Subatomic particles and the nuclear atom
· Atoms contain a small, dense, positively charged nucleus made of protons and neutrons (nucleons).
· Electrons occupy the space outside the nucleus.
· Know the relative charge and relative mass of each subatomic particle:
· Proton: +1 charge, relative mass 1
· Neutron: 0 charge, relative mass 1
· Electron: –1 charge, relative mass 1/1836 (usually treated as negligible mass)
· Almost all atomic mass comes from the nucleus because electron mass is negligible in IB calculations.
· A neutral atom has equal numbers of protons and electrons.
· An ion has gained or lost electrons, so number of electrons ≠ number of protons.
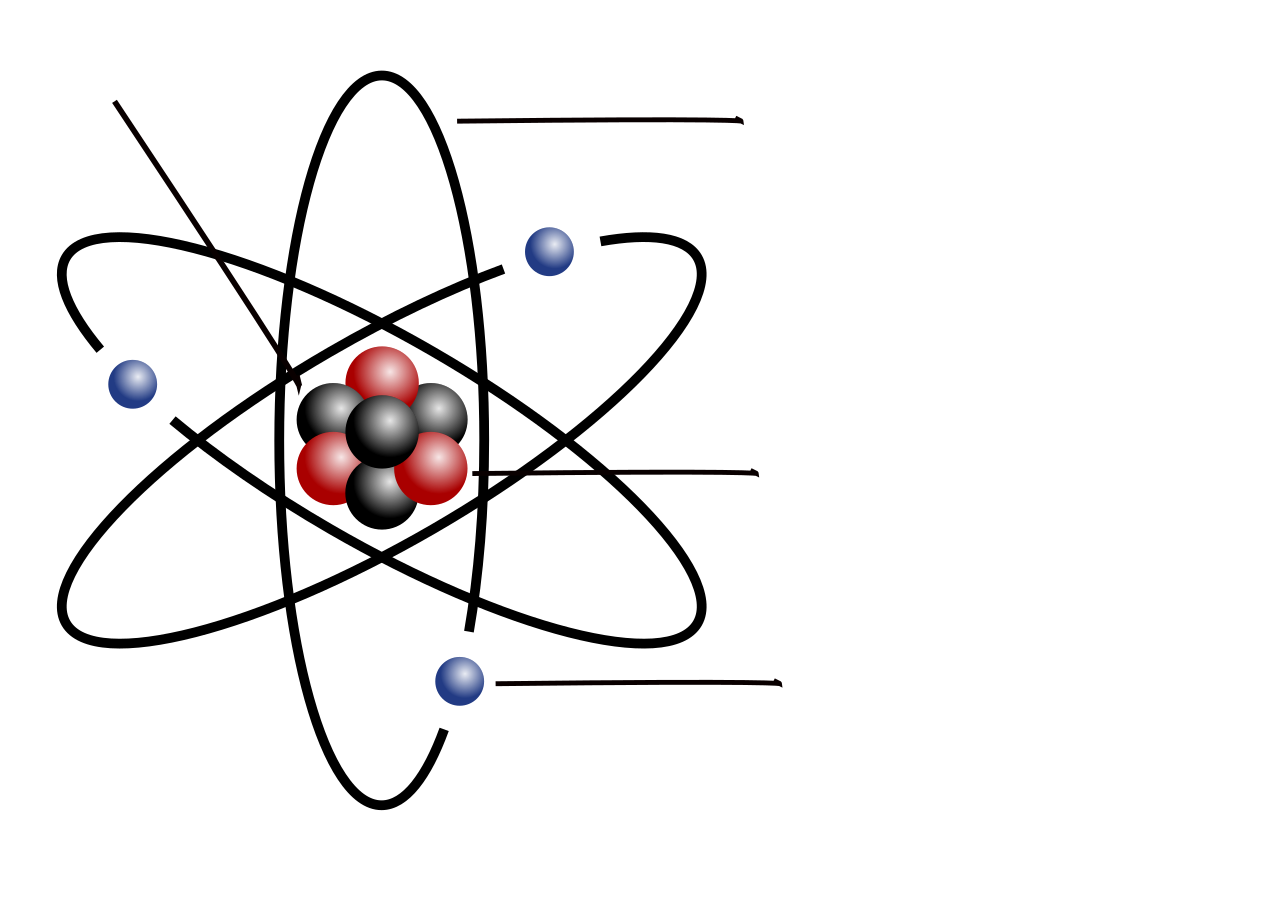
This diagram shows a simplified atom with a central nucleus and electrons outside it. Use it to reinforce that protons and neutrons are in the nucleus, while electrons occupy space around the nucleus. Source
Nuclear symbol and particle counting
· The nuclear symbol is written as .
