AP Syllabus focus: ‘In any pure sample of a compound, the mass ratio of its constituent elements is constant; this is the law of definite proportions.’
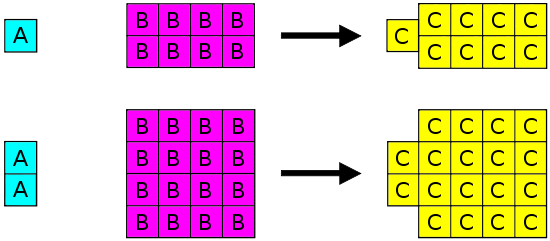
This diagram visualizes the scaling idea behind definite proportions: if a fixed mass ratio (e.g., 1 g of A to 8 g of B) forms a compound, doubling the amount doubles each reactant mass while preserving the same ratio. It reinforces that the composition of a pure compound is independent of sample size. Source
The law of definite proportions connects the microscopic idea of fixed atomic ratios to macroscopic measurements. It explains why a compound’s composition is predictable and reproducible, enabling consistent analysis, identification, and reaction planning.
Core Idea: Fixed Mass Ratios in Compounds
Compounds are substances made of elements chemically combined in fixed, whole-number ratios. Because the atom ratio in a compound is fixed, the mass ratio of the elements is also fixed (masses scale directly with numbers of atoms).
Law of Definite Proportions: In any pure sample of a compound, the mass ratio of its constituent elements is constant.
This law applies regardless of:
Sample size (small or large)
Source (lab-made or naturally occurring)
Physical form (powder, crystal, dissolved—so long as it is still the same pure compound)
Why the Mass Ratio Must Be Constant
At the particle level, a chemical formula indicates a fixed ratio of atoms per “unit” of the compound. When you scale up to a measurable amount, you are just combining many identical units, so the element masses increase proportionally.
The constant mass ratio arises from:
Fixed whole-number atom ratios in a chemical formula
Constant atomic masses for each element (using average atomic mass from isotopic abundance)
Additivity of mass when many formula units are present
Connecting Formula to Mass Ratio (Conceptual)
For a compound with formula , each formula unit contains:
atoms of
atoms of
So the mass contribution from each element in one formula unit is proportional to and , where is molar mass.
A useful way to express the idea is:
= subscript for element in the chemical formula (unitless)
= molar mass of element (g/mol)
= subscript for element in the chemical formula (unitless)
= molar mass of element (g/mol)
This relationship is not a “calculation requirement” by itself; it formalizes the reasoning that a fixed formula produces a fixed mass ratio.
What “Pure Sample” Means (and Why It Matters)
The specification statement hinges on pure sample.
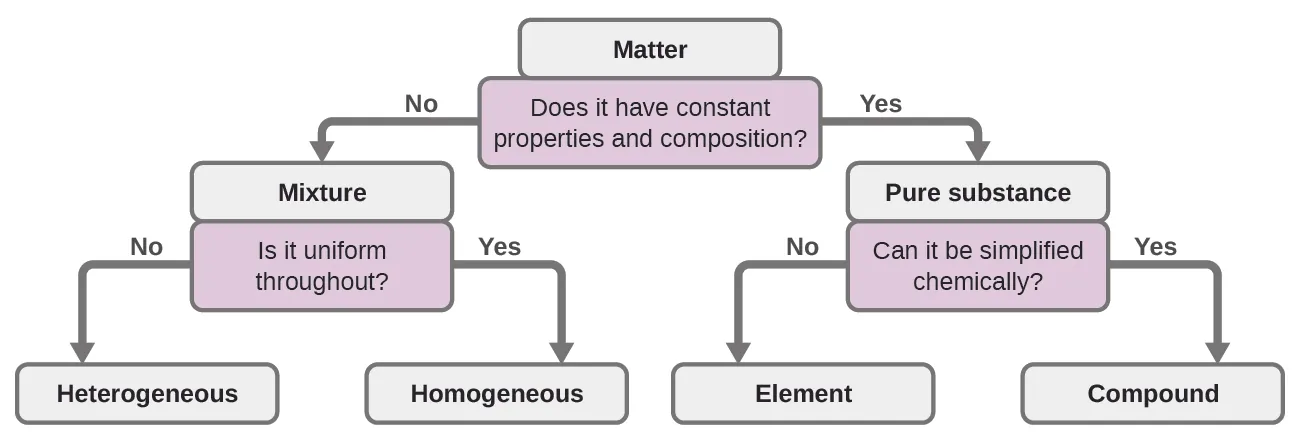
This flowchart classifies matter by whether it has constant composition, separating mixtures (variable composition) from pure substances (constant composition). It provides a clean conceptual bridge to why the law of definite proportions applies only when the sample is truly one compound rather than a mixture. Source
Common reasons apparent mass ratios can change (without violating the law):
Mixtures of different compounds containing the same elements
Hydrates vs anhydrous salts (different compounds with different fixed ratios)
Contamination (extra material adds mass without matching the compound’s composition)
Incomplete reaction leaving unreacted reactant mixed with product
When the identity is truly one compound, the mass ratio is constant; when the identity is not one compound, the ratio may vary.
How This Law Is Used in Chemistry
Identifying or confirming a compound
Because each compound has a characteristic mass composition, composition data can support whether two samples are the same compound (assuming both are pure).
Checking consistency in synthesis and manufacturing
Industrial chemistry relies on definite proportions to ensure that a product has the correct composition and therefore consistent properties (such as reactivity and safety).
Supporting atomic theory
The law is evidence that matter is made of discrete particles that combine in consistent, countable ways, rather than in arbitrary fractions.
Common Pitfalls and Clarifications
The law does not say different compounds made from the same elements have the same ratio; different formulas give different fixed ratios.
A compound can deviate from “ideal” measurements due to experimental error, but the underlying ratio remains constant.
“Constant” refers to the mass ratio of elements, not necessarily the total mass (which depends on how much sample you have).
FAQ
Yes. Whether a substance is molecular or ionic, a pure compound has a fixed formula, so the element mass ratio remains constant.
Natural isotopic abundances make atomic masses effectively constant for typical samples. Only artificially enriched isotopic samples can shift measured ratios slightly.
They can be different compounds with different formulas (for example, different oxides), each with its own constant mass ratio.
Residual solvent/water on the sample
Impure reagents
Incomplete drying or reaction
Poor mixing leading to non-representative subsamples
Not always. It can also indicate inconsistent experimental handling (e.g., variable moisture content). Reproducibility after proper purification/drying helps distinguish these cases.
Practice Questions
(2 marks) State the law of definite proportions and describe what it implies about the composition of a pure compound sample.
1 mark: Correct statement that a pure compound has a constant mass ratio of its elements.
1 mark: Clear implication (e.g., ratio unchanged with sample size/source; fixed composition).
(5 marks) A student analyses two solid samples, X and Y, each containing only carbon and oxygen. Sample X gives a constant carbon:oxygen mass ratio across repeated trials. Sample Y gives different carbon:oxygen mass ratios in different portions tested. Using the law of definite proportions, explain what can be inferred about X and Y, and give two plausible reasons for Y’s varying results.
1 mark: X is consistent with a single pure compound (fixed mass ratio).
1 mark: Y is not consistent with a single pure compound as tested (ratio varies).
1 mark: Reason 1 linked to mixtures/contamination (e.g., mixture of two C–O compounds).
1 mark: Reason 2 linked to differing composition compounds (e.g., presence of hydrate/impurity/unreacted reactant) or sampling inhomogeneity.
1 mark: Explicit connection back to the law (pure compound → constant mass ratio; variation indicates not purely one compound or not uniform).

