Bonding continuum: the big idea
Bonding is a continuum, not three completely separate categories.
Real substances often show a mixture of ionic, covalent and metallic character.
The bonding triangle is used to place substances between these three extremes.
In exam answers, always link bonding + structure + properties.
Key properties to predict: melting point, electrical conductivity, solubility, hardness / brittleness / malleability.
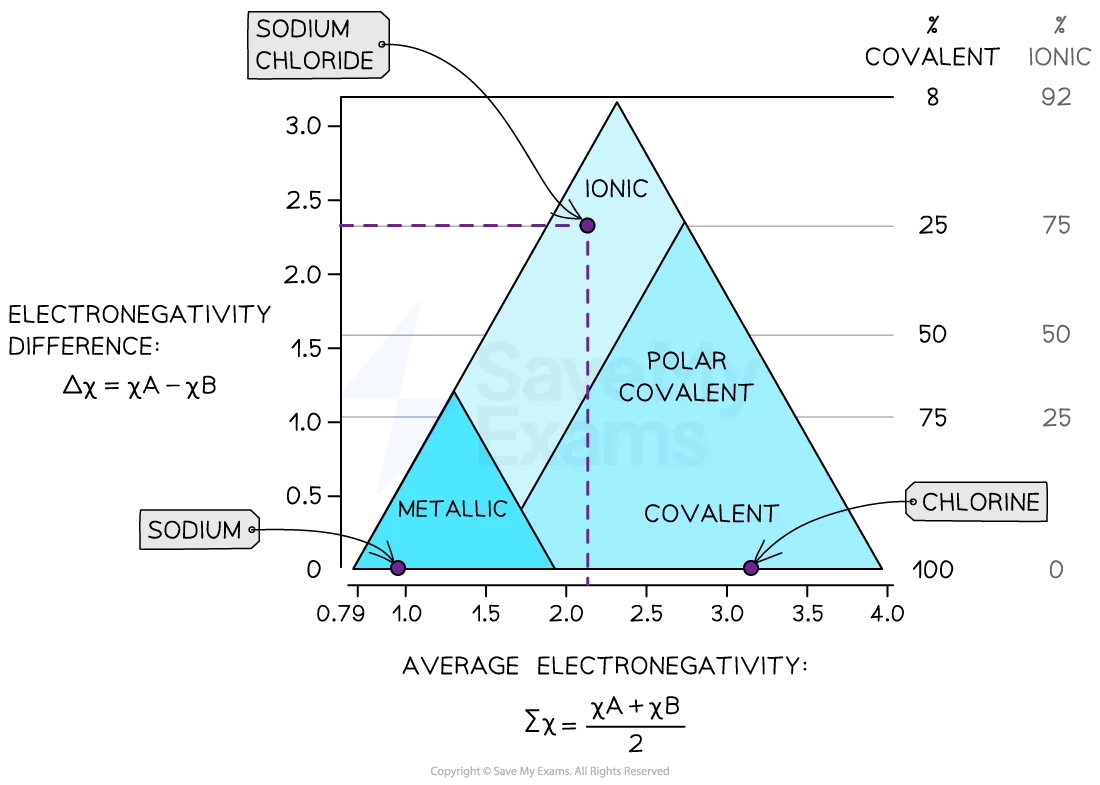
This image shows how a substance is placed on the bonding triangle using average electronegativity and electronegativity difference. It helps explain why real materials can have mixed bonding character rather than being purely ionic, covalent or metallic. A useful visual for predicting properties from position in the triangle. Source
Using the bonding triangle
A compound’s position depends on the relative contributions of ionic, covalent and metallic bonding.
Use electronegativity data from the data booklet to estimate where a binary compound lies.
Large electronegativity difference ⟶ more ionic character.
Small electronegativity difference between non-metals ⟶ more covalent character.
Low electronegativity values and small difference between metals ⟶ more metallic character.
You may be asked to determine position and then predict properties.
Percentage ionic character calculations are not required.
Predicting properties from position
Near ionic region: usually high melting point, brittle, often soluble in water, conducts when molten / aqueous, does not conduct when solid.
Near covalent molecular region: usually low melting / boiling point, often volatile, poor electrical conductivity.
Near giant covalent / network region: very high melting point, usually insoluble, usually non-conducting except forms like graphite / graphene.
Near metallic region: good electrical conductivity, good thermal conductivity, malleable, often high melting point.
Borderline substances can show intermediate properties, so avoid over-simplified statements.
Alloys
Alloys are mixtures of a metal with other metals or non-metals.
They have enhanced properties compared with the pure metal.
Metallic bonding is non-directional, so layers can still move, but less easily than in a pure metal.
