Core idea: metallic bonding
· Metallic bonding = electrostatic attraction between a lattice of positive metal ions (cations) and delocalized electrons.
· Metals form a giant lattice: the positive ions are in fixed positions, while the valence electrons are free to move throughout the structure.
· This is why metallic bonding is often described as a “sea of delocalized electrons” around metal cations.
· In exams, always link structure → bonding → property.
· Key wording: non-directional bonding, mobile electrons, strong electrostatic attraction.
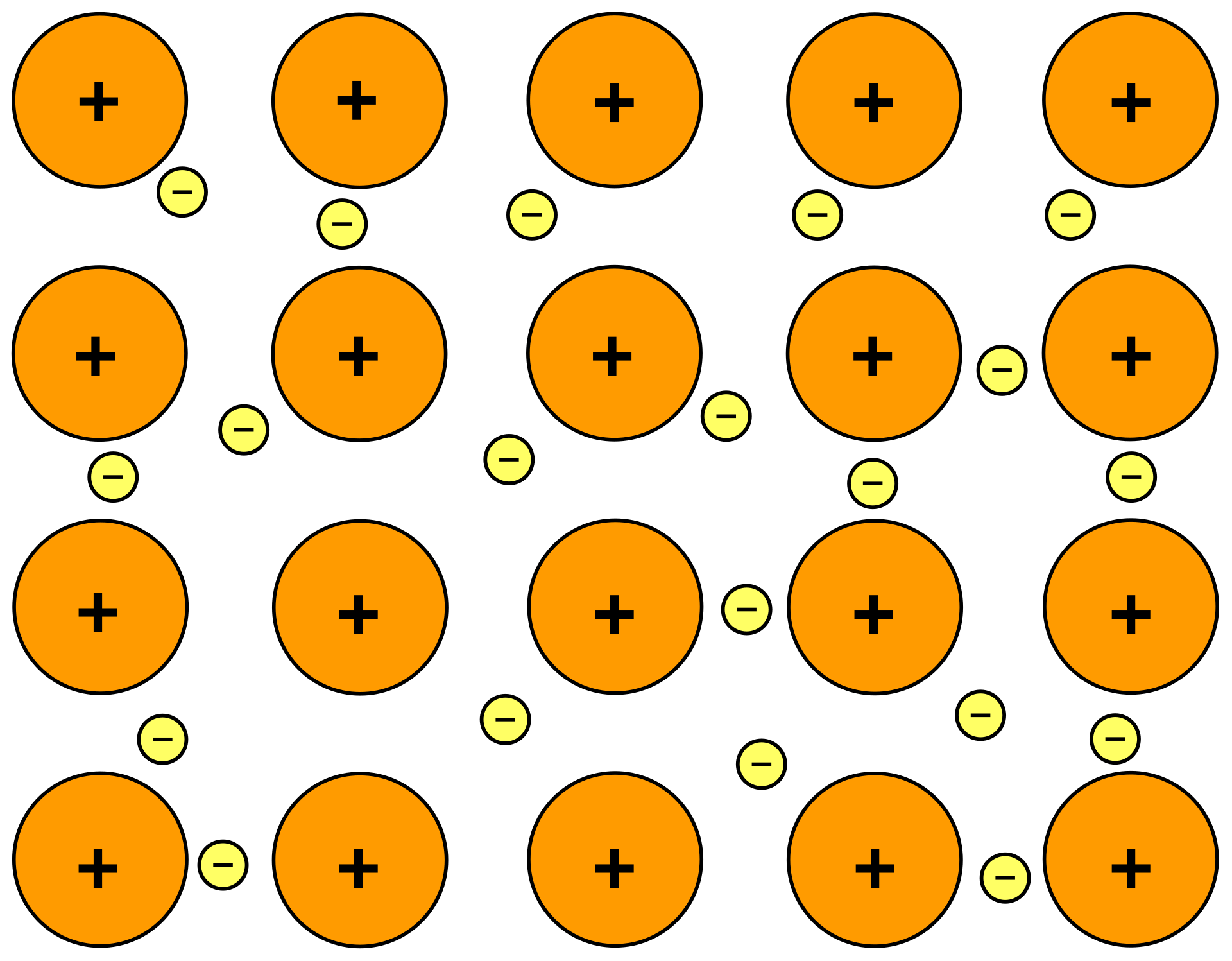
This diagram shows a regular lattice of metal cations surrounded by delocalized electrons. It is ideal for explaining why metals conduct electricity and why metallic bonding is non-directional. Use it to label cations, delocalized electrons, and electrostatic attraction. Source
Why metals conduct electricity and heat
· Electrical conductivity: metals conduct because delocalized electrons can move and carry charge through the lattice.
