AP Syllabus focus:
‘Facilitated diffusion uses transport or channel proteins to move charged ions like sodium and potassium across membranes.’
Cells must move ions across hydrophobic membranes to maintain function. Channel proteins provide selective, fast pathways for ions to cross the membrane down gradients, enabling essential transport without directly using cellular ATP.
Core idea: why channels are needed
The interior of the phospholipid bilayer is hydrophobic, so charged ions (e.g., Na⁺, K⁺) cannot readily cross on their own. Channel proteins solve this by creating an aqueous route through the membrane while preserving membrane selectivity.
Key term
Channel protein: A transmembrane protein that forms a selective, hydrophilic pore allowing specific ions to cross the membrane by facilitated diffusion (down a gradient) without direct ATP input.
How channel proteins enable ion movement
Channel proteins support the syllabus focus: facilitated diffusion of charged ions (including sodium and potassium) across membranes.
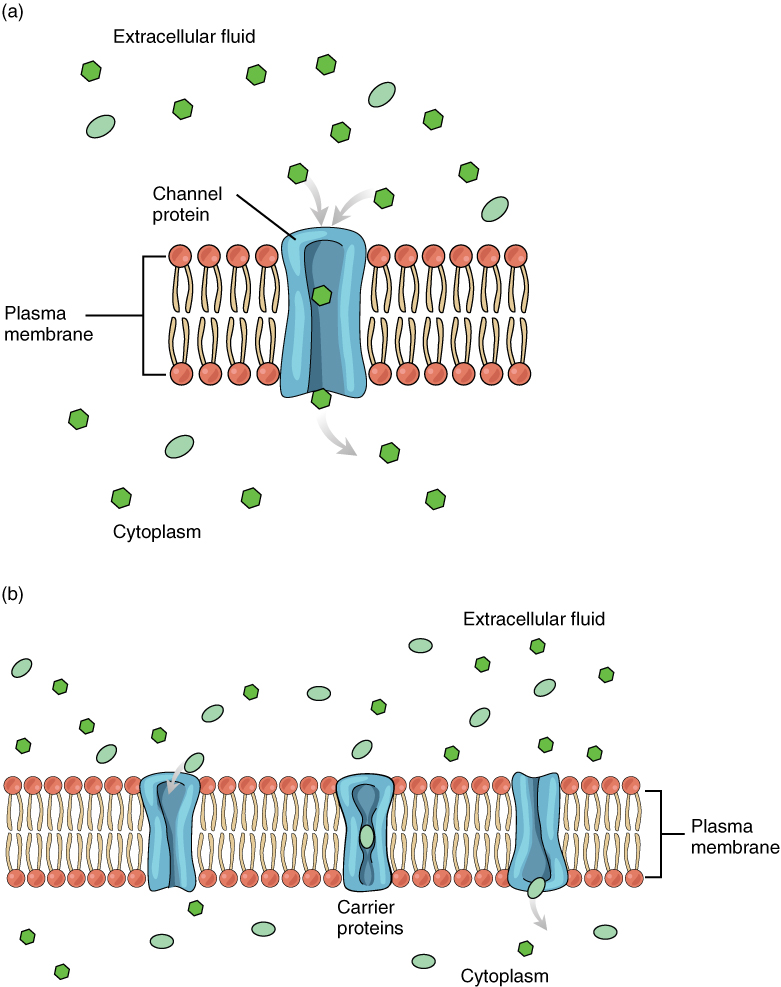
This diagram summarizes facilitated diffusion across the plasma membrane and contrasts channel-mediated transport with carrier-mediated transport. It emphasizes that movement occurs down a concentration gradient and does not require direct ATP input, matching the definition of passive, protein-assisted transport. Source
Mechanism (what actually happens)
Ions move through the channel down their concentration gradient (from higher ion concentration to lower ion concentration).
The channel’s pore provides a hydrophilic environment, lowering the energetic barrier for an ion to pass through the membrane.
