Ideal gas model: core assumptions
Ideal gas particles are treated as point particles with negligible volume.
There are no intermolecular forces between particles in an ideal gas.
Particles move in constant random motion.
All collisions are perfectly elastic: no kinetic energy is lost overall during collisions.
The model is used to predict gas behaviour, even though no real gas is perfectly ideal.
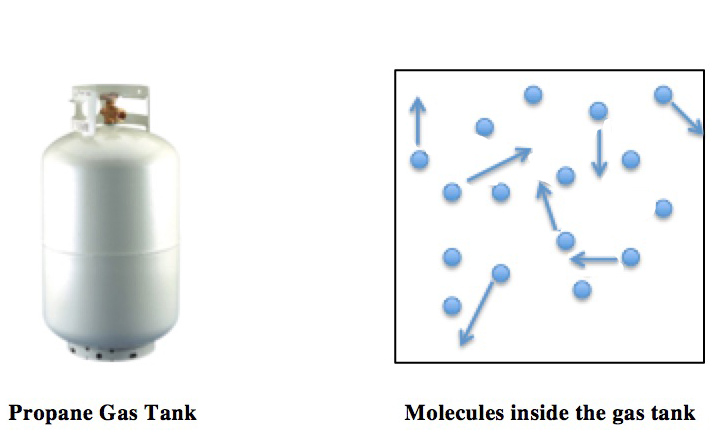
Educational caption: This diagram shows gas particles moving randomly in a container and colliding with the walls. It helps explain how pressure arises from particle-wall collisions and why higher temperature increases collision energy and pressure. Source
Why real gases deviate from ideal behaviour
Real gases deviate because actual particles do have volume and do experience intermolecular attractions.
Deviation is greatest at low temperature because particles move more slowly, so attractive forces matter more.
Deviation is greatest at high pressure because particles are forced closer together, so particle volume is no longer negligible.
In exams, explain non-ideal behaviour using the two key ideas: intermolecular forces and finite particle volume.
The ideal gas model works best at high temperature and low pressure.
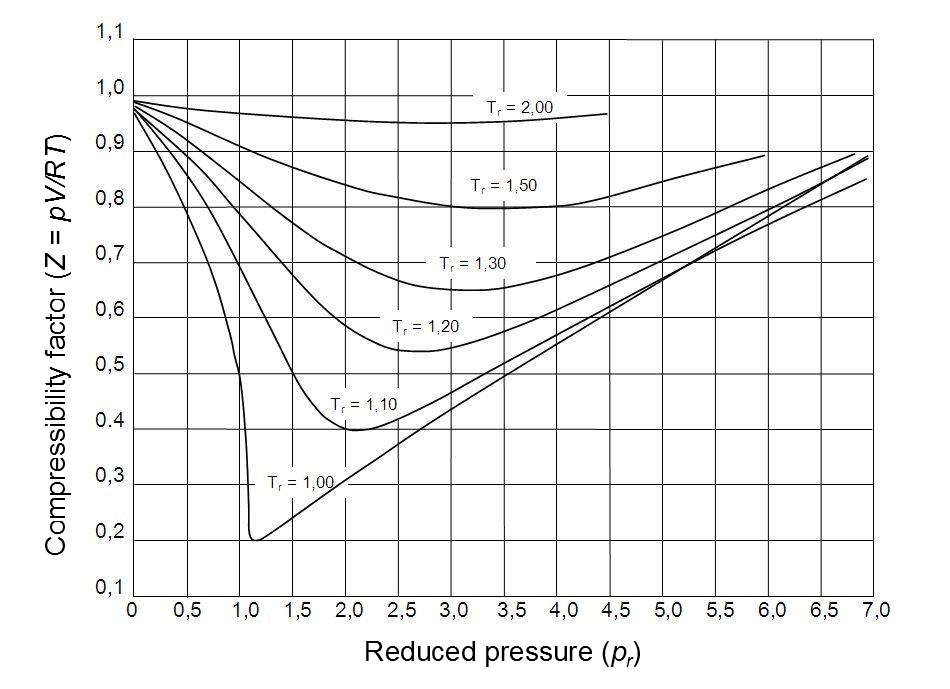
Educational caption: This graph shows how real gases deviate from ideal behaviour under different conditions. It is useful for seeing that gases behave most ideally when pressure is low and deviations become larger as conditions become more extreme. Source
Molar volume of an ideal gas
At a given temperature and pressure, the molar volume of an ideal gas is constant.
This means 1 mol of any ideal gas occupies the same volume under the same conditions.
At STP, use the molar volume value given in the IB data booklet.
This idea links directly to Avogadro’s law: equal volumes of gases at the same temperature and pressure contain equal numbers of molecules.
