AP Syllabus focus: ‘Use composition-by-mass information to calculate how much of each substance is present in a mixture and to connect mass data to particle ratios.’
Mixture-composition problems translate experimental mass data into chemically meaningful amounts. You will use mass relationships to determine how much of each component is present, then convert to moles to compare particle-level ratios.
What “composition of a mixture” means in practice
A mixture contains multiple substances present together, so the key task is to determine each component’s mass and often its amount (moles) from limited information (total mass, percent by mass, elemental analysis, or a recoverable product).
Mass-based descriptions you may be given
Many mixture questions start from a component’s share of the total sample.
Mass percent: The mass of a component divided by the total mass of the mixture, multiplied by 100%.
A mass percent statement implies a direct proportionality between each component’s mass and the mixture’s total mass.
= mass of a specific substance in the mixture (g)
= total mass of the mixture (g)
If you are given mass fraction (rather than percent), treat it as the same relationship without the factor of 100.
Converting mass information into chemical amounts
Mass data alone does not tell you how many particles are present, because different substances have different molar masses.

OpenStax Figure 3.5 compares samples of different elements where each sample contains exactly 1.00 mol (the same number of atoms), but the masses are different. The figure makes the macroscopic–microscopic link explicit: equal moles imply equal particle counts, while differing molar masses produce differing masses for the same mole amount. This is the conceptual reason mixture problems must convert grams to moles before forming particle ratios. Source
To connect composition-by-mass to particle counts, convert each component’s mass into moles.
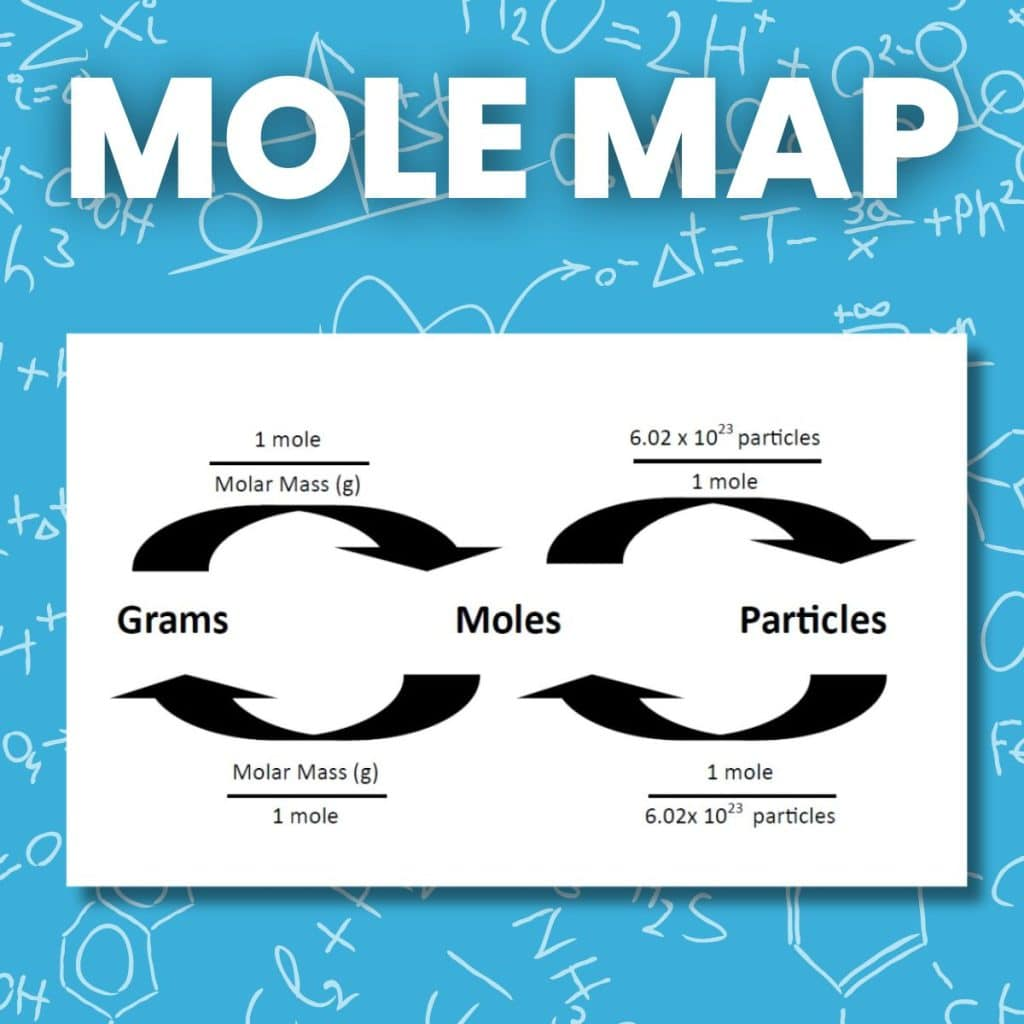
A “mole map” conversion diagram showing the allowed pathways between mass (g), amount (mol), and particle count (entities). The arrows encode the two key conversion factors: molar mass (g/mol) and Avogadro’s number ( entities/mol). This is a visual workflow for ensuring particle ratios are computed from moles rather than directly from masses. Source
= amount of substance (mol)
= sample mass (g)
= molar mass (g mol)
Once each component’s moles are found, you can compare components on a particle basis (for molecules, ions, or formula units).
Connecting mixture composition to particle ratios
“Particle ratios” are mole ratios, because moles are proportional to numbers of entities. After converting each component to moles, form ratios such as and simplify if requested (often to compare relative numbers of particles or relative reacting amounts).
When a mixture is analysed via a chemical change
Sometimes a mixture is characterised by converting one component selectively into a measurable product (e.g., a precipitate, a gas, or a mass change). The essential logic stays within mixture composition:
Use the measured mass (or other quantity) of the product to infer the moles of that product.
Use the reaction stoichiometry (given in the prompt) to infer moles of the original component.
Convert those moles back to mass to determine how much of that component was in the mixture.
Use the total mixture mass to determine the remaining component mass by difference.
This approach is still “composition-by-mass” because the final goal is the mass (and often percent by mass) of each mixture component, justified by particle ratios through moles.
High-utility workflow (no matter the data format)
Identify what is known: total mass, one component’s mass/percent, or a measurable product linked to a component.
Write expressions for unknown component masses that must sum to the total mass.
Convert any necessary masses to moles to connect to particle ratios.
If a reaction is involved, use the provided mole ratio (stoichiometric coefficients) to relate component moles to product moles.
Check internal consistency: component masses must be positive and sum to the mixture mass; significant figures should match measured data.
Common pitfalls to avoid
Mixing up mass percent and mole (particle) percent; they are generally not the same.
Forgetting to convert percent to a decimal (or vice versa) before solving.
Using the wrong molar mass (especially for ionic compounds vs molecular formulas).
Reporting a particle ratio from masses without converting to moles first.
FAQ
Treat “$x$ g per 100 g” as $x%$ by mass.
Use the given values as-is and carry appropriate significant figures; minor mismatch is usually rounding.
Yes; multiply each amount in moles by $N_A$ to get counts. The ratio is unchanged because both are scaled by the same constant.
Use the exact formula provided for that component when finding $M$; otherwise you will convert mass to the wrong mole amount.
Compare $m/M$ values: a larger $M$ should give fewer moles for the same mass, and the component with smaller $M$ often dominates the particle ratio.
Practice Questions
(2 marks) A 25.0 g mixture contains 40.0% by mass of substance X and 60.0% by mass of substance Y. Determine the mass of X and the mass of Y.
(1)
(1)
(5 marks) A 10.0 g mixture contains substances A and B. It is known that 6.00 g of A are present. The molar mass of A is and the molar mass of B is .
(a) Determine the mass of B.
(b) Determine the moles of A and B.
(c) Determine the simplest whole-number ratio of particles (moles) in the mixture.
(a) (1)
(b) (1)
(b) (1)
(c) Ratio (1)
(c) Simplest ratio (1)