· = atomic number = number of protons.
· = mass number = number of protons + number of neutrons.
· Number of neutrons = .
· Number of electrons:
· neutral atom: electrons =
· positive ion: electrons = charge
· negative ion: electrons = magnitude of charge
· Always identify the element from the atomic number, not the mass number.
· Exam trap: changing electrons makes an ion; changing neutrons makes an isotope.
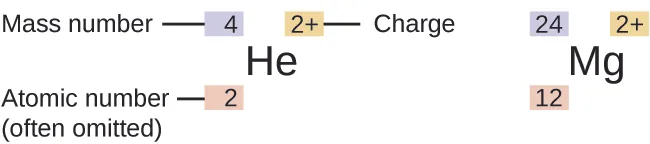
This figure labels the parts of a nuclear symbol: mass number, atomic number, element symbol, and ionic charge. It is ideal for practising how to count protons, neutrons, and electrons from notation. Source
Isotopes
· Isotopes are atoms of the same element with different numbers of neutrons.
· Isotopes have the same number of protons and therefore the same chemical identity.
· Isotopes have different mass numbers because their neutron numbers differ.
· Chemical properties of isotopes are very similar because chemical behaviour depends mainly on electron arrangement, not neutron number.
· Physical properties can differ because isotopes have different masses.
· You do not need to memorize specific isotope examples unless they are provided in a question.
Relative atomic mass and isotopic abundance
· The relative atomic mass, , in the periodic table is usually not a whole number because it is a weighted mean of the isotopes of an element.
· Weighted mean idea: more abundant isotopes contribute more to the final value.
· Core calculation:
·
· If abundances are given in %, divide by 100 or keep them over 100 total consistently.
· If only two isotopes are present, the abundances must add to 100%.
· Exam skill: rearrange weighted-average calculations to find unknown abundance or unknown isotopic mass.
Differences in physical properties of isotopes
· Isotopes can differ in mass-dependent physical properties.
· Heavier isotopes generally move slightly more slowly and may show small differences in diffusion or other physical behaviour.
· In IB questions, the key idea is simply: same chemical properties overall, but different physical properties because mass differs.
Checklist: can you do this?
· Deduce numbers of protons, neutrons, and electrons from for both atoms and ions.
· Explain what isotopes are and distinguish them from ions.
· Calculate relative atomic mass from isotopic masses and abundances.
· Calculate isotopic abundance from a non-integer relative atomic mass.
· State why isotopes have similar chemical properties but different physical properties.
HL only – Mass spectra and isotopic composition
· Mass spectra are used to determine the relative atomic mass of an element from its isotopic composition.
· In a mass spectrum, the x-axis is mass-to-charge ratio, and the y-axis is relative abundance.
· For IB Chemistry at this point, spectra are usually interpreted assuming ions have charge +1, so values are effectively isotopic masses.
· Each peak represents an isotope of the element.
· The height or area of a peak shows the isotope’s relative abundance.
· The weighted average of the isotopic masses using the abundances gives the relative atomic mass.
· A taller peak means that isotope is more abundant in the natural sample.
· You are expected to interpret spectra, not explain the operational details of the instrument.
