uniform composition throughout (for example, a solution).
· Heterogeneous mixture = non-uniform composition with visible phases or particles.
· In exams, always link fixed ratio + chemical bonding to compounds, and variable composition + physical separation to mixtures.

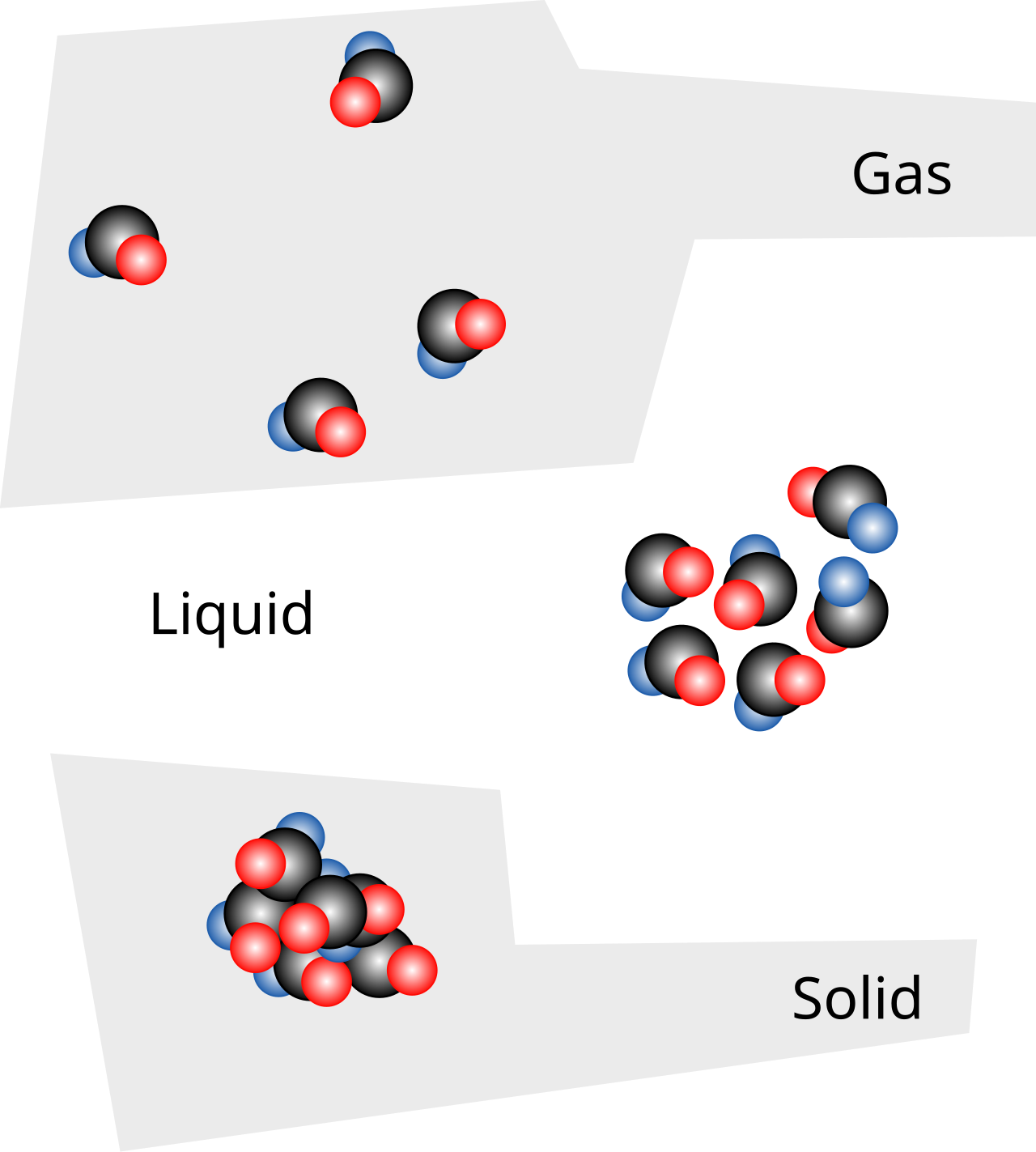
This diagram compares homogeneous mixtures, heterogeneous mixtures, compounds, and elements at the particle level. It is especially useful for IB questions that ask you to classify matter from a particle diagram rather than from words alone. Students should use it to connect fixed ratio, chemical bonding, and uniformity of composition to the correct type of substance. Source
Separating mixtures
· Choose a separation method by comparing particle size, solubility, boiling point, and attraction to stationary/mobile phases.
· Filtration separates an insoluble solid from a liquid.
· Evaporation removes the solvent to leave a dissolved solid behind.
