The mole and Avogadro constant
The mole (mol) is the SI unit of amount of substance.
1 mol contains exactly elementary entities (Avogadro constant, ).
Elementary entities can be atoms, molecules, ions, electrons or a specified group of particles.
Convert between amount in moles and number of particles using:
In exam questions, always check what particle is being counted.
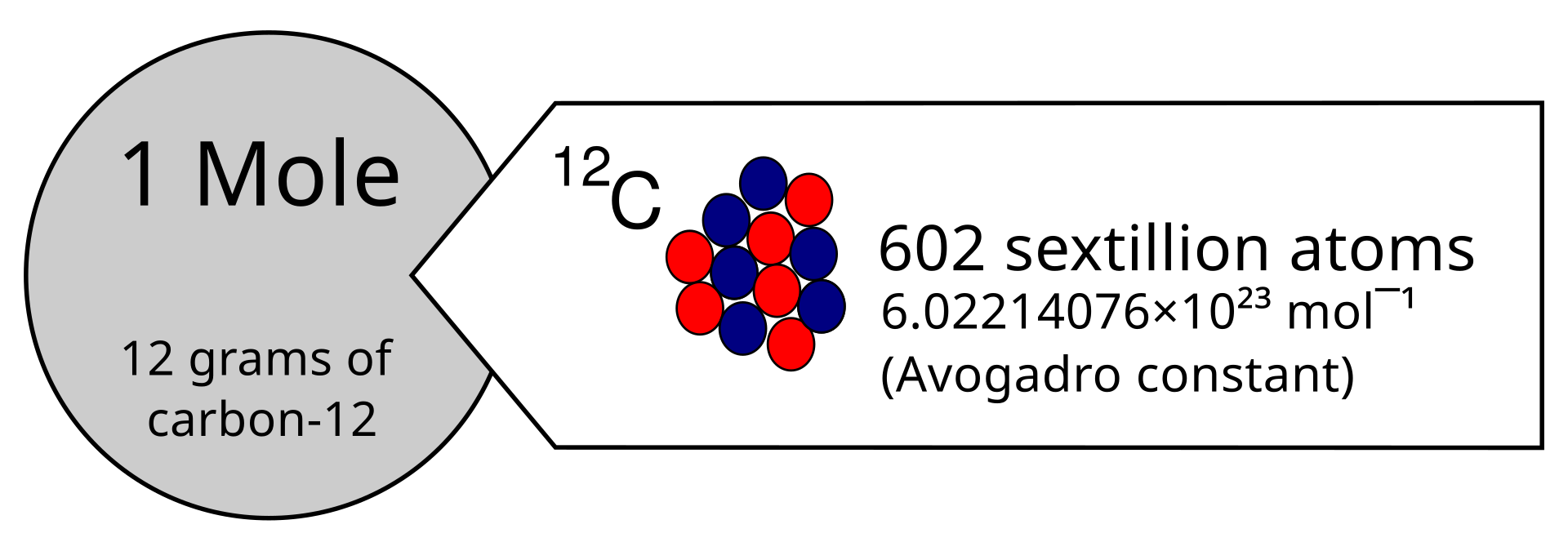
This diagram shows the definition of one mole using carbon-12, linking 12 g of carbon-12 to Avogadro’s number of atoms. It is useful for visualizing why the mole connects mass to particle number. Source
Relative atomic mass, relative formula mass and molar mass
Relative atomic mass, compares the average mass of an atom with of the mass of a carbon-12 atom.
Relative formula mass, = sum of all the values in a formula.
and have no units.
Molar mass, is the mass of 1 mol of a substance and has units g mol.
Numerically, is usually the same as , but has units and does not.
Use data booklet values to 2 decimal places in calculations.
Core mole calculations
Key relationship:
Rearrangements:
Typical calculation route:
mass moles particles
particles moles mass
Common exam trap: do not confuse number of moles with number of particles.
Always write units clearly: g, mol, g mol.
Empirical formula and molecular formula
Empirical formula = simplest whole-number ratio of atoms in a compound.
Molecular formula = actual number of each type of atom in a molecule.
To find an empirical formula from percentage composition:
Assume 100 g if percentages are given.
Convert each element’s mass to moles.
Divide all mole values by the smallest.
Convert to the simplest whole-number ratio.
If ratios are not whole numbers, multiply all by the same factor:
0.5
0.25
0.33
0.67
To find a molecular formula:
Find the empirical formula mass.
Calculate factor = .
Multiply all subscripts in the empirical formula by this whole-number factor.
Concentration of solutions
Molar concentration depends on amount of solute and volume of solution.
Main equation:
Rearrangements:
Square brackets show molar concentration, e.g. .
Required concentration units:
mol dm
g dm
Important conversion:
Remember: volume in concentration questions is usually required in dm, not cm.
Conversion: .
The image shows a volumetric flask, the key apparatus for preparing a solution of known volume and concentration. It is directly relevant to concentration calculations and standard-solution practical work. Source
Gas volumes and Avogadro’s law
Avogadro’s law: equal volumes of gases at the same temperature and pressure contain equal numbers of molecules.
For gases at the same and , volume ratios = mole ratios.
This allows mole calculations from gas volumes in reaction questions.
Always compare gases only when temperature and pressure are the same.
In stoichiometry, convert gas volumes to moles before using the balanced equation unless a direct volume ratio can be used.
This is helpful for seeing that gas volume is directly linked to amount of substance when temperature and pressure are fixed. It supports the IB idea that gas volume ratios can be used like mole ratios. Source
Problem-solving strategy for mole questions
Start by identifying the data type given: mass, particles, concentration/volume, gas volume, or percentage composition.
Convert the given quantity into moles first whenever possible.
Use the balanced equation only after you have found moles of the known substance.
Convert final moles into the required form: mass, particles, volume, or concentration.
Check that the answer has the correct units and sensible significant figures.
Checklist: can you do this?
Convert between mass, moles and number of particles.
Calculate and molar mass correctly, including units.
Find an empirical formula from percentage composition or experimental mass data.
Determine a molecular formula from empirical formula + molar mass.
Solve concentration and gas-volume stoichiometry problems using and Avogadro’s law.
Exam traps and quick reminders
and have no units; molar mass does.
Moles are not particles: use when converting.
For solutions, convert cm to dm before using .
In empirical formula questions, convert mass to moles, not directly to ratios.
A molecular formula must be a whole-number multiple of the empirical formula.
For gas-volume ratios, only compare gases under the same temperature and pressure.