AP Syllabus focus: ‘The average mass of one particle in amu is numerically equal to the molar mass in grams per mole, creating a quantitative link between mass and particle count.’
Chemists constantly move between the microscopic world of individual atoms and the macroscopic world of lab-scale masses. This subtopic explains the two mass scales—amu and g/mol—and why their numbers match.
Two mass scales for the same particles
Atomic mass unit (amu): mass per particle
Atomic and molecular masses are often reported in atomic mass units (amu) because individual particles are extremely small.
Atomic mass unit (amu) — a microscopic unit of mass used for atoms and molecules; values on the periodic table reflect the average mass of atoms in amu.
A key idea for AP Chemistry is that an element’s atomic mass (in amu) describes the mass of a single atom on this microscopic scale, while a compound’s molecular/formula mass (in amu) describes the mass of a single molecule or formula unit.
Molar mass: mass per mole of particles
In the lab, you measure mass in grams, so chemists use moles to count particles indirectly and connect particle numbers to measurable mass.
Molar mass — the mass of 1 mole of a substance, expressed in grams per mole (g/mol).
For an element, the molar mass corresponds to 1 mole of atoms. For a molecular substance, it corresponds to 1 mole of molecules. For an ionic compound, it corresponds to 1 mole of formula units.
The numerical equality that makes chemistry practical
The central AP relationship is:
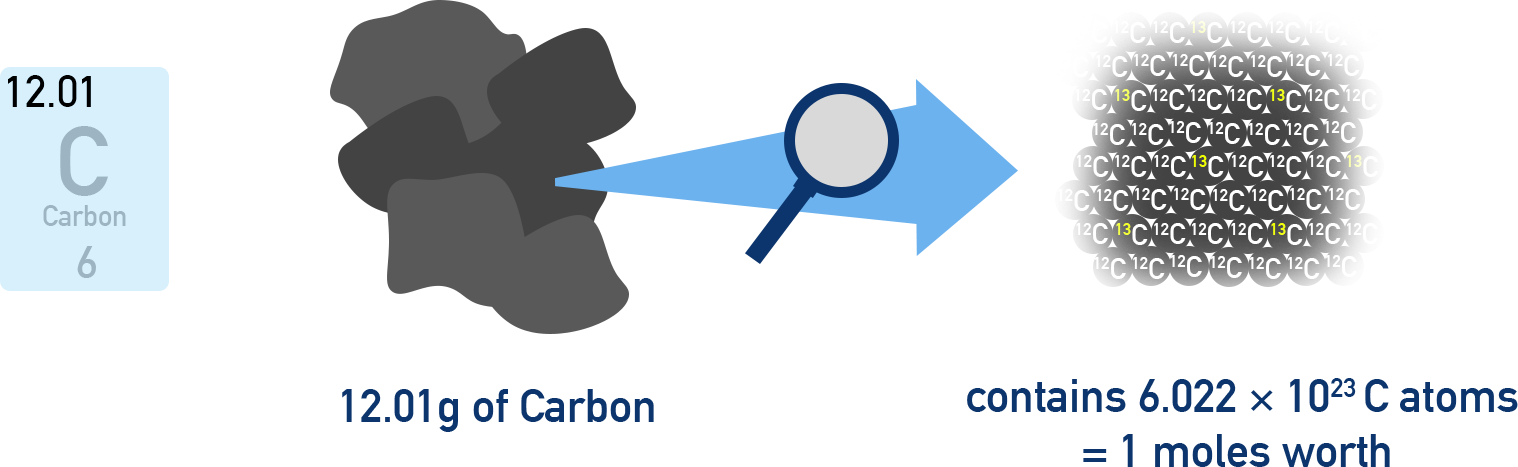
This diagram illustrates the mole concept using carbon: an atomic mass of 12.01 amu corresponds to a molar mass of 12.01 g/mol. It visually links a lab-scale sample (12.01 g C) to a particle count of one mole, carbon atoms. Source
The average mass of one particle in amu is numerically equal to the molar mass in g/mol.
This is what allows you to treat a periodic-table atomic mass (amu) as a molar mass (g/mol) without additional conversion steps in most AP problems.