Transport is passive: channels do not “pump” ions uphill and do not directly hydrolyse ATP.
What makes channels selective
Selectivity depends on how the pore interacts with ions:
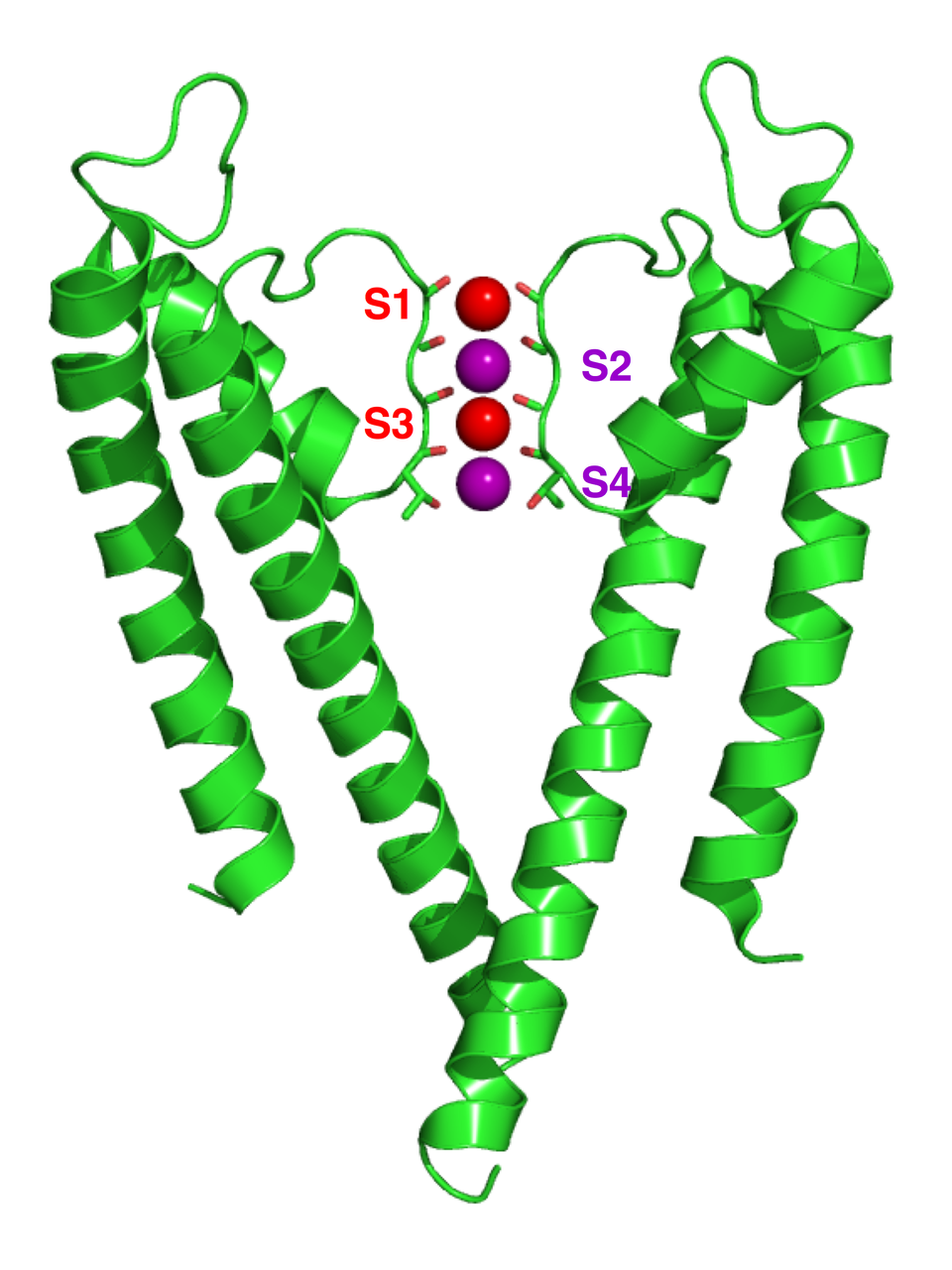
This structural figure of a potassium channel (KcsA) highlights the selectivity filter region where K⁺ ions occupy discrete binding sites (S1–S4). It visually supports the idea that channels can be highly selective because the pore’s geometry and lining stabilize specific ions while excluding others (e.g., Na⁺). Source
Pore size/shape: the channel fits certain ions better than others.
Chemical properties of the pore lining: polar/charged amino acid side chains stabilise particular ions.
Ion hydration effects: ions in water are surrounded by hydration shells; selective channels help ions shed/reform these interactions appropriately during passage.
Types of ion channels (functional categories)
Ion channels vary in when they allow ions through.
Leak channels (often open)
Provide a baseline permeability for certain ions.
Contribute to steady ion movement when gradients exist.
Gated channels (open/close in response to a signal)
Ligand-gated: a specific molecule binds the channel (or a nearby receptor site) and changes channel conformation.
Voltage-gated: changes in membrane voltage alter the channel’s shape, switching it between closed and open states.
Mechanically gated: physical deformation of the membrane or associated structures opens the channel.
These gating mechanisms let cells control when ions cross, while still moving ions by facilitated diffusion when the channel is open.
Rate and direction: what determines ion flow through channels?
Even though channels are passive, the net direction and magnitude of ion movement depend on conditions across the membrane:
Gradient strength: a steeper concentration difference increases net flux when channels are open.
Number of open channels: more open channels increases total membrane permeability to that ion.
Time open (gating dynamics): longer open states allow more ions to pass.
Ion specificity: only ions that the channel selects will move efficiently.
Biological importance of sodium and potassium channels
Channel-mediated movement of Na⁺ and K⁺ is especially important because:
These ions are common and tightly regulated in cells.
Their movement rapidly changes local ion distributions across the membrane.
Controlled permeability to Na⁺ and K⁺ supports many cell-level processes that require quick, reversible changes in ion movement.
FAQ
Patch-clamp electrophysiology isolates a tiny patch of membrane and records current through individual channels.
It can reveal single-channel conductance, opening frequency, and how gating changes in response to ligands or voltage.
Many channels have movable “gate” regions formed by transmembrane helices.
Small conformational shifts can occlude or expose the pore, switching between closed and open states.
Blockers often mimic an ion’s size/charge or bind within the pore.
“Pore plug” binding physically stops ion passage
Allosteric binding stabilises the closed (or inactive) conformation
A channelopathy is a disorder caused by abnormal ion channel function due to mutation or misregulation.
Effects can include altered gating, reduced selectivity, or fewer functional channels in the membrane.
Inactivation is a time-dependent switch to a non-conducting state distinct from simple closure.
It can limit ion flow during sustained stimulation and allows cells to reset responsiveness after a signalling event.
Practice Questions
Explain why sodium ions require channel proteins to cross the plasma membrane by facilitated diffusion. (2 marks)
Na⁺ is charged/polar and cannot pass through the hydrophobic core of the phospholipid bilayer (1).
Channel proteins provide a hydrophilic pathway allowing Na⁺ to move down its gradient without ATP (1).
Describe how channel proteins enable selective movement of potassium ions across membranes and explain two factors that affect the net rate of potassium ion movement through channels. (5 marks)
Channel protein forms a hydrophilic pore through the membrane (1).
Selectivity due to pore size/shape and/or chemical interactions with the ion (1).
K⁺ moves by facilitated diffusion down a concentration gradient when the channel is open (1).
Factor affecting net rate: steeper concentration gradient increases net flux (1).
Factor affecting net rate: increased number of open channels and/or longer open time increases permeability and flux (1).