Different-sized atoms distort the lattice and make it harder for layers to slide.
This usually makes alloys harder, stronger, and sometimes more corrosion resistant.
Common examples named in the syllabus: bronze, brass, stainless steel.
Exam wording: explain alloy properties using delocalized electrons, positive metal ions, and distortion of layers.
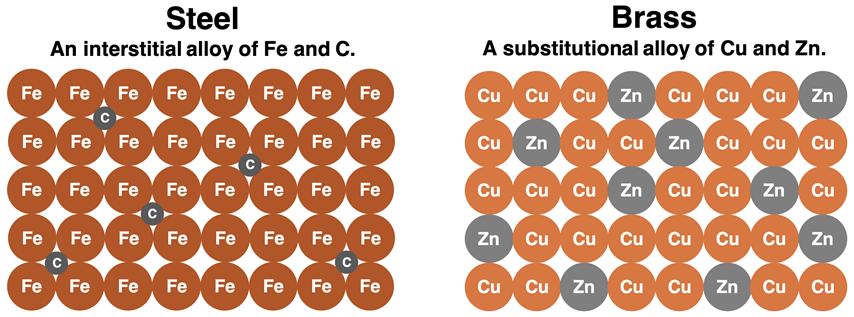
These diagrams show substitutional and interstitial alloys. They help explain why adding different atoms disrupts regular metal layers, making an alloy harder and stronger than the pure metal. This directly supports IB explanations of enhanced alloy properties. Source
Polymers and plastics
Polymers are macromolecules made from repeating monomers.
Plastics are common polymeric materials.
Typical plastic properties depend on structure: often low density, durable, chemically resistant, poor electrical conductors, and can be moulded easily.
Many plastics are useful because strong covalent bonds run along the polymer chains.
The forces between chains are weaker than the covalent bonds within chains, helping explain flexibility and melting behaviour.
Be prepared to discuss both natural polymers and synthetic polymers.
Addition polymers
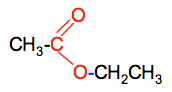
Addition polymerization occurs when monomers with a C=C double bond react.
The double bond breaks and monomers join to form a long chain.
Only one product forms, so atom economy is 100%.
You must be able to draw / identify the repeating unit from a given monomer.
The repeating unit is shown inside square brackets with continuation bonds through the brackets.
The monomer for an addition polymer is usually an alkene.
In reverse questions, identify the monomer by restoring the double bond from the repeating unit.

This diagram shows addition polymerization of ethene into poly(ethene). It is a clear model for the IB idea that the C=C bond opens and forms a chain of repeating units. Use it to practise converting between monomer and polymer repeating unit. Source
Checklist: drawing addition polymer repeating units
Identify the C=C double bond in the monomer.
Change the double bond to a single bond in the polymer backbone.
Keep side groups attached to the same carbon atoms.
Put the repeating unit in square brackets.
Add continuation bonds through the brackets.
HL only: condensation polymers
Condensation polymerization happens when monomers with two functional groups react.
A small molecule is eliminated, commonly or HCl.
You must be able to represent the repeating units of polyamides and polyesters.
Polyamides contain amide links: –CONH–.
Polyesters contain ester links: –COO–.
Biological macromolecules also form by condensation reactions and break down by hydrolysis.
In exams, identify the monomers by locating the linkage group in the repeating unit.
Typical monomer pairs:
Dicarboxylic acid + diol ⟶ polyester
Dicarboxylic acid + diamine ⟶ polyamide
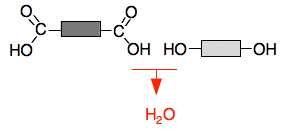
These diagrams show how condensation polymers form from monomers with two functional groups, with loss of a small molecule. They are especially useful for spotting ester links and amide links in repeating units. Use them to practise identifying whether a polymer is a polyester or polyamide. Source
Exam patterns and common traps
Do not say a substance is purely ionic / purely covalent / purely metallic unless the question clearly expects an idealized model.
For bonding triangle questions, use electronegativity values and then link to property predictions.
For alloys, do not call them compounds; they are mixtures.
For polymers, distinguish clearly between monomer, polymer, and repeating unit.
For addition polymers, no small molecule is lost.
For condensation polymers, a small molecule is released.
If asked why alkenes are useful in industry, link to their ability to undergo addition reactions and form polymers.
Checklist: can you do this?
Use electronegativity data to place a binary compound on the bonding triangle.
Predict material properties from bonding type and structure.
Explain why alloys are harder / stronger than pure metals.
Draw the repeating unit of an addition polymer from a monomer.
Identify polyamide vs polyester and the small molecule eliminated in HL questions.