This equality is not a coincidence: the mole is defined so that a mole of particles has a mass in grams that matches the particle’s mass in amu. As a result:
An atom with mass 12.01 amu corresponds to a molar mass of 12.01 g/mol.
A molecule with mass 18.02 amu corresponds to a molar mass of 18.02 g/mol.
= molar mass of the substance in g/mol
= mass of one atom, molecule, or formula unit on the amu scale
= Avogadro’s constant in (particles per mole)
The first relationship is the AP “shortcut” used constantly: match the number, change the unit context (amu per particle becomes g/mol per mole). The second relationship explains the deeper link between the two scales: since 1 mole contains particles, “per particle” masses and “per mole” masses differ by a factor of .
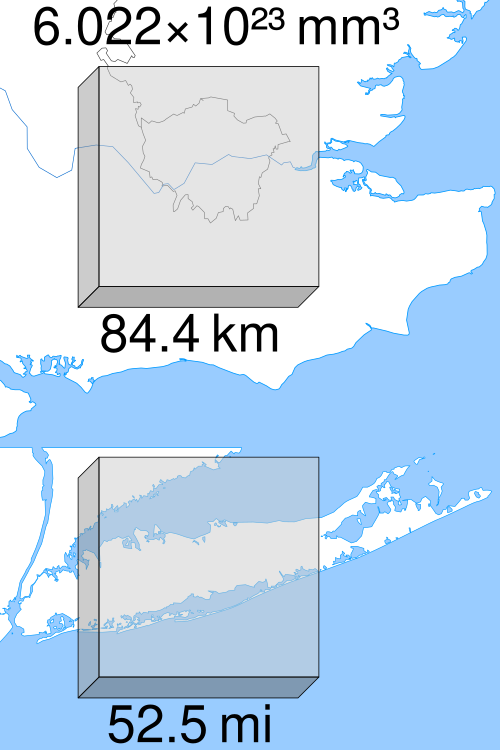
This visualization represents a mole by imagining tiny cubes assembled into one large cube. By comparing that cube’s side length to real geographic maps, the graphic emphasizes the physical scale implied by Avogadro’s constant, . Source
Using atomic masses to build molar masses
To find a substance’s molar mass, you add contributions from each element using subscripts as particle counts.
For an element:
Molar mass (g/mol) = atomic mass from the periodic table (amu) with units changed to g/mol.
For a compound:
Multiply each element’s atomic mass by its subscript.
Add the results to get the total molar mass in g/mol.
This works because the periodic table provides average atomic masses, so the resulting molar mass is also an average over the naturally occurring isotopes.
What to watch for (common AP pitfalls)
Units must match the context:
amu refers to one particle; g/mol refers to one mole of particles.
Don’t confuse mass number with atomic mass:
mass number is an integer for a specific isotope; atomic mass is usually non-integer because it’s an average.
Be clear about the counted entity:
covalent substances: count molecules
ionic substances: count formula units
Significant figures:
use the periodic table’s reported precision; don’t round too early when summing atomic masses.
FAQ
Historically, 1 amu was set relative to carbon-12 so atomic masses could be compared consistently.
Atomic mass: weighted average on the periodic table.
Mass number: protons + neutrons for one isotope.
Isotopic mass: mass of one specific isotope.
They are averages over naturally occurring isotopes, so the mean is rarely an integer.
Formula mass is in amu for one formula unit; molar mass is in g/mol for one mole of formula units. The numerical value matches.
You can use $1\ \text{amu} \approx 1.6605 \times 10^{-24}\ \text{g}$ for converting a single particle’s mass into grams.
Practice Questions
Magnesium has an atomic mass of 24.3 amu. State the molar mass of magnesium atoms, with units. (2 marks)
24.3 (1)
g/mol (1)
A compound has a particle mass of 98.0 amu per molecule.
(a) State its molar mass. (2 marks)
(b) Using , determine the mass in grams of one molecule. (3 marks)
98.0 (1)
g/mol (1) (b)
Uses or equivalent reasoning linking amu to grams via (1)
Correct substitution leading to g (1)
Correct numerical value to appropriate significant figures, e.g. g (1)