· Recrystallization purifies a soluble solid by dissolving it in a hot solvent and then cooling to form pure crystals.
· Distillation separates substances using differences in boiling point; simple distillation is commonly used to recover a solvent from a solution.
· Paper chromatography separates dissolved substances because they have different relative attractions to the stationary phase and mobile phase.
· Solvation means particles of a solute become surrounded by solvent particles when dissolving.
· For purification questions, state both the method used and the property difference it depends on.
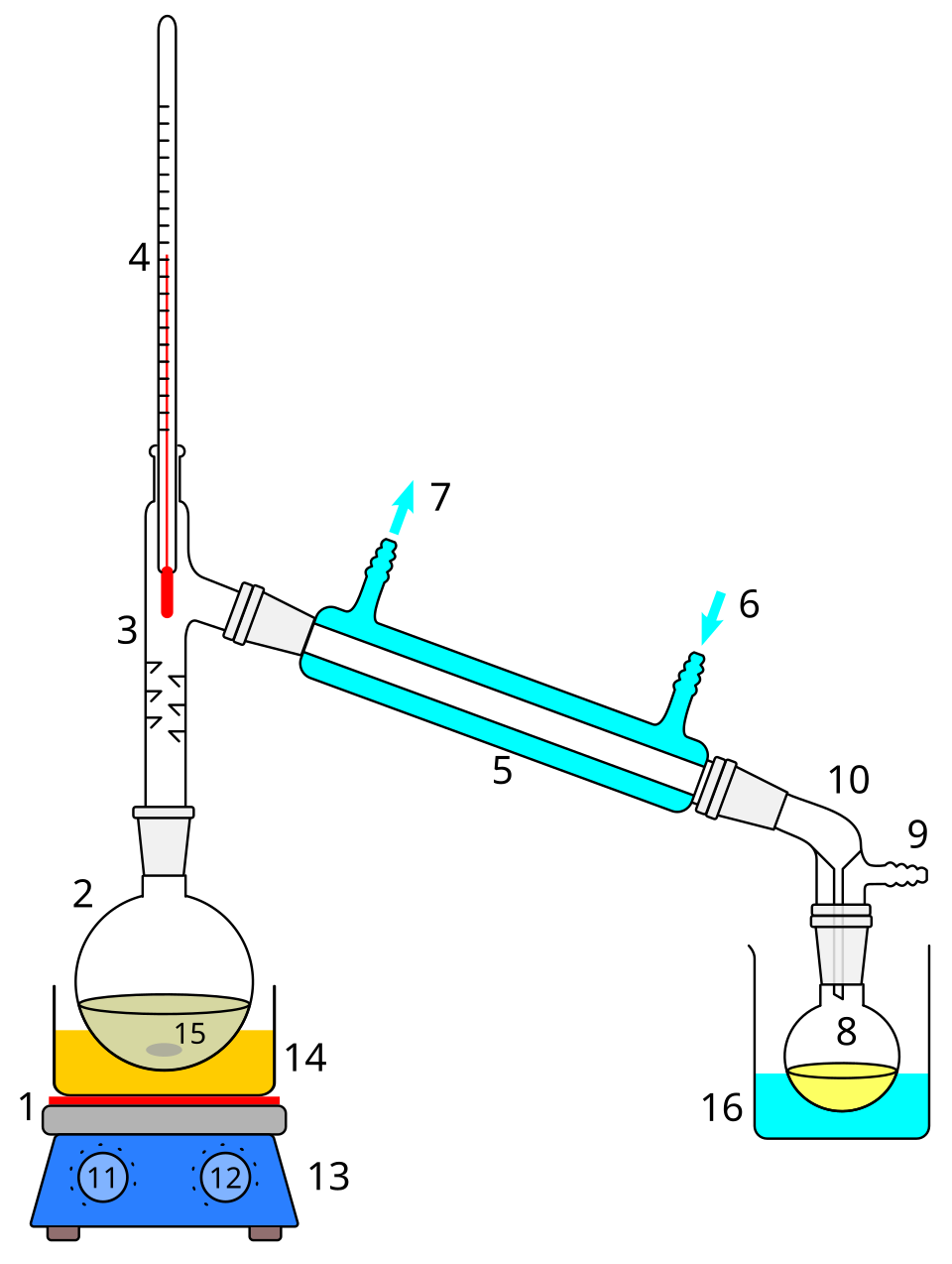
This labelled apparatus diagram shows how simple distillation separates substances using differences in boiling point. It helps students identify the flask, condenser, and receiving vessel, and explain how a vapor is formed and then condensed. This is useful for practical-method and purification questions. Source
Kinetic molecular theory
· Kinetic molecular theory (KMT) explains the behaviour of solids, liquids and gases in terms of moving particles.
