AP Syllabus focus: ‘Atoms contain negatively charged electrons and a positively charged nucleus made of protons and neutrons.’
Understanding subatomic particles explains why atoms are electrically neutral (or become ions) and why nearly all atomic mass is concentrated in a tiny nucleus. These ideas support later topics like formulas and reactions.
The basic structure of an atom
Atoms are composed of a dense central nucleus surrounded by electrons in the space around the nucleus.
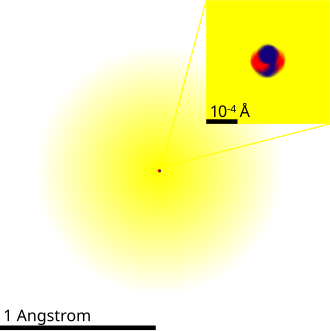
Schematic depiction of an atom in which the electron probability cloud surrounds a compact nucleus. The nucleus is explicitly shown as protons (red) and neutrons (blue), emphasizing that nearly all mass resides in the nucleus while electrons occupy most of the atom’s volume. Source
The nucleus contains protons and neutrons, which together are called nucleons.
Proton, neutron, and electron properties
Proton: A subatomic particle in the nucleus with a charge of (in elementary charge units) and a mass of about amu.
A proton’s count determines the identity of the element.
Neutron: A subatomic particle in the nucleus with a charge of and a mass of about amu.
Neutrons strongly affect atomic mass and nuclear stability without changing the element.
Electron: A subatomic particle outside the nucleus with a charge of and a very small mass (about amu, often treated as negligible for mass calculations).
Electrons largely determine how atoms interact chemically, but in this subtopic the key idea is their negative charge.
Charge: neutrality and ions
An atom is neutral when it has equal numbers of protons and electrons, so the total positive and negative charges cancel. If electrons are gained or lost, the atom becomes an ion with a net charge.
= number of protons (unitless count)
= number of electrons (unitless count)
= elementary charge magnitude,
= net charge of the ion, in coulombs (C)
In AP Chemistry, it is often sufficient to report charge in elementary charge units (for example, or ) rather than coulombs.
Key implications of charge
Protons contribute positive charge and essentially all of an atom’s identity.
Electrons contribute negative charge and can change in number when ions form.
Neutrons do not change charge; they change mass.
Counting subatomic particles with nuclear notation
Nuclear symbols are commonly written as , where:
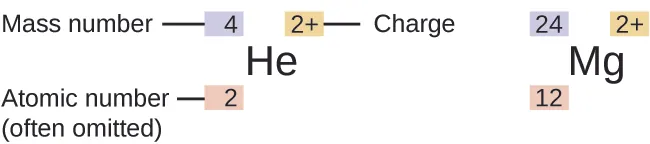
Diagram showing how isotope/ion symbols encode nuclear information: mass number as a left superscript, atomic number as a left subscript (often omitted in practice), and charge as a right superscript. This directly supports using and to determine particle counts (protons and neutrons ). Source
is the atomic number (the number of protons).
is the mass number (the number of protons plus neutrons).
Atomic number (): The number of protons in the nucleus of an atom; it uniquely identifies the element.
Because fixes the element, changing the number of protons changes the element itself.
Mass number (): The total number of protons and neutrons in the nucleus of a specific atom.
From these ideas:
number of protons
number of neutrons
for a neutral atom, number of electrons
for an ion, number of electrons differs from by the ion’s charge (electrons lost positive charge; electrons gained negative charge)
Isotopes: same element, different mass
Atoms of the same element can have different numbers of neutrons.
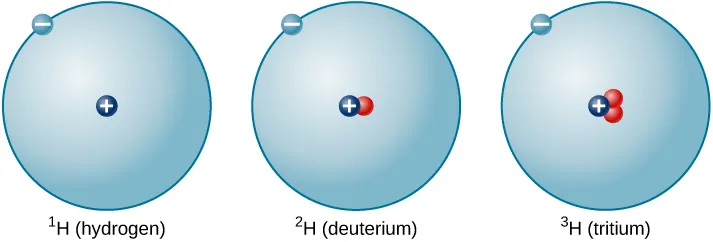
Side-by-side schematic of hydrogen isotopes showing that each isotope has one proton (so the element identity is unchanged) while the number of neutrons varies. This makes the definition of isotope visually concrete: same , different due to different neutron counts. Source
Isotope: Atoms of the same element (same ) that have different numbers of neutrons (different ), and therefore different masses.
Isotopes have the same proton count (so they are the same element) and, when neutral, the same electron count, but differ in neutron count and mass.
FAQ
Protons and neutrons each have a mass close to $1$ amu, while an electron’s mass is about $1/1836$ of a proton’s.
Even though electrons take up most of the atom’s volume, they contribute negligibly to its mass.
Elementary charge, $e$, is the fixed magnitude of charge carried by a single proton ($+e$) or electron ($-e$).
Charge is quantised because particles carry integer multiples of $e$, so observable net charges come in whole-number steps (e.g., $2+$, $1-$).
Electrons are not nucleons; the nucleus is composed of protons and neutrons.
Confining an electron to nuclear dimensions would require extreme conditions and leads to physics beyond the scope of AP Chemistry; chemically, electrons occupy regions outside the nucleus.
They change the mass of the atom and can change nuclear stability.
Different neutron counts can alter which isotopes are stable or radioactive, even though the element (proton count) stays the same.
Periodic table atomic masses are weighted averages of naturally occurring isotopes.
Because isotopes have different masses and natural abundances, the average is typically not an integer, even though individual mass numbers ($A$) are whole numbers.
Practice Questions
(2 marks): State the charge and location (nucleus or outside nucleus) of (i) a proton and (ii) an electron.
Proton: and in the nucleus (1)
Electron: and outside the nucleus (1)
(5 marks): An ion of an element is written as . Determine (i) the number of protons, (ii) the number of neutrons, (iii) the number of electrons, and (iv) explain what the charge indicates in terms of electron transfer.
Protons (1)
Neutrons (1)
Electrons (1)
Explains means 3 fewer electrons than protons / loss of 3 electrons (1)
States protons unchanged when forming ions (1)

