AP Syllabus focus: ‘The empirical formula lists the lowest whole-number ratio of atoms of each element in a compound.’
An empirical formula is the simplest “recipe” for a compound’s composition. It communicates atom ratios without implying an exact molecule size, making it a compact, high-utility representation for many AP Chemistry tasks.
What an Empirical Formula Represents
An empirical formula expresses relative composition only: it tells you how many atoms of each element are present compared to the others, reduced to the smallest whole numbers.
Empirical formula: the chemical formula showing the lowest whole-number ratio of atoms (or ions) of each element in a compound.
Because it is ratio-based, an empirical formula does not necessarily match how the compound exists as discrete particles.
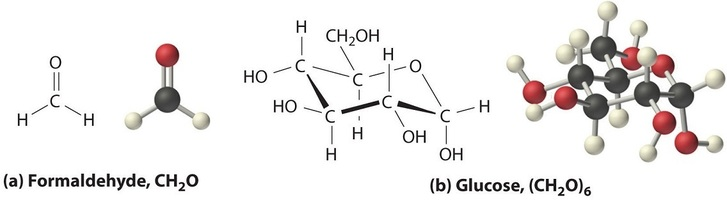
Side-by-side structural representations of (a) formaldehyde () and (b) glucose (), highlighting that very different molecules can share the same empirical formula. The figure visually connects “simplest ratio” to “actual molecule,” showing how molecular formulas can be whole-number multiples of an empirical formula. Source
Some substances exist as molecules, while others are extended ionic or network structures; the empirical formula remains a valid way to express composition in each case.
Key Features of Empirical Formulas
Subscripts are integers in the smallest possible ratio (no common factor greater than 1).
The ratio refers to atoms of elements (or, for ionic compounds, ions in a formula unit) present in the compound.
Empirical formulas are often used when composition data are available but true molecular size is not.
Empirical Formula vs Molecular Formula
Many molecular compounds have an empirical formula that is a reduced form of the molecular formula. For example, a compound with molecular formula has empirical formula because all subscripts share a factor of 6.
Molecular formula: the chemical formula that gives the actual number of atoms of each element in a single molecule of a molecular compound.
The relationship between these two formulas is always a whole-number scaling: a molecular formula is an integer multiple of the empirical formula (and equals it only when the empirical ratio is already the true atom count).
The “Lowest Whole-Number Ratio” Requirement
Reducing to the lowest whole-number ratio means:
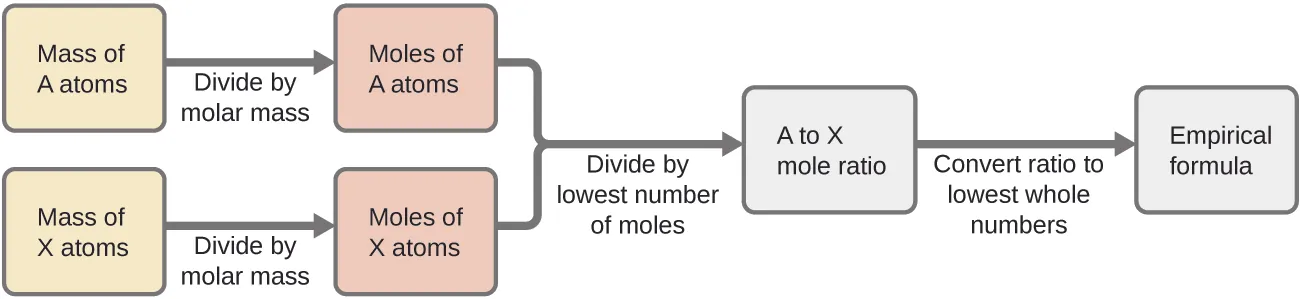
Flowchart showing the standard empirical-formula workflow: convert each element’s measured mass to moles (using molar mass), divide all mole amounts by the smallest to get a ratio, then scale to the lowest whole-number subscripts. This visual emphasizes that subscripts come from mole ratios, not from raw masses. Source
Identify a common factor across all subscripts and divide by it.
The final subscripts must remain whole numbers (chemical formulas do not use fractional subscripts).
When Ratios Are Not Obvious Integers
Real data can imply ratios that are close to, but not exactly, integers. Conceptually, the empirical formula corresponds to the nearest chemically reasonable small-integer ratio consistent with the composition. In AP Chemistry contexts, ratios are typically designed to reduce cleanly or with simple integer scaling.
Empirical Formula Mass and Scaling to a Molecular Formula
To connect an empirical formula to a molecular formula, you compare the compound’s molar mass to the mass represented by one empirical “unit.” This introduces the idea of an integer multiplier .
A key point: if , the empirical and molecular formulas are identical; if , every subscript in the empirical formula is doubled, and so on.
= integer multiplier (unitless)
= mass per mole of the compound, in
= mass of one empirical-formula “set” of atoms, in
Common Pitfalls (Conceptual)
Confusing simplest ratio (empirical) with actual formula (molecular).
Attempting to “simplify” ionic compound formulas beyond charge balance; empirical formulas must still reflect a valid neutral composition.
Treating subscripts as percentages or masses; they are counts in a ratio, not direct mass information.
FAQ
If the subscripts in the molecular formula share no common factor greater than 1, the formula is already in the lowest whole-number ratio.
Examples include $H_2O$ and $CO_2$.
Yes. A subscript of 1 is implied and normally not written.
For instance, $NaCl$ means $Na_1Cl_1$ in a 1:1 ratio.
Not necessarily. If the molecular formula is already in lowest terms, it will look the same as the empirical formula.
“Simpler” refers to reducibility, not visual length.
An empirical formula can represent the simplest elemental ratio, but it may not match a polymer’s repeating unit.
A polymer might have a repeat unit that is a multiple of the empirical ratio.
In correct chemical contexts, $n$ should be a whole number within rounding. A non-integer usually indicates inconsistent data or an incorrect molar mass/empirical formula mass.
Check significant figures and whether the empirical formula mass was computed correctly.
Practice Questions
(2 marks) State what is meant by an empirical formula.
Defines it as a formula showing the simplest/lowest whole-number ratio of atoms of each element (1)
Makes clear it is a ratio and not necessarily the actual numbers in a molecule (1)
(5 marks) A compound has empirical formula and molar mass . Determine the molecular formula.
Calculates empirical formula mass: (1)
Uses (1)
States that molecular formula is an integer multiple of empirical formula (1)
Multiplies each subscript by 6 to obtain (1)
Correct final molecular formula given (1)

