AP Syllabus focus: ‘Because particles are too small to count directly, chemists relate the mass of substances in a reaction to the number of particles changing during the reaction.’
Chemistry connects the visible world of measurable mass to the invisible world of atoms, ions, and molecules. The mole is the bridge unit that lets chemists count particles indirectly and compare substances on a consistent scale.
The core problem: particles are unimaginably small
A typical lab sample contains an enormous number of microscopic entities (atoms, molecules, ions). Direct counting is impractical because:
Individual particles are far too small to see or isolate routinely.
Chemical changes involve astronomically large particle counts, even in gram-sized samples.
Reactions depend on numbers of particles, but laboratories measure mass most easily and accurately.
The mole as chemistry’s counting unit
To solve the counting problem, chemists use a counting unit analogous to “dozen,” but far larger, called the mole. This allows particle-level thinking without direct particle counting.
Mole (mol): The SI unit for amount of substance; it represents a fixed number of elementary entities (such as atoms, molecules, or ions).
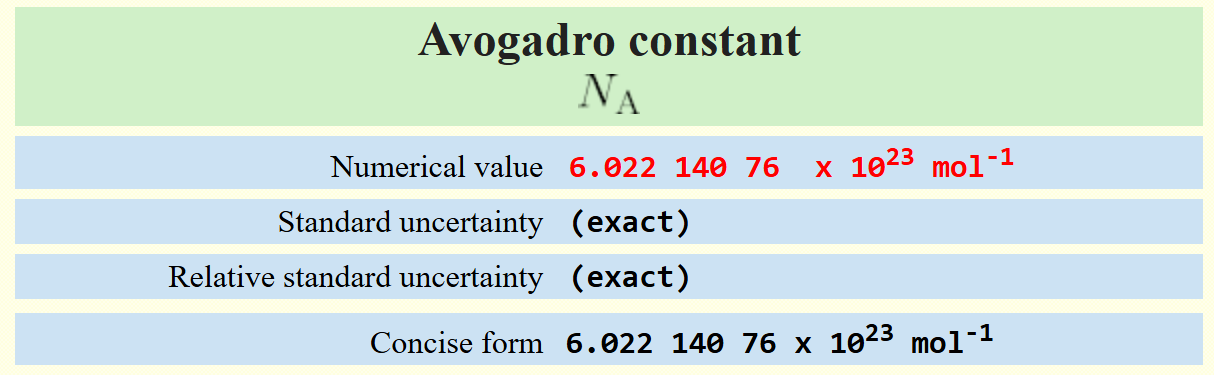
A concise NIST presentation of the Avogadro constant, , with its defined exact numerical value. This emphasizes that the mole is anchored to a fixed count of elementary entities, making “amount of substance” a true counting concept rather than a mass-based one. It provides an authoritative reference point for the definition used throughout mole and stoichiometry calculations. Source
Using moles allows chemists to:
Treat reacting particles in manageable “packages”
Compare different substances using the same counting scale
Convert experimental measurements (mass) into chemically meaningful quantities (amount of substance)
“Amount of substance” is not the same as mass
Amount of substance tracks “how many particles’ worth” of a substance you have, not how heavy it is. Two samples can have the same mass but different amounts of substance because their particles have different masses.
Why mass is the lab-friendly quantity
In typical experiments, mass is preferred because it is:
Fast to measure with balances
Highly precise and reproducible
Additive (masses of components can be combined or separated conceptually)
However, reactions don’t occur because “10 grams collided with 5 grams.” They occur because particles interact.