Typical exam use: convert between gas volume and amount of substance, .
Relationships between pressure, volume, temperature and amount
For a fixed mass of gas, changing one variable changes the others in a predictable way.
Pressure increases when volume decreases if temperature is constant.
Volume increases when temperature increases if pressure is constant.
For a fixed amount of gas, temperature in gas equations must always be in Kelvin (K).
The syllabus expects you to analyse graphs showing relationships between , and .
The names of individual gas laws are not assessed, but the relationships are.
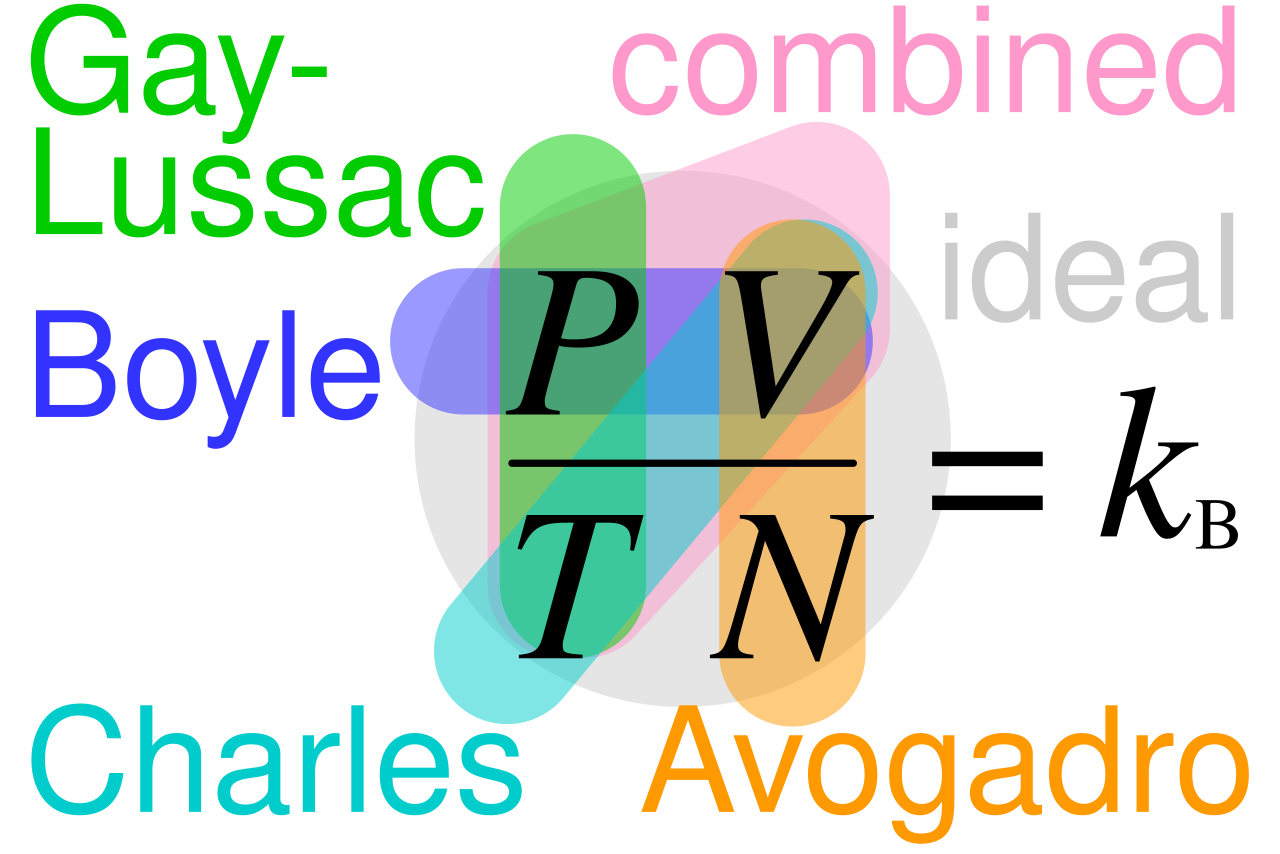
Educational caption: This diagram summarizes how pressure, volume, temperature and amount of gas are linked. It is especially helpful for revising which variable is held constant in common gas-law relationships and how they combine into the ideal gas law. Source
Graph skills for IB exams
Be able to interpret how , and change for a fixed mass of gas.
A vs graph at constant temperature shows an inverse relationship.
A vs graph at constant pressure shows a direct relationship when is in Kelvin.
A vs graph at constant volume shows a direct relationship when is in Kelvin.
Always check whether the graph is a sketch or accurately plotted data.
