AP Syllabus focus:
‘Movement of ions through channels can polarize membranes, creating electrochemical gradients important for cell signaling and homeostasis.’
Cells use ion channels to shape electrical conditions across the plasma membrane. By controlling ion movement, membranes become polarized, storing potential energy as electrochemical gradients that support signalling and stable internal conditions.
How ion movement creates membrane polarization
Ions are charged, so their movement across the membrane changes both solute distribution and voltage. When channel proteins open, specific ions diffuse down their existing driving forces, which can make one side of the membrane more positive or more negative.
Membrane polarization: The development of an unequal distribution of electrical charge across a membrane, producing a voltage difference between the cytosol and the extracellular fluid.
Because the plasma membrane’s hydrophobic core limits free ion passage, charge imbalances can persist and be precisely adjusted by regulated channel opening and closing.
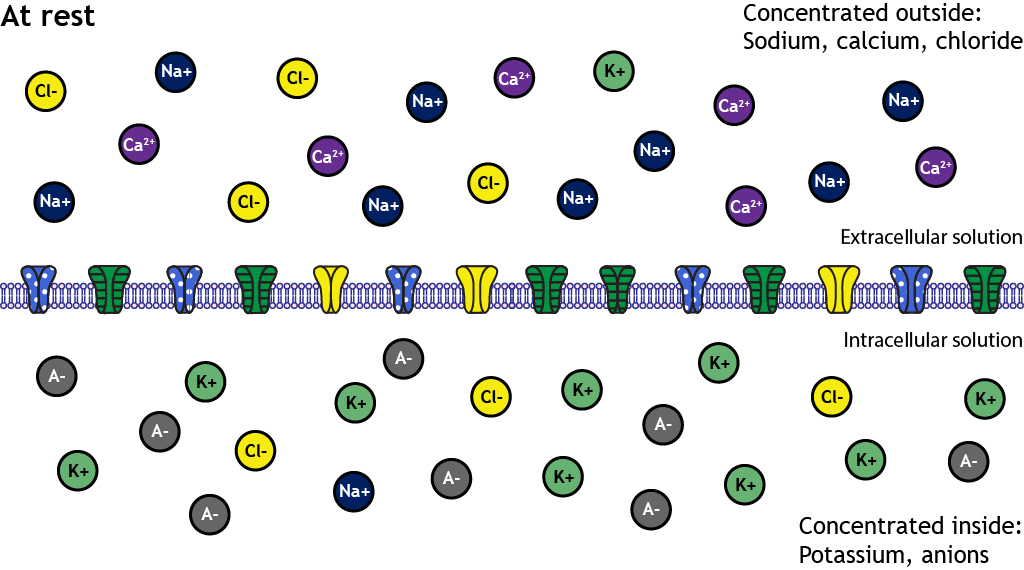
This diagram summarizes the typical resting distribution of major ions across a cell membrane: sodium, calcium, and chloride are higher outside, while potassium and large intracellular anions are higher inside. The separation of these charges (plus selective membrane permeability) underlies a negative resting membrane potential and provides stored energy for signaling and transport. Source
What “voltage across the membrane” means
Even a tiny imbalance in the number of charges near the membrane can create a measurable voltage because charges line up along the membrane surfaces.
Membrane potential: The electrical potential difference across a membrane, typically expressed in millivolts (mV), reflecting charge separation between the inside and outside of the cell.
Membrane potential is dynamic: opening different channels can quickly shift the direction and magnitude of the voltage difference.
Electrochemical gradients: two forces acting together
Ion movement depends on more than concentration alone. A charged ion experiences:
a chemical (concentration) gradient that favours diffusion from high to low concentration
an electrical gradient that favours movement toward the opposite charge
Electrochemical gradient: The combined effect of a solute’s concentration gradient and the electrical gradient across a membrane on that solute’s net movement.
These two components can reinforce each other or oppose each other.