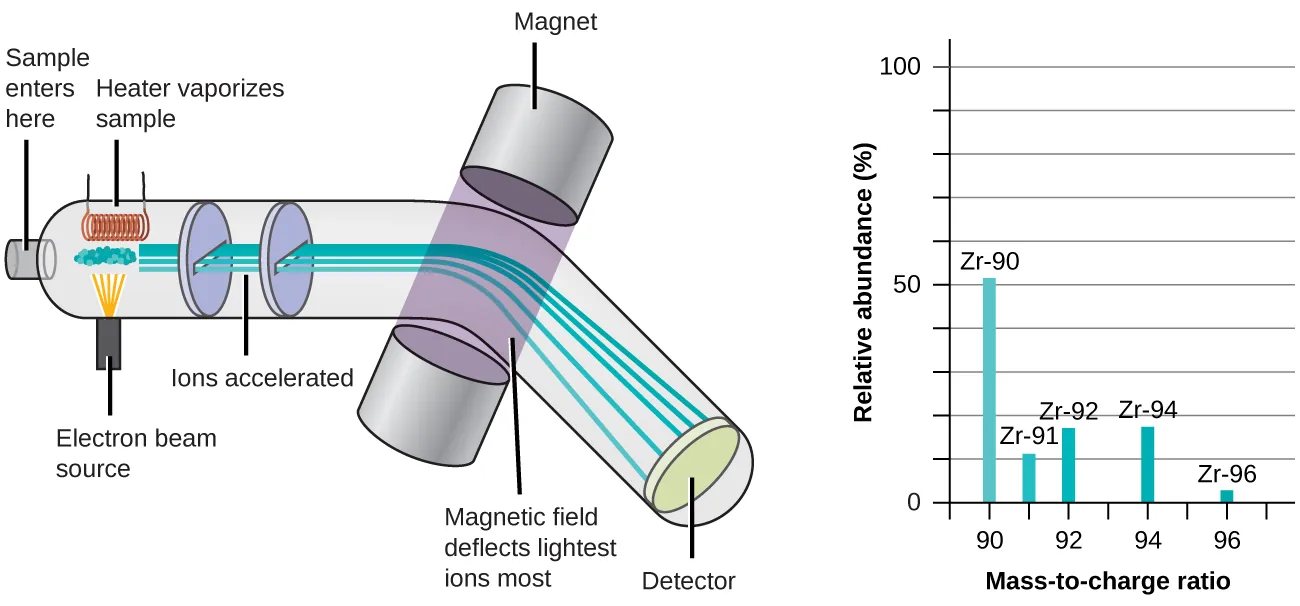
This figure shows how ions are separated by mass-to-charge ratio and how the resulting spectrum peaks represent different isotopes. Focus on reading the peak positions and relative abundances, since that is the exam skill. Source
Mass spectrum interpretation tips
· First identify the number of peaks → this usually tells you the number of isotopes.
· Read the value of each peak → this gives the isotopic mass (usually for ions).
· Read the relative abundances from the peak heights or values given.
· Use the weighted-average method to calculate relative atomic mass.
· Check whether the final answer is between the lightest and heaviest isotope masses.
· A relative atomic mass closer to one isotope means that isotope has the greater abundance.
