AP Syllabus focus: ‘Some pure substances consist of individual molecules, while others are lattices of atoms or ions held together in fixed ratios described by a formula unit.’
Chemists describe pure substances by the type of particles present. Distinguishing discrete molecules from extended lattices (ionic, atomic, or metallic) determines how formulas are interpreted and how particle-level models are drawn.
Pure substances and what “particle” means
A pure substance contains only one type of chemical species throughout (even if that species contains more than one element). In AP Chemistry, the key question is whether the substance is made of discrete units or an extended lattice.
Discrete units: substances made of molecules
Molecule: A discrete, electrically neutral group of atoms held together by covalent bonds, existing as an individual unit.
Molecular substances are composed of many separate molecules with strong covalent bonds inside each molecule and comparatively weaker forces between molecules (often intermolecular forces). Their chemical formula is a molecular formula that gives the actual number of each type of atom in one molecule (for example, is one molecule with 2 H and 1 O).
Common cues that a substance is molecular:
It is composed only of nonmetals (or a nonmetal + metalloid in many covalent compounds).
The particles can be pictured as separate clusters (molecule-by-molecule).
The formula can, in principle, be scaled down to one particle that still “exists” as that same unit.
Extended structures: lattices of ions or atoms
Many pure substances are not made of individual molecules.
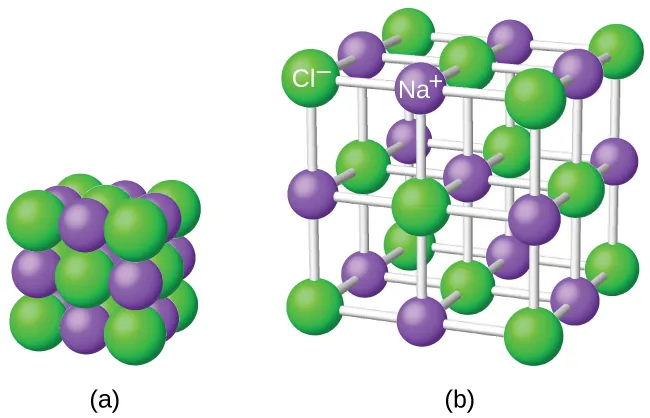
Two complementary particulate diagrams of solid NaCl: a compact 3D lattice view and an expanded view that clarifies the repeating ion arrangement. The labels emphasize that the structure is an extended array of and ions rather than discrete “NaCl molecules.” This is a concrete visual anchor for interpreting NaCl as a formula unit (fixed ratio) within a continuous crystal. Source
Instead, they are continuous repeating structures with no smallest “molecule” that preserves the structure.
Lattice: A repeating, three-dimensional arrangement of particles (ions or atoms) extending throughout a solid.
A lattice description is used for:
Ionic solids: a repeating arrangement of cations and anions
Covalent network solids: a repeating arrangement of atoms connected by covalent bonds
Metallic solids: a repeating arrangement of metal atoms (often modeled as metal cations in a sea of delocalized electrons)
Because there is no discrete molecule to count, the written chemical formula is interpreted differently.
Formula units and fixed ratios
When a pure substance is an ionic solid (and in some lattice-type solids), chemists use the idea of a formula unit to communicate composition.
Formula unit: The lowest whole-number ratio of ions (or atoms, in some lattices) represented by the chemical formula of an extended solid.
A formula unit is not a “piece” you can physically isolate from the crystal; it is a ratio statement that reflects how the lattice repeats.
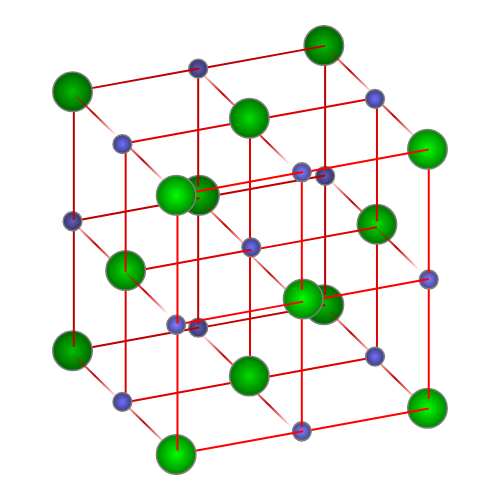

A 3D particulate model of an ionic crystal lattice, showing alternating ions packed into a repeating array. The visual reinforces why a “formula unit” is a composition ratio (e.g., in NaCl) rather than a separable chunk of the solid. In an actual crystal, the pattern extends throughout the entire solid, with no smallest self-contained molecule. Source
This directly matches the syllabus emphasis: some pure substances are lattices “held together in fixed ratios described by a formula unit.”
Key implications:
The formula in an ionic compound gives the simplest ratio of ions that results in overall charge neutrality.
The “particles” in an ionic solid are ions, but the formula unit combines them in the smallest neutral ratio (e.g., one cation with one anion, or multiple ions as needed).
For covalent network solids (e.g., many silicon/oxygen solids), the formula likewise indicates a repeating ratio, not a discrete molecule.
Interpreting chemical formulas correctly
To decide whether a formula refers to molecules or formula units, focus on structure type:
Molecular formula interpretation (discrete molecules)
The subscripts describe atoms within one molecule
Many identical molecules make up the sample
Formula unit interpretation (extended lattice)
The subscripts describe the lowest repeating ratio in the lattice
The sample is one continuous network of repeating units
Common student pitfalls to avoid
Treating an ionic compound’s formula as if it describes a single “molecule” floating around.
Assuming every solid is a lattice of ions; many solids are molecular (made of molecules packed together) while others are covalent network or metallic.
Forgetting that “lowest ratio” language applies to formula units: the written formula is a compositional simplification, not necessarily a count of atoms in a discrete particle.
FAQ
Check element types. Compounds of a metal with a nonmetal are usually ionic lattices (formula units). Compounds of only nonmetals are usually molecular (molecules), unless known network solids.
Yes. Their formulas give a repeating ratio in an extended covalent lattice, not a discrete molecule you can isolate.
No. Some solids are molecular (packed molecules). Others are ionic, metallic, or covalent network lattices; the bonding/structure type matters.
They change what repeats in the lattice. The formula unit still gives the lowest ratio, but one “ion” in that ratio may be a multi-atom polyatomic ion.
A molecule is a discrete bonded unit. A formula unit is a bookkeeping ratio for an extended lattice; separating exactly one formula unit would disrupt the continuous structure.
Practice Questions
State whether the substance represented by is best described as consisting of molecules or formula units. Give one reason. (2 marks)
Molecules (1)
Reason linked to discrete covalent units / nonmetals covalently bonded (1)
A student claims “ exists as molecules because it has a chemical formula.” Explain why this statement is incorrect, using the ideas of lattices and formula units. (5 marks)
Identifies as an ionic solid with a lattice of ions (1)
Explains there are no discrete molecules in an ionic lattice (1)
Defines/interprets “formula unit” as lowest whole-number ratio in the lattice (1)
States the ratio reflects repeating composition, not a single separable particle (1)
Links to charge balance: with two gives neutral ratio (1)