· In a question about solid metals, remember: unlike ionic compounds, metals conduct in the solid state because their charge carriers are already mobile.
· Thermal conductivity: the mobile electrons transfer kinetic energy rapidly through the structure.
· The closely packed lattice also helps energy pass from particle to particle efficiently.
· Exam link: free-moving electrons → good conductor of electricity and heat.
Why metals are malleable
· Malleability = metals can be hammered into shape without shattering.
· Metallic bonding is non-directional, so when layers of ions slide past one another, the attraction to the delocalized electrons remains.
· Because the bonding is not tied to one fixed pair of atoms, the structure can change shape without the whole lattice breaking apart.
· This contrasts with ionic lattices, where shifting layers can bring like charges together and cause repulsion.
· Application to uses: metals are useful for wires, sheets, car bodies, tools, and structural materials because they are conductive and/or malleable.
Strength of metallic bonding
· The strength of metallic bonding depends mainly on:
· 1. Charge of the metal ions: higher positive charge means stronger attraction to delocalized electrons.
· 2. Radius of the metal ion: smaller ions attract delocalized electrons more strongly because the electrons are closer to the positive nuclei.
· A stronger metallic bond usually gives a higher melting point.
· Simple exam idea: higher charge + smaller ion = stronger metallic bond = higher melting point.
Trends in melting points of s- and p-block metals
· Across metals in the same period, melting point often increases when:
· more electrons are delocalized per atom
· cation charge increases
· metal ions become smaller
· Example trend often used: Na < Mg < Al in melting point.
· Reason: from Na to Mg to Al, the ions have higher charge, contribute more delocalized electrons, and get smaller, so metallic bonding becomes stronger.
· For IB, keep the treatment simple: focus on cation charge, electron density, and ion size/radius.
· Do not overcomplicate with advanced lattice details unless specifically asked.
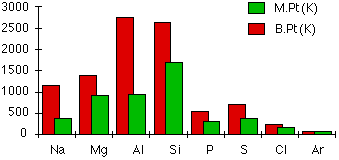
This resource supports the trend Na < Mg < Al and links it to stronger metallic bonding across Period 3 metals. It is useful for showing that increasing charge density and number of delocalized electrons strengthen the metallic bond. Source
Relating metallic properties to uses
· Electrical conductivity → used in electrical wiring and circuits.
· Thermal conductivity → used in cookware, heat exchangers, and radiators.
· Malleability → used to make metal sheets and shaped components.
· High melting point and strength → used in high-temperature or load-bearing applications.
· In exam answers, always connect the specific property to the specific use with a reason.
HL only: transition elements and delocalized d-electrons
· Transition elements have delocalized d-electrons as well as delocalized outer electrons.
· This can make metallic bonding especially strong.
· As a result, transition elements often have high melting points and good electrical conductivity.
· Their melting-point trend across the d-block is less simple / less regular than across the s-block because the number of delocalized electrons and bonding interactions changes in a more complex way.
· If asked why transition metals often have higher melting points than Group 1 metals, mention more delocalized electrons and stronger attraction between cations and electron cloud.
This source explains that transition metals can involve d-electrons in delocalization, helping explain their high melting points and good conductivity. It is useful for the Additional HL content in this subtopic. Source
Alloys link
· Alloys are mixtures of a metal with other metals or non-metals.
· They still have metallic bonding, but the atoms are different sizes, so the layers cannot slide as easily.
· This often makes alloys harder and less easily deformed than pure metals.
· Key exam phrase: distortion of layers reduces slipping.
· This links directly to the next topic on materials and alloys.