In written explanations, link graph shape to particle motion, collision frequency and collision force.
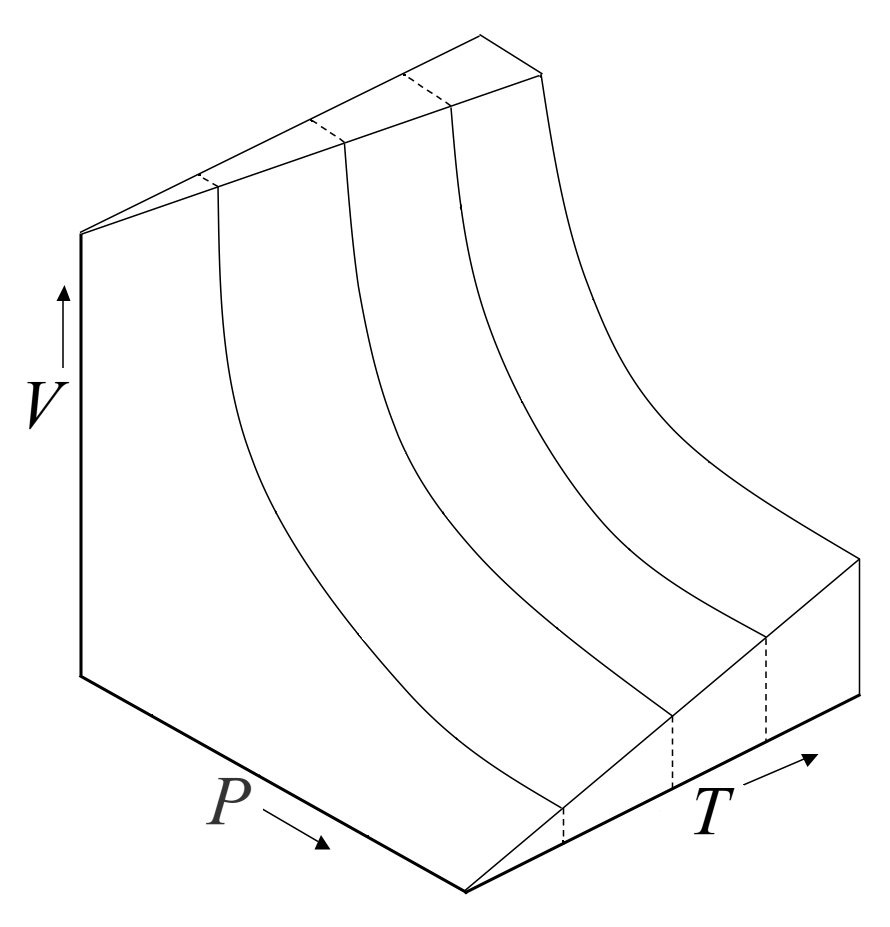
Educational caption: This diagram shows several isotherms for an ideal gas, helping you visualize how pressure and volume are related at different constant temperatures. Higher-temperature curves correspond to gas particles with greater average kinetic energy. Source
Essential equations and unit rules
Ideal gas equation:
Combined gas law:
Use SI units only for pressure and volume in ideal gas calculations, as required by the syllabus.
Temperature must be in Kelvin, never in °C, in gas-law calculations.
Rearranging equations accurately is a common exam skill: solve for , , or .
The values of and the molar volume at STP are provided in the IB data booklet.
Problem-solving moves that score marks
Identify which variables are constant and which change.
Choose the right model:
when you have a single gas state or need moles.
Combined gas law when amount of gas stays constant between two states.
Convert all data before substituting: especially °C to K and non-SI units to SI units.
Show substitution clearly and keep track of units.
Check whether your answer is physically sensible: for example, increasing temperature at constant pressure should increase volume.
Practical / data-based applications
The ideal gas law can be used to determine molar mass of a gas from experimental data.
In practical questions, gas data may come from measured pressure, temperature and volume.
You may need to combine gas calculations with mole calculations from earlier topics.
State assumptions clearly if the gas is treated as ideal.
Be ready to comment on why experimental values differ from theoretical values: gas loss, measurement error, or non-ideal behaviour.
Common exam traps
Using °C instead of K.
Using the combined gas law when changes.
Forgetting that real gases deviate most at low and high .
Not converting pressure and volume into the required units.
Giving a correct number with the wrong unit.
Checklist: can you do this?
State the assumptions of the ideal gas model.
Explain why real gases deviate from ideal behaviour at low temperature and high pressure.
Interpret -, - and - graphs for a fixed mass of gas.
Solve numerical problems using and the combined gas law.
Use the molar volume at STP from the IB data booklet in calculations.