AP Syllabus focus: ‘Avogadro’s number (6.022 × 10^23 per mole) links the amount of substance in moles to the number of atoms, molecules, ions, or formula units present.’
Chemistry connects measurable laboratory quantities to invisible particles. Avogadro’s number provides the bridge between macroscopic moles and microscopic entities, letting you translate “how much” into “how many.”
Core idea: counting by the mole
Amount of substance and Avogadro’s number
Mole (mol): The amount of substance containing exactly 6.022×1023 specified particles (entities).
Because atoms and molecules are far too small to count directly, chemists count them indirectly by counting moles.
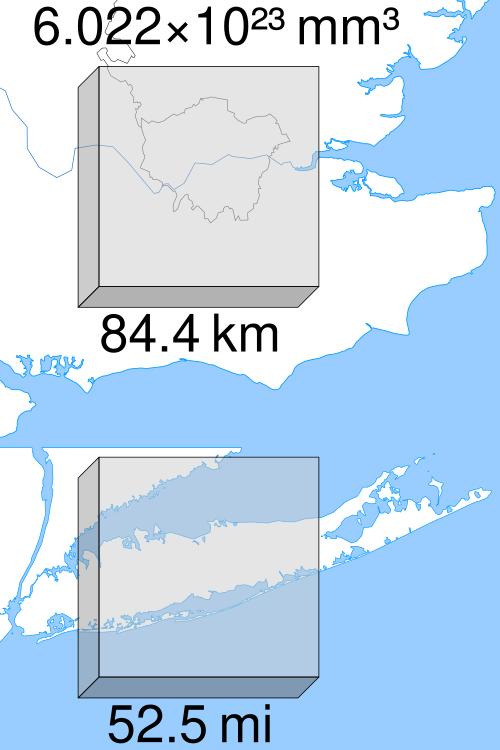
A scale visualization of Avogadro’s number: it represents a mole as 6.022×1023 small cubes aggregated into a much larger cube, overlaid on real maps for size context. The purpose is to build intuition for how extraordinarily large “one mole” is, even though the entities themselves are microscopic. Source
The key constant is Avogadro’s number (also called the Avogadro constant, NA), which tells you the number of entities per 1 mole.
Avogadro’s number (NA): 6.022×1023 entities per mole; the conversion factor between moles and number of particles.
An “entity” must be clearly identified, since different substances are counted differently:
Atoms (e.g., He atoms in helium gas)
Molecules (e.g., H2O molecules in water)
Ions (e.g., Na+ ions in sodium chloride)
Formula units (e.g., NaCl formula units in an ionic solid)
The mole–particle conversion relationship
The central quantitative link is between amount of substance (n) and number of particles (N). Always label what the particles are (atoms, molecules, ions, formula units) to avoid interpretation errors.
N=nNA
N = number of entities (particles)
$</p><p>n=amountofsubstance(mol)</p><p></p><p>N_A=Avogadro’snumber(6.022\times10^{23}\ \text{mol}^{-1})</p></div><p>Thisrelationshipworksforanypuresubstanceaslongastheentityisspecifiedcorrectly.</p><imgsrc="https://tutorchase−production.s3.eu−west−2.amazonaws.com/f1568f5a−2111−4ffd−affa−faf723c99759−file.png"alt="Pastedimage"style="max−width:100N_A),eventhoughthegaseshavedifferentmolarmassesandthereforedifferentsamplemasses.</em><atarget="blank"rel="noopenernoreferrernofollow"href="https://chem.libretexts.org/Bookshelves/IntroductoryChemistry/IntroductoryChemistry(CK−12)/10_2).Whenyouareaskedfor“numberofparticles,”identifywhethertheparticlesare<strong>atoms</strong>or<strong>molecules</strong>.</p><p>Commoninterpretationrules:</p><ul><li><p>Ifthesubstanceiswrittenasasingleelementsymbol(e.g.,Fe),particlesare<strong>atoms</strong>.</p></li><li><p>Ifthesubstanceiswrittenasamolecularformula(e.g.,O_2,P_4),particlesare<strong>molecules</strong>unlessthequestionexplicitlyasksforatoms.</p></li></ul><h3class="editor−heading"><strong>Compounds:moleculesvsformulaunits</strong></h3><p>Forcovalentcompounds(e.g.,CO_2),therepresentativeparticlesare<strong>molecules</strong>.Forioniccompounds(e.g.,CaF_2),therepresentativeparticlesare<strong>formulaunits</strong>,notmolecules.</p><p>Usethesedistinctionscarefully:</p><ul><li><p><strong>Molecules</strong>arediscreteunitswithaspecificcountofatomscovalentlybonded.</p></li><li><p><strong>Formulaunits</strong>representthesimplestwhole−numberratioofionsinanioniclattice(therearenoindividual“NaClmolecules”insolidNaCl).</p></li></ul><h3class="editor−heading"><strong>Ionsandtotalparticlecounts</strong></h3><p>Questionsmayaskforthenumberofaspecifictypeofionratherthanformulaunits.Inthatcase:</p><ul><li><p>Firstinterprettheformulaunitcomposition(e.g.,1Ca^{2+}and2F^-perCaF_2formulaunit).</p></li><li><p>Thenscalethecountofionsrelativetothecountofformulaunits.</p></li></ul><h2class="editor−heading"id="using−units−and−language−precisely"><strong>Usingunitsandlanguageprecisely</strong></h2><h3class="editor−heading"><strong>Conversion−factormindset</strong></h3><p>Avogadro’snumberfunctionslikea“particles−per−mole”conversion:</p><ul><li><p>multiplybyN_Atoconvert<strong>mol→particles</strong></p></li><li><p>dividebyN_Atoconvert<strong>particles→mol</strong></p><p></p></li></ul><divclass="example−section"><p>n = \dfrac{N}{N_A}</p><p>n=amountofsubstance(mol)</p><p></p><p>N=numberofentities(particles)</p><p></p><p>N_A=Avogadro’snumber(6.022\times10^{23}\ \text{mol}^{-1})</p></div><h3class="editor−heading"><strong>Whattostateinyoursetup</strong></h3><p>Tocommunicateclearly(andearnfullcredit),consistentlyinclude:</p><ul><li><p>the<strong>identityoftheentity</strong>(atoms,molecules,ions,formulaunits)</p></li><li><p>theunit\text{mol}^{-1}withN_A$
the correct interpretation of what “particle” means in context (especially for ionic solids and diatomic elements)


