Formation of ions
Metals lose electrons to form positive ions (cations).
Non-metals gain electrons to form negative ions (anions).
Ion formation is explained using electron configuration: atoms tend to achieve a full outer shell.
Be able to predict ion charge from group / valence electrons for main-group elements.
Transition elements can form more than one ion, so learn to accept variable charges (for example, Fe and Fe).
In exam questions, link ion charge to the atom becoming more stable after losing or gaining electrons.
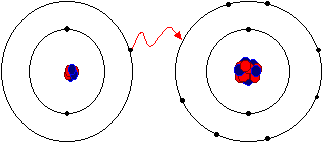
This diagram shows how an electron is transferred from a metal atom to a non-metal atom to form a cation and anion. It is useful for visualizing why ions end up with full outer shells. Use it to practise explaining ion formation in words and with electron-shell diagrams. Source
Ionic bonding
An ionic bond is the electrostatic attraction between oppositely charged ions.
Ionic bonding is non-directional: each ion attracts ions of opposite charge in all directions.
Ionic compounds usually form between a metal and a non-metal.
Ionic bonding is strong because there are many electrostatic attractions throughout the structure.
Stronger ionic attraction occurs when ions have:
higher charges
smaller ionic radii
This is why compounds such as MgO generally have stronger ionic bonding than NaCl.
This image shows that ionic bonding is not a single shared pair of electrons, but a network of attractions between oppositely charged ions. It helps explain why ionic compounds form giant lattices rather than small molecules. Use it when revising the definition of an ionic bond. Source
Formulas and naming of ionic compounds
The formula of an ionic compound must give an overall neutral charge.
Write formulas by balancing the total positive charge and total negative charge.
The formula of an ionic compound is an empirical formula: the simplest whole-number ratio of ions.
Binary ionic compounds are named:
cation first
anion second with the suffix “-ide”
Examples:
NaCl = sodium chloride
MgO = magnesium oxide
CaBr = calcium bromide
AlO = aluminium oxide
You must be able to interconvert names and formulas.
Polyatomic ions you must know
Ammonium = NH
Hydroxide = OH
Nitrate = NO
Hydrogencarbonate = HCO
Carbonate = CO
Sulfate = SO
Phosphate = PO
When writing formulas, treat each polyatomic ion as a single charged unit.
Use brackets when more than one polyatomic ion is needed, for example Ca(OH) and (NH)SO.
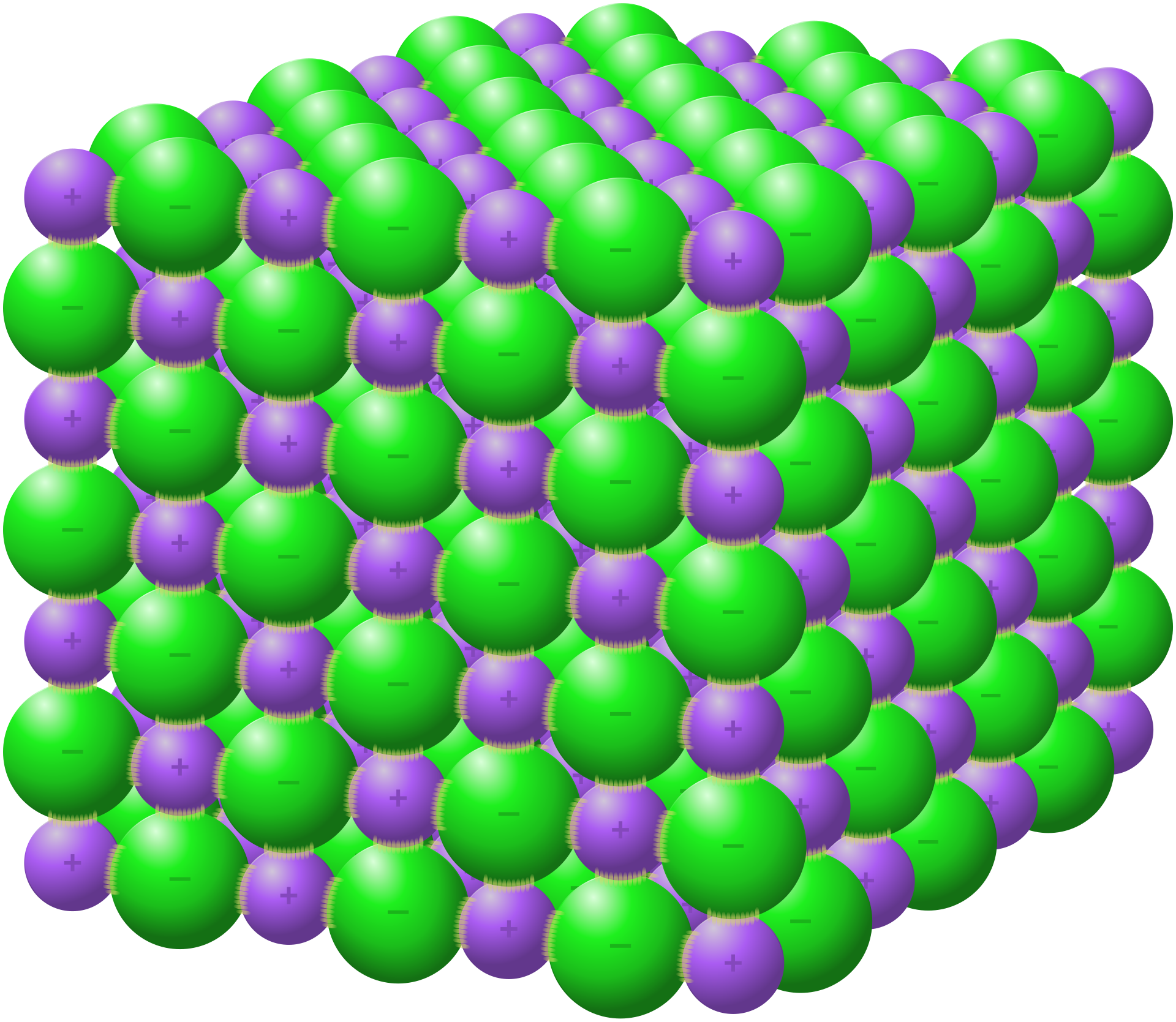
Giant ionic lattice
Ionic compounds exist as giant three-dimensional lattice structures.
The lattice contains a regular repeating arrangement of cations and anions.
There are no separate molecules in an ionic solid.
This is why ionic substances are represented by an empirical formula, not a molecular formula.
In markschemes, phrases such as “giant ionic lattice”, “3D arrangement”, and “strong electrostatic attractions between oppositely charged ions” score well.

This diagram shows the repeating 3D arrangement of ions inside an ionic crystal. It is especially useful for linking structure to high melting point and empirical formula. Use it when describing why ionic compounds are not made of discrete molecules. Source
Physical properties of ionic compounds
Ionic compounds have high melting points and high boiling points because a lot of energy is needed to overcome strong electrostatic attractions in the lattice.
They are usually low in volatility because the ions are held strongly in place.
Solid ionic compounds do not conduct electricity because the ions are fixed in position and cannot move.
Molten ionic compounds and aqueous solutions do conduct electricity because the ions are free to move and carry charge.
Many ionic compounds are soluble in water because water molecules can attract and separate ions from the lattice.
Ionic compounds are often insoluble in non-polar solvents.
They are often hard but brittle: when layers shift, like charges line up, causing repulsion and the crystal to crack.
This diagram explains why ionic solids are brittle rather than malleable. When stress shifts ion layers, ions of the same charge repel strongly, causing the crystal to break. This is a common structure–property explanation in exams. Source
Electrical conductivity: exam wording
Solid ionic compounds: do not conduct because ions are not mobile.
Molten ionic compounds: conduct because ions are free to move.
Aqueous ionic solutions: conduct because dissolved ions are mobile charge carriers.
Do not say electrons move through an ionic solid; in IB questions, conduction is due to moving ions, not electrons.
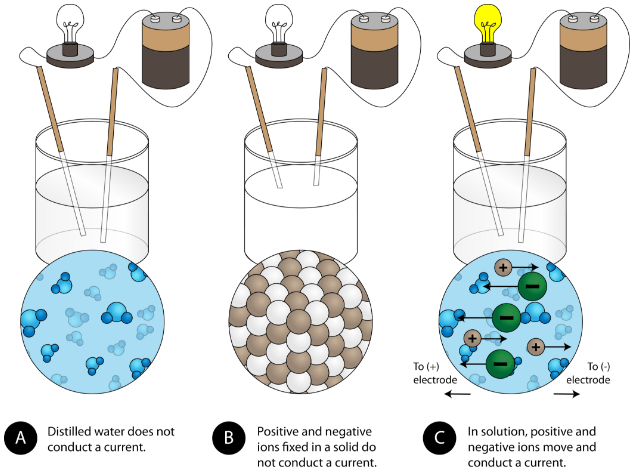
This source supports the key idea that ionic compounds conduct only when ions can move. It is useful for comparing solid, molten, and aqueous ionic substances. Use it to refine your explanations of conductivity and solubility. Source
Lattice enthalpy and bond strength
Lattice enthalpy is a measure of the strength of ionic bonding in a compound.
The more negative the lattice enthalpy, the stronger the attractions between ions.
Lattice enthalpy becomes larger in magnitude when:
ions have greater charges
ions have smaller radii
Therefore, compounds with small, highly charged ions have especially strong ionic bonding.
This helps explain trends in melting point and stability.
Typical comparison: MgO has a much stronger ionic lattice than NaCl because Mg and O have higher charges.
Common exam traps
Do not describe ionic compounds as made of molecules.
Do not say ionic bonding involves sharing electrons.
Do not forget that formulas must be charge-balanced.
Do not assume all metals form only one ion; transition elements often show variable charge.
For conductivity, always mention mobility of ions.
For melting point / boiling point, always mention strong electrostatic attractions in the lattice.
Checklist: can you do this?
Predict the charge of an ion from an atom’s electron configuration or position in the periodic table.
Write formulas and names for binary ionic compounds and compounds containing polyatomic ions.
Explain conductivity, solubility, brittleness, volatility, melting point and boiling point using the giant ionic lattice model.
Compare ionic bond strength using ion charge and ionic radius.
State why ionic compounds are represented by empirical formulas, not molecular formulas.