· All matter is made of tiny particles that are in constant motion.
· The average kinetic energy of particles increases as temperature increases.
· Solids: particles are closely packed, vibrate in fixed positions, and have the least freedom of movement.
· Liquids: particles are still close together but can move past one another, so liquids flow.
· Gases: particles are far apart, move rapidly and randomly, and are easily compressed.
· Use KMT to explain properties such as shape, volume, compressibility, density, and ability to flow.
This diagram compares the particle arrangement and spacing in a solid, liquid, and gas. It is useful for explaining why gases are compressible, liquids flow, and solids keep a fixed shape and volume. It directly supports particle-level explanations expected in IB Chemistry. Source
States of matter and their properties
· Solid = fixed shape and fixed volume because particles remain in set positions.
· Liquid = no fixed shape but fixed volume because particles can move while remaining close together.
· Gas = no fixed shape and no fixed volume because particles move freely and spread out to fill the container.
· In particle explanations, always refer to particle arrangement, particle motion, and strength of attractions where relevant.
· A gas has low density and is highly compressible because there is a lot of empty space between particles.
· Solids and liquids are much less compressible because particles are already close together.
Changes of state
· Melting: solid → liquid.
· Freezing: liquid → solid.
· Vaporization: liquid → gas.
· Evaporation is vaporization at the surface; boiling occurs throughout the liquid.
· Condensation: gas → liquid.
· Sublimation: solid → gas.
· Deposition: gas → solid.
· During a change of state, energy changes are used to change particle spacing/attractions, so temperature stays constant while the change happens.
· In exam answers, distinguish physical change from chemical change: the substance remains the same, only the state changes.
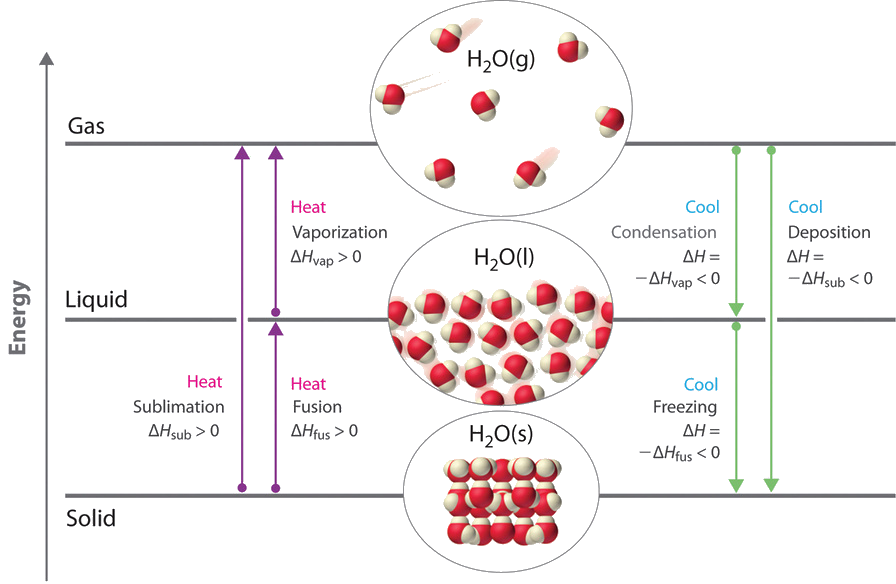
This diagram shows the six main changes of state between solid, liquid, and gas. It helps students match each process to the correct direction of change, especially sublimation, deposition, vaporization, and condensation. It is also useful for linking phase changes to energy transfer. Source
State symbols in equations
· Use (s) for solid, (l) for liquid, (g) for gas, and (aq) for aqueous.
· (aq) means the substance is dissolved in water.
· State symbols are essential in chemical equations because they show the physical state of each substance.
· Example: NaCl(s) → NaCl(aq) shows dissolving, not a chemical reaction.
· Do not confuse (l) with (aq): a liquid is a pure liquid, while aqueous means dissolved in water.
Temperature and kinetic energy
· Temperature measures the average kinetic energy of particles.
· Higher temperature means particles have a greater average kinetic energy and move faster.
· The SI unit of temperature is the kelvin (K).
· The Kelvin and Celsius scales have the same size interval: a change of 1 °C = 1 K.
· Convert using: .
· Convert back using: .
· In IB questions, use kelvin when temperature is linked to particle kinetic energy.