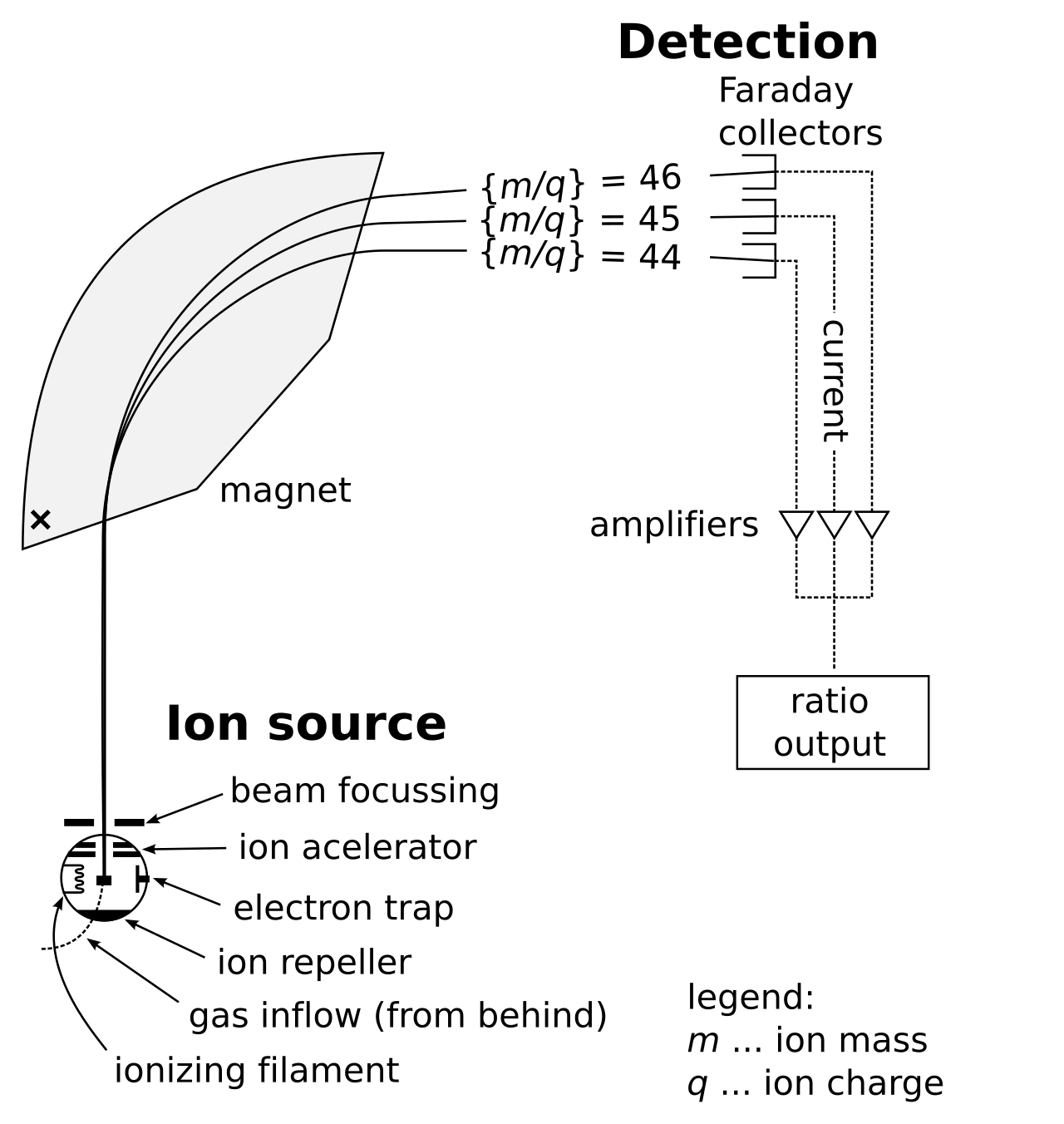
This schematic shows the main stages of a mass spectrometer, helping you visualize why ions separate before detection. For IB, use it only to support the big idea that different isotopes produce different peaks, not to memorize instrument mechanics. Source
Common exam traps
· Atomic number identifies the element; mass number does not.
· Neutrons = mass number – atomic number, not the other way around.
· For ions, only the electron number changes.
· Relative atomic mass is a weighted average, so it is often decimal/non-integer.
· In mass spectra, do not confuse peak height (abundance) with peak position ().
· Do not say isotopes have different chemical properties as the main rule; IB expects similar chemical properties but different physical properties.
Fast exam method
· Step 1: Read to get protons.
· Step 2: Calculate neutrons = .
· Step 3: Adjust for charge to get electrons.
· Step 4: For isotope questions, compare neutron number only.
· Step 5: For abundance questions, apply the weighted average carefully and check that total abundance = 100%.