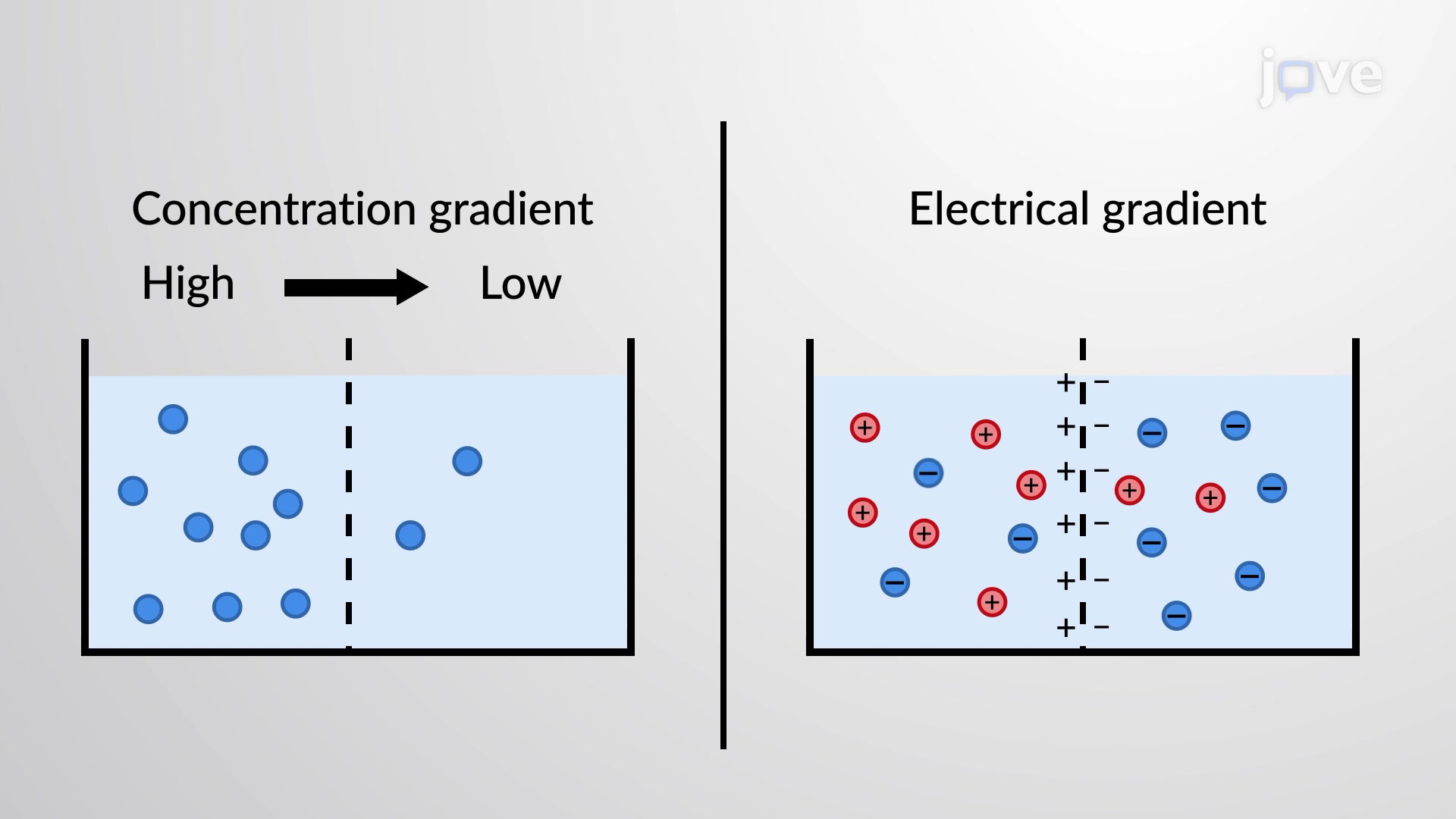
This illustration pairs the concepts of chemical (concentration) gradients and electrical gradients to show how they combine into a single electrochemical gradient. It reinforces that the net direction of ion movement depends on both forces at once, not concentration alone. Source
For example, a positively charged ion may be chemically driven into the cell but electrically pushed out if the inside becomes too positive.
Direction of ion movement through channels
Net diffusion through an open channel depends on the overall electrochemical “push”:
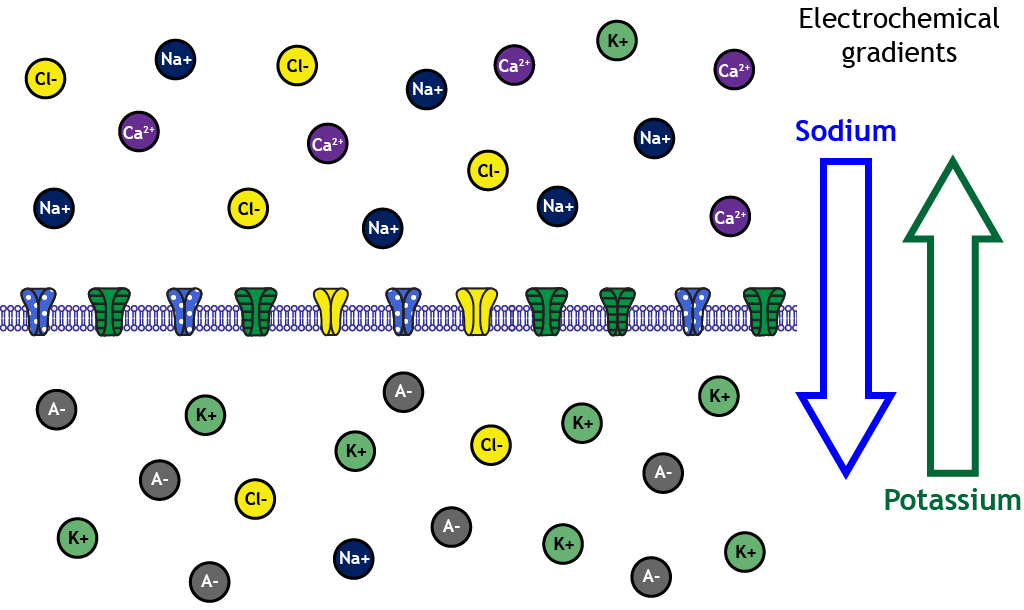
This figure visualizes electrochemical gradients as a net “push” on ions: sodium experiences a strong inward driving force, while potassium tends to move outward under typical resting conditions. It helps connect concentration differences and membrane voltage to the predicted direction of diffusion through open ion channels. Source
If both gradients favour the same direction, movement is rapid in that direction.
