AP Syllabus focus: ‘Pure substances contain only one type of particle. Mixtures contain two or more types of particles, and their relative proportions can vary.’
Chemists classify matter as pure substances or mixtures to predict properties and interpret experimental data. The key distinction is particle identity: one kind of particle throughout versus multiple kinds whose amounts can change.
Core idea: what “pure” means in chemistry
A chemical classification is based on the types of particles present (atoms, molecules, ions), not on appearance.
Pure substance: Matter made of only one type of particle (one chemical species) with a fixed, characteristic composition.
A pure substance can be:
An element (only one type of atom)
A compound (one type of molecule or one repeating ionic formula unit)
Pure substances have constant composition regardless of sample size or source because every particle is the same chemical species.
Core idea: what a mixture is
A mixture contains more than one particle type present together without being chemically bonded into a single, fixed composition.
Mixture: Matter containing two or more types of particles (two or more substances) physically combined, with proportions that can vary.
Because different samples can contain different ratios of components, mixtures have variable composition. This is the meaning of “their relative proportions can vary” in the syllabus statement.
Components and uniformity
When describing mixtures, it helps to name the substances present.
Component: One of the pure substances that makes up a mixture.
A mixture may be uniform or non-uniform on the scale you can observe.
Homogeneous mixture: A mixture with uniform composition throughout (single visible region), so any small sample has the same composition as the whole.
Homogeneous mixtures are often called solutions in AP Chemistry contexts (for example, salt dissolved in water).
Heterogeneous mixture: A mixture whose composition is not uniform throughout (multiple visible regions), so different samples can have different compositions.
In heterogeneous mixtures, different parts can contain different proportions of components, consistent with variable composition.
How to classify a sample: practical decision points
Use these checks to decide whether a sample is a pure substance or a mixture, staying focused on particle types and variability.
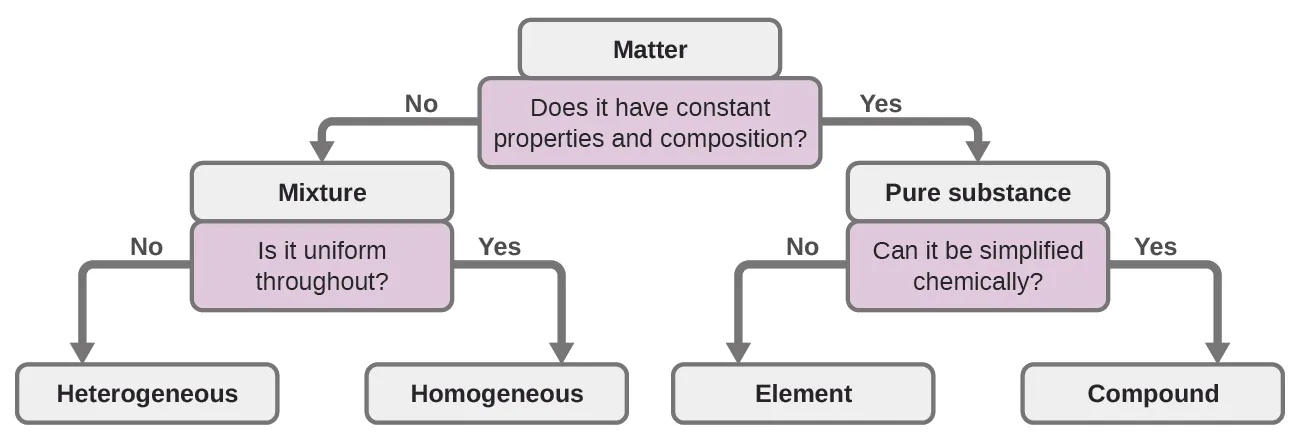
Flowchart for classifying matter by composition and uniformity. It emphasizes the key branching ideas you use in these notes: constant composition implies a pure substance (element or compound), while variable composition implies a mixture (homogeneous or heterogeneous). Source
Check 1: How many particle types are present?
Pure substance: one particle type (all identical atoms, identical molecules, or identical formula units)
Mixture: at least two particle types present together
Check 2: Does the composition have to be fixed?
Pure substance: fixed composition (a defining feature)
Mixture: composition can change (for example, more or less of one component can be added without creating a new chemical substance)
Check 3: Is the sample uniform throughout?
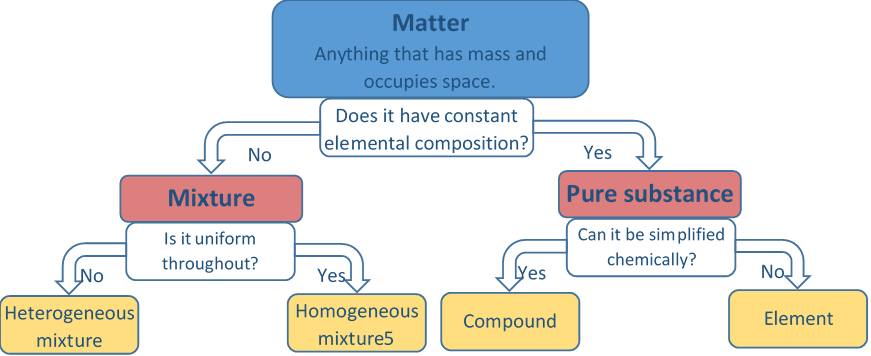
Schematic decision tree for classifying matter into pure substances (elements/compounds) versus mixtures (homogeneous/heterogeneous). The diagram visually encodes the logic that ‘uniform throughout’ is a test for mixture type, not a guarantee of purity. Source
Uniform throughout suggests homogeneous mixture or pure substance
Non-uniform suggests heterogeneous mixture Uniform appearance alone does not guarantee purity; it only addresses whether the mixture is homogeneous.
Particle-level perspective (what you should picture)
Classification is strongest when tied to a particulate model:

Comparative particle-diagram figure contrasting a solution (homogeneous mixture) with a colloid and a suspension (heterogeneous mixture). The particle sketches emphasize that homogeneous mixtures have dispersed species that do not settle, whereas heterogeneous mixtures contain larger dispersed particles that can settle or form multiple regions/phases. Source
Element (pure): one kind of atom only
Compound (pure): identical molecules repeated, or identical ionic formula units repeated
Mixture: a physical intermingling of different particles (different molecules and/or ions and/or atoms) in variable ratios
The AP distinction is therefore about particle identity and whether the relative amounts are constrained (pure substances) or adjustable (mixtures).
FAQ
No. “Pure” only means one chemical species is present.
A pure substance can be hazardous (e.g., pure methanol), while a mixture can be safe (e.g., air in normal conditions).
Because it looks uniform.
At the particle level it still contains multiple particle types, so the composition can be changed by adding/removing components without forming a new substance.
Under normal conditions, no.
Air contains multiple gases (different particles), and humidity and trace gas levels vary, so the relative proportions can vary from place to place and time to time.
Not necessarily.
Filtering can remove undissolved solids from a heterogeneous mixture, but dissolved substances remain in a homogeneous mixture; the filtrate can still contain multiple particle types.
For ionic compounds, the repeating unit is a single formula unit (fixed ratio of ions).
Even though cations and anions are present, the compound is one pure substance because it has one fixed composition and one chemical identity.
Practice Questions
(2 marks) Explain, in terms of particles, the difference between a pure substance and a mixture.
States that a pure substance contains only one type of particle / one chemical species. (1)
States that a mixture contains two or more types of particles / substances and that their proportions can vary. (1)
(5 marks) A student is given four samples: A contains only molecules; B contains and molecules uniformly mixed; C contains visible solid particles suspended in a liquid; D is described as “a substance with the same composition in every portion of the sample”.
(a) Classify A, B, and C as pure substance, homogeneous mixture, or heterogeneous mixture.
(b) For B and D, state one statement about composition that justifies your classification.
A: pure substance (one particle type, only). (1)
B: homogeneous mixture (two particle types uniformly mixed). (1)
C: heterogeneous mixture (non-uniform; more than one visible region/particle distribution). (1) (b)
B: proportions can vary / contains more than one particle type. (1)
D: composition is fixed throughout / consistent with a pure substance (or homogeneous mixture only if explicitly stated as multiple substances; accept “fixed composition implies pure”). (1)