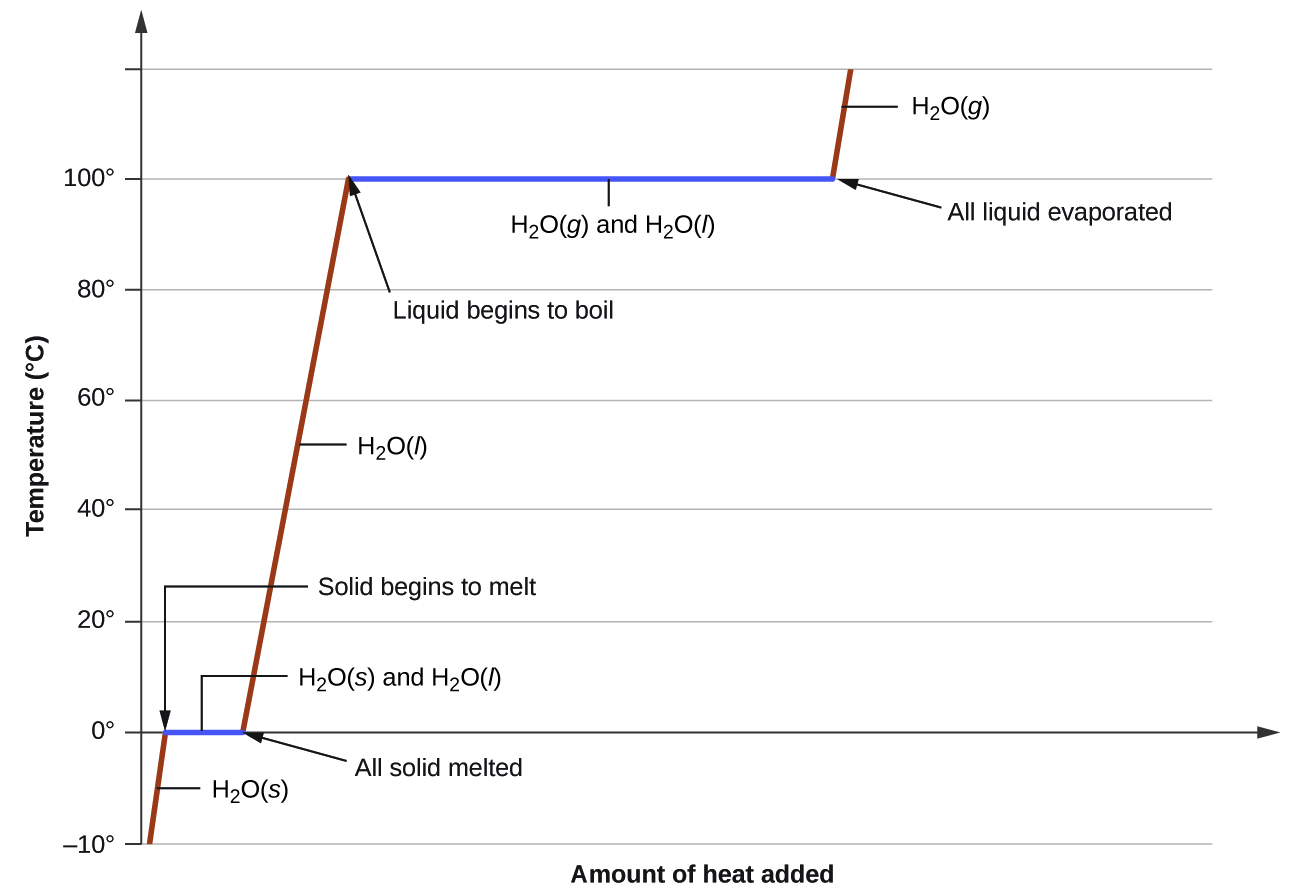
This heating-curve graph shows that temperature rises within a state but stays constant during a phase change. It is ideal for explaining why flat sections represent energy being used to change particle arrangement/attractions rather than increase average kinetic energy. This directly supports IB graph-interpretation questions. Source
Interpreting heating and cooling changes
· As a substance is heated within one state, temperature increases because average kinetic energy increases.
· At a melting point or boiling point, the temperature remains constant until the state change is complete.
· A flat section on a heating/cooling graph shows a change of state.
· Sloped sections show temperature change within one state.
· During cooling, the reverse applies: condensation and freezing occur at constant temperature.
· Be ready to interpret observable changes such as formation of bubbles, crystals, or condensation droplets.
Checklist: can you do this?
· Distinguish between an element, compound, and mixture using both definitions and particle diagrams.
· Choose and justify a suitable method for separating or purifying a mixture.
· Explain the properties of solids, liquids and gases using kinetic molecular theory.
· Name and identify all changes of state and use the correct state symbols in equations.
· Convert between °C and K and interpret heating/cooling graphs in terms of kinetic energy and changes of state.