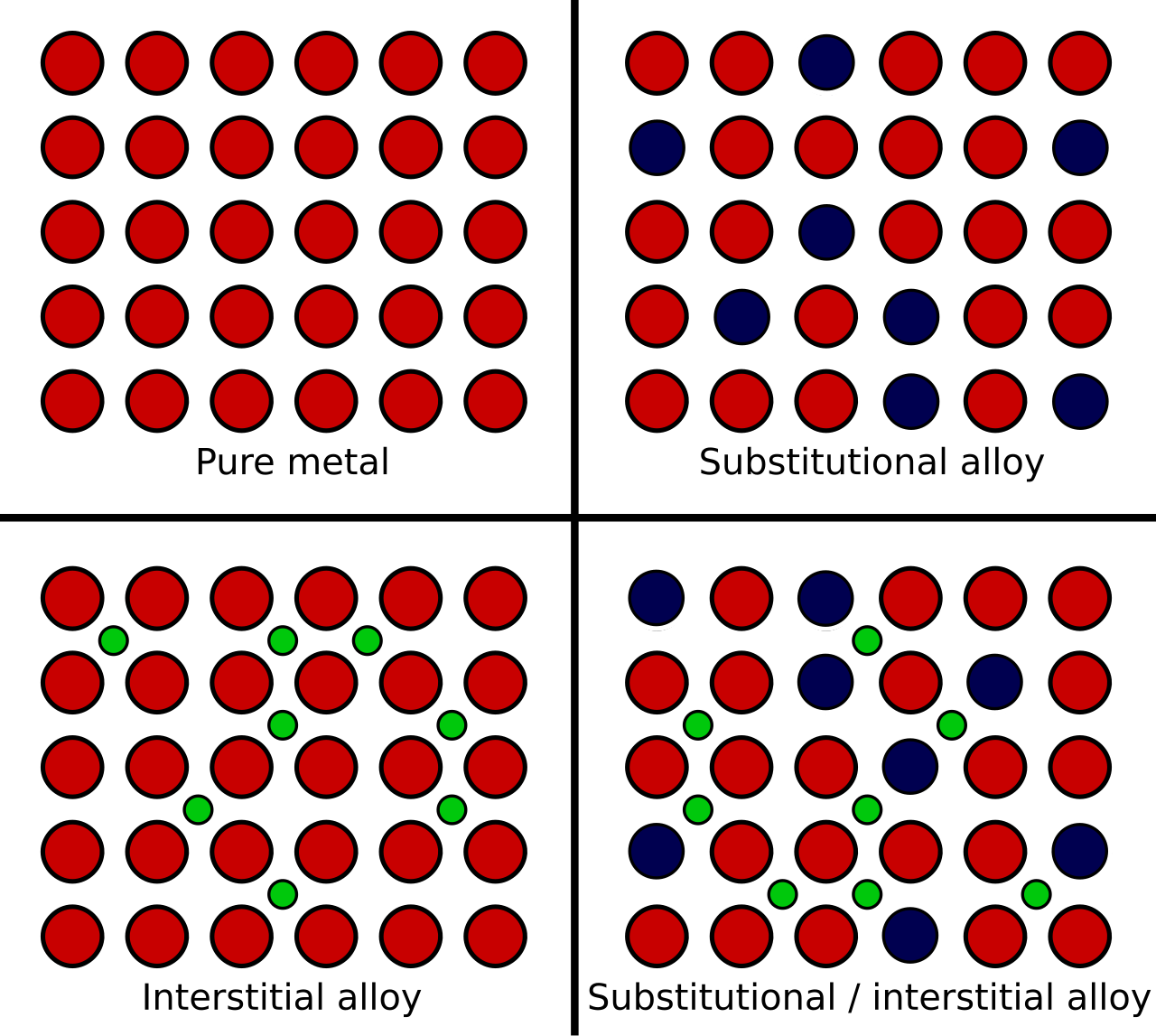
This diagram shows pure metal, substitutional alloy, and interstitial alloy arrangements. It helps explain why different-sized atoms disrupt layer movement, making alloys generally harder than pure metals. Source
Checklist: can you do this?
· Define metallic bonding using the phrase electrostatic attraction between cations and delocalized electrons.
· Explain electrical conductivity, thermal conductivity, and malleability using the metallic model.
· Explain melting-point trends in s- and p-block metals using ion charge, ion radius, and electron density.
· Apply the model to uses of metals by linking property → bonding/structure → use.
· HL: explain why transition metals often have high melting points and good conductivity using delocalized d-electrons.
Common exam mistakes to avoid
· Do not say metals contain molecules. Metals have a giant metallic lattice.
· Do not describe metallic bonding as sharing electrons between two specific atoms; the electrons are delocalized.
· Do not explain conductivity by saying ions move in solid metals; in metals, it is the electrons that move.
· Do not forget to compare charge and radius when explaining stronger metallic bonding.
· For HL, do not just say transition metals are strong because they are “transition metals” — mention delocalized d-electrons.