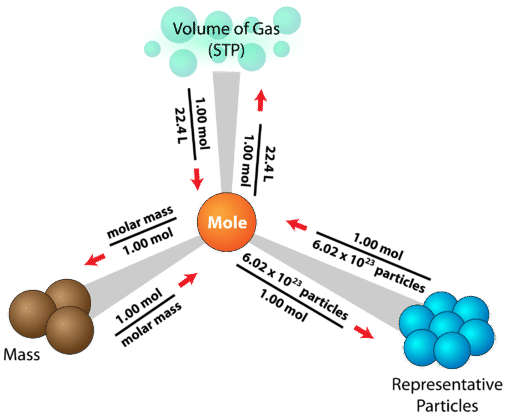
A “mole road map” that organizes the standard conversion pathways among mass (g), amount of substance (mol), and representative particles. The diagram highlights moles as the central hub: molar mass connects grams to moles, and Avogadro’s number connects moles to particle counts. This reinforces why laboratory measurements of mass can be translated into particle-level accounting. Source
The mole provides a standard route from measurable mass to particle count.
Relating mass to particle changes in reactions
The syllabus focus is that chemists relate mass of substances to the number of particles changing during a reaction. This matters because:
Balanced chemical equations describe particle ratios (molecule-to-molecule, ion-to-ion) via coefficients.
Those coefficients must be interpreted as mole ratios in the lab, because moles scale directly with particle number.
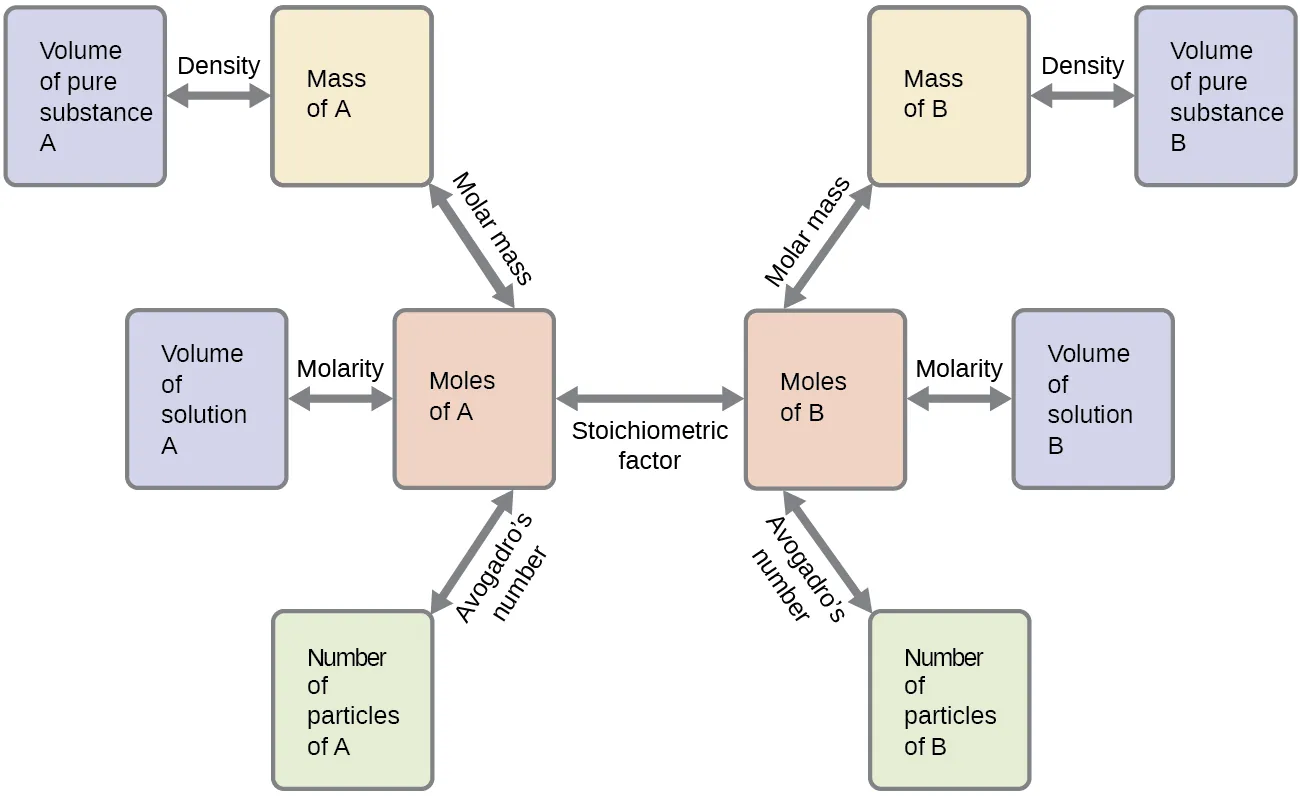
A stoichiometry “workflow” flowchart showing how typical reaction problems move between mass, moles, and particle counts. The central step is converting to moles, applying the stoichiometric factor (mole ratio from the balanced equation), and then converting to the desired quantity (mass, particles, or solution volume). This makes the role of coefficients as mole ratios operational and visually explicit. Source
Once substances are compared in moles, the reaction can be carried out using mass measurements.
Stoichiometry depends on particle ratios
A balanced equation communicates that:
Reactants combine in fixed whole-number ratios of particles.
Products form in fixed whole-number ratios of particles.
In the lab, those same ratios are used as mole ratios, enabling prediction of how much reactant is needed or how much product can form.
Why chemists don’t use mass ratios alone
Mass ratios vary from reaction to reaction and depend on the identities (particle masses) of substances. Particle ratios, in contrast, come directly from chemical formulas and conservation principles.
Key idea:
Conservation of atoms means reactions rearrange atoms rather than create/destroy them.
Therefore, correct reaction accounting requires tracking entities in proportional counts.
The mole is the practical unit for scaling those counts to macroscopic samples.
What the mole enables in real chemical work
Using the mole lets chemists:
Prepare solutions with controlled chemical amounts (consistent reactivity)
Compare yields across substances with different particle masses
Link experimental data (mass change, precipitate collected) to chemical interpretation (how many particles reacted)
Conceptual checkpoints for students
Chemical formulas tell you the relative numbers of atoms in a compound’s particles.
Balanced equations tell you the relative numbers of particles that react.
Laboratory tools give you mass, so you need a standard counting unit (the mole) to connect mass measurements to reaction particle ratios.
FAQ
It is a unit (mol) for amount of substance.
It corresponds to a fixed count of entities, but in calculations you treat it as a measurable unit, like grams or litres.
It depends on what is specified. Common entities include:
atoms (e.g., Ar)
molecules (e.g., CO$_2$)
ions (e.g., Na$^+$)
formula units (e.g., NaCl)
Always match the entity to the chemical description.
$6.02 \times 10^{23}$ is a count, not a mass.
The mass of 1 mol depends on the substance’s particle mass; different substances have different masses per mole.
Balances measure mass precisely, and repeating mass measurements is straightforward.
By converting mass to moles, chemists compare amounts on a particle-based scale while still leveraging accurate macroscopic measurements.
Volume depends strongly on conditions (temperature and pressure), so it is not inherently a counting measure.
Moles provide a condition-independent way to track particle amounts, while volume can be a convenient measurement only when conditions are controlled.
Practice Questions
(2 marks) Explain why chemists use the mole rather than counting individual particles when describing reactions.
States that particles are too small/too numerous to count directly (1)
Explains that the mole links measurable mass to number of particles involved in reactions (1)
(5 marks) A student claims: “A balanced chemical equation is only about mass, so using grams is enough to predict how much product forms.” Evaluate this claim using ideas about particles and the mole.
States that balanced equations represent ratios of particles (or formula units/ions/molecules), not masses (1)
Explains that these ratios are interpreted as mole ratios in practice (1)
States that reactions occur due to interactions between particles, so particle counts matter (1)
Explains that mass is measured experimentally and must be related to particle number via the mole (1)
Concludes the claim is incorrect or incomplete, because grams alone do not reflect the fixed particle ratios given by the equation (1)