If gradients oppose, net movement is reduced and can reach no net flux when forces balance.
The stronger gradient determines the direction of net movement at that moment.
Crucially, channels do not “pull” ions; they provide a selective pathway that allows ions to respond to existing gradients.
Why polarization and gradients matter for cell signalling
Changes in membrane potential can function as information. By opening or closing ion channels, cells can convert a chemical event (like ligand binding) into an electrical change that affects downstream processes.
Key signalling roles include:
Rapid responsiveness: Channel gating can alter membrane potential in milliseconds.
Local to global effects: A change in voltage can influence other voltage-sensitive proteins in the membrane.
Signal specificity: Different channel types (ion selectivity, gating stimuli, and kinetics) produce distinct electrical responses.
In many cells, transient shifts in polarization act as signals that coordinate cellular activities without changing gene expression.
Why polarization and gradients matter for homeostasis
Electrochemical gradients help stabilise internal conditions by enabling controlled ion fluxes that influence:
osmotic balance, since ion distributions affect water movement indirectly
pH stability, because charged solutes interact with buffering systems and transport processes
enzyme and protein function, which can depend on ionic conditions and local electrical environment
steady internal ion levels, maintained by balancing passive channel leakage with regulated channel activity
Homeostasis depends on maintaining gradients while still permitting regulated, purposeful dissipation of those gradients when cells need to communicate or adjust internal conditions.
Channel regulation ties signalling to stability
Cells regulate ion channel activity to avoid uncontrolled gradient collapse:
gated opening (responding to voltage, ligands, or mechanical forces)
inactivation/desensitisation to limit prolonged ion flux
selectivity filters that restrict passage to particular ions, preventing nonspecific charge equalisation
This coordination lets cells use ion diffusion for fast signalling while preserving the electrochemical conditions needed for normal function.
FAQ
Key factors include ion valence ($z$), the steepness of the concentration difference, and how permeable the membrane is to that ion at that moment.
Even with the same concentration gradient, a $2^+$ ion can shift charge distribution differently from a $1^+$ ion when flux occurs.
Common approaches use microelectrodes or patch-clamp methods to compare electrical potential inside versus outside the cell.
Patch clamp can isolate current through single channels, linking channel opening events to changes in voltage or ion flow.
Each ion’s balance point depends on its own concentration difference across the membrane and the membrane voltage.
Because ions differ in permeability and distribution, the membrane can be closer to balance for one ion while far from balance for another, enabling selective responsiveness.
Temperature affects diffusion rates and can alter channel gating kinetics (how quickly channels open/close).
It also changes how strongly concentration differences contribute to the overall driving force, potentially shifting how readily ions move when channels open.
They may block pores, force channels open, or alter gating, changing permeability inappropriately.
Consequences can include persistent depolarisation or hyperpolarisation, reduced signalling precision, and failure to maintain stable internal ionic conditions.
Practice Questions
Explain how the movement of ions through channels can polarise a cell membrane. (2 marks)
States that ions are charged and their movement changes charge distribution across the membrane (1)
Links this to a voltage difference/membrane potential forming between inside and outside (1)
A cell opens a set of ion channels in its plasma membrane. Describe how electrochemical gradients determine the direction and extent of ion movement, and explain why this is important for cell signalling and homeostasis. (6 marks)
Defines/identifies the concentration (chemical) gradient as one component affecting ion movement (1)
Defines/identifies the electrical gradient as the other component affecting ion movement (1)
Explains that the combined effect is the electrochemical gradient (1)
States gradients may reinforce or oppose each other, affecting net flux and direction (1)
Links changes in membrane potential to signalling (e.g. rapid electrical changes act as information) (1)
Links maintaining/controlling gradients to homeostasis (e.g. stable internal conditions/ion balance